Abstract
Purpose
Although development of immune checkpoint inhibitors has revolutionized the treatment of metastatic melanoma, more than a half of treated patients experience disease progression during therapy. Cases of spontaneous vitiligo-like leukoderma have been described in melanoma patients and have been associated with a favorable outcome. This vitiligo-like leukoderma can also appear in melanoma patients undergoing immune therapies such as immune checkpoint inhibitors. However, no consensus exists about the relationship between vitiligo-like leukoderma onset and improved overall survival. Our study investigates the possible association between the onset of vitiligo-like leukoderma during immune checkpoint inhibitor treatment and a better prognosis.
Methods
A non-concurrent cohort study was conducted by identifying retrospectively 280 patients who had inoperable or metastatic melanoma and had undergone immune therapy with checkpoint inhibitors in any line of treatment. Toxicities developed during therapy were evaluated.
Results
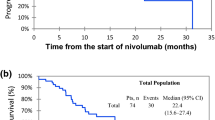
Among the 280 study participants, 50% developed at least one type of toxicity, and vitiligo-like leukoderma was observed in 43 patients (15.4%). In the multivariate Cox model, a protective effect for mortality was observed for patients with vitiligo-like leukoderma development (HR : 0.23; 95% CI 0.11–0.44, p < 0.0001). In a sub-group analysis comprising only cutaneous melanoma in first line of treatment (N = 153), occurrence of vitiligo-like leukoderma was also an independent predictor factor for duration of clinical benefits measured by time to the next treatment (HR: 0.17; 95% CI 0.06–0.44).
Conclusion
Our findings indicate that onset of vitiligo-like leukoderma during melanoma treatment could be a marker of favorable outcome in patients treated with immune checkpoint inhibitors.


Similar content being viewed by others
Availability of data and materials
All relevant data are within the papers.
Abbreviations
- MEK:
-
Mitogen-activated protein kinase
- ICI:
-
Immune checkpoint inhibitors
- CTLA-4:
-
Cytotoxic T-lymphocyte antigen-4
- PD-1:
-
Programmed cell death protein 1
- PFS:
-
Progression free survival
- OS:
-
Overall survival
- TMB:
-
Tumor mutational burden
- VLL:
-
Vitiligo-like leukoderma
- IL:
-
Interleukin
- irAE:
-
Immune-related adverse events
- LDH:
-
Lactate dehydrogenase
- TTNT:
-
Time-to-next treatment
- RECIST:
-
Response Evaluation Criteria in Solid Tumors
- CR:
-
Complete response
- PR:
-
Partial response
- SD:
-
Stable disease
- PD:
-
Progressive disease
- SD:
-
Standard deviation
- IQR:
-
Interquartile range
- HR :
-
Hazard ratio
- PD-L1:
-
PD-ligand-1
References
Babai S, Voisin AL, Bertin C et al (2020) Occurrences and outcomes of immune checkpoint inhibitors-induced vitiligo in cancer patients: a retrospective cohort study. Drug Saf 43:111–117. https://doi.org/10.1007/s40264-019-00875-6
Belum VR, Benhuri B, Postow MA et al (2016) Characterisation and management of dermatologic adverse events to agents targeting the PD-1 receptor. Eur J Cancer 60:12–25. https://doi.org/10.1016/j.ejca.2016.02.010
Barnhill RL, Piepkorn M, Busam KJ (2004) Melanocytes. In: Pathology of melanocytic nevi and malignant melanoma Springer: New York, pp 1–10
Breslow A (1970) Thickness, cross-sectional areas and depth of invasion in the prognosis of cutaneous melanoma. Ann Surg 172:902–908. https://doi.org/10.1097/00000658-197011000-00017
Büttner R, Longshore JW, López-Ríos F et al (2019) Implementing TMB measurement in clinical practice: considerations on assay requirements. ESMO Open 4:e000442
Cancer Institute N (2017) Common Terminology Criteria for Adverse Events (CTCAE) Common Terminology Criteria for Adverse Events (CTCAE) v5.0. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/ctc.htm
Chabanon RM, Pedrero M, Lefebvre C et al (2016) Mutational landscape and sensitivity to immune checkpoint blockers. Clin Cancer Res 22:4309–4321
Clark WH, From L, Bernardino EA, Mihm MC (1969) The histogenesis and biologic behavior of primary human malignant melanomas of the skin. Cancer Res 29:705–727
Clark WH, Elder DE, Guerry D et al (1989) Model predicting survival in stage I melanoma based on tumor progression. J Natl Cancer Inst 81:1893–1904. https://doi.org/10.1093/jnci/81.24.1893
Corcoran CD, Senchaudhuri P, Mehta CR, Patel NR (2005) Exact inference for categorical data. Encycl Biostat. https://doi.org/10.1002/0470011815.b2a10019
Cortellini A, Buti S, Agostinelli V, Bersanelli M (2019) A systematic review on the emerging association between the occurrence of immune-related adverse events and clinical outcomes with checkpoint inhibitors in advanced cancer patients. Semin Oncol 46:362–371
Di Giacomo AM, Valente M, Cerase A et al (2019) Immunotherapy of brain metastases: breaking a “dogma.” J Exp Clin Cancer Res 38:419
Dimitriou F, Mangana J, Dummer R (2020) Hair depigmentation and hair loss in advanced melanoma treated with combined immunotherapy and targeted therapy. Acta Derm Venereol 100:1–2. https://doi.org/10.2340/00015555-3355
Dreno B, Ascierto PA, McArthur GA et al (2018) Efficacy and safety of cobimetinib (C) combined with vemurafenib (V) in patients (pts) with BRAF V600 mutation–positive metastatic melanoma: analysis from the 4-year extended follow-up of the phase 3 coBRIM study. J Clin Oncol 36:9522–9522. https://doi.org/10.1200/jco.2018.36.15_suppl.9522
Eisenhauer EA, Therasse P, Bogaerts J et al (2009) New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 45:228–247. https://doi.org/10.1016/j.ejca.2008.10.026
Failla CM, Carbone ML, Fortes C et al (2019) Melanoma and vitiligo: in good company. Int J Mol Sci. https://doi.org/10.3390/ijms20225731
Gambichler T, Seifert C, Lehmann M et al (2020) Concurrent Vogt-Koyanagi-Harada disease and impressive response to immune checkpoint blockade in metastatic melanoma. Immunotherapy 12:439–444. https://doi.org/10.2217/imt-2019-0206
Gershenwald JE, Scolyer RA (2018) Melanoma Staging: American Joint Committee on Cancer (AJCC) 8th Edition and Beyond. Ann Surg Oncol 25:2105–2110
Goodman AM, Kato S, Bazhenova L et al (2017) Tumor mutational burden as an independent predictor of response to immunotherapy in diverse cancers. Mol Cancer Ther 16:2598–2608. https://doi.org/10.1158/1535-7163.MCT-17-0386
Guida M, Strippoli S, Maule M et al (2021) Immune checkpoint inhibitor associated vitiligo and its impact on survival in patients with metastatic melanoma: an Italian Melanoma Intergroup study. ESMO Open 6:100064. https://doi.org/10.1016/j.esmoop.2021.100064
Hua C, Boussemart L, Mateus C et al (2016) Association of vitiligo with tumor response in patients with metastatic melanoma treated with pembrolizumab. JAMA Dermatol 152:45–51. https://doi.org/10.1001/jamadermatol.2015.2707
Hwang SJE, Park JJW, Wakade D et al (2019) Cutaneous adverse events of anti-programmed death 1 antibodies combined with anti-cytotoxic T-lymphocyte-Associated protein 4 therapy use in patients with metastatic melanoma. Melanoma Res 29:172–177. https://doi.org/10.1097/CMR.0000000000000518
Keung EZ, & Gershenwald JE, (2020) Clinicopathological Features, Staging, and Current Approaches to Treatment in High-Risk Resectable Melanoma, J. Natl. Cancer Inst 112(9):875–885. https://doi.org/10.1093/jnci/djaa012
Klein SL, Flanagan KL (2016) Sex differences in immune responses. Nat Rev Immunol 16:626–638
Larkin J, Chiarion-Sileni V, Gonzalez R et al (2015) Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N Engl J Med 373:23–34. https://doi.org/10.1056/nejmoa1504030
León-Letelier RA, Bonifaz LC, Fuentes-Pananá EM (2019) OMIC signatures to understand cancer immunosurveillance and immunoediting: melanoma and immune cells interplay in immunotherapy. J Leukoc Biol 105:915–933
Mason CP, Gawkrodger DJ (2005) Vitiligo presentation in adults. Clin Exp Dermatol 30:344–345. https://doi.org/10.1111/j.1365-2230.2005.01779.x
Maverakis E, Cornelius LA, Bowen GM et al (2015) Metastatic melanoma—a review of current and future treatment options. Acta Derm Venereol 95:516–524
Nakamura Y, Tanaka R, Asami Y et al (2017) Correlation between vitiligo occurrence and clinical benefit in advanced melanoma patients treated with nivolumab: a multi-institutional retrospective study. J Dermatol 44:117–122. https://doi.org/10.1111/1346-8138.13520
Parmiani G (2001) Melanoma antigens and their recognition by T cells. Keio J Med 50:86–90
Robert C, Grob JJ, Stroyakovskiy D et al (2019a) Five-year outcomes with dabrafenib plus trametinib in metastatic melanoma. N Engl J Med 381:626–636. https://doi.org/10.1056/nejmoa1904059
Robert C, Ribas A, Schachter J et al (2019b) Pembrolizumab versus ipilimumab in advanced melanoma (KEYNOTE-006): post-hoc 5-year results from an open-label, multicentre, randomised, controlled, phase 3 study. Lancet Oncol 20:1239–1251. https://doi.org/10.1016/S1470-2045(19)30388-2
Rodrigues M, Ezzedine K, Hamzavi I et al (2017) New discoveries in the pathogenesis and classification of vitiligo. J Am Acad Dermatol 77:1–13. https://doi.org/10.1016/j.jaad.2016.10.048
Simeone E, Grimaldi AM, Festino L et al (2019) Immunotherapy in metastatic melanoma: a novel scenario of new toxicities and their management. Melanoma Manag 6:MMT30. https://doi.org/10.2217/mmt-2019-0005
Smalley KSM (2020) Two worlds collide: Unraveling the role of the immune system in Braf–Mek inhibitor responses. Cancer Discov 10:176–178. https://doi.org/10.1158/2159-8290.CD-19-1441
Teulings HE, Willemsen KJ, Glykofridis I et al (2014) The antibody response against MART-1 differs in patients with melanoma-associated leucoderma and vitiligo. Pigment Cell Melanoma Res 27:1086–1096. https://doi.org/10.1111/pcmr.12294
Teulings HE, Limpens J, Jansen SN et al (2015) Vitiligo-like depigmentation in patients with stage III-IV melanoma receiving immunotherapy and its association with survival: a systematic review and meta-analysis. J Clin Oncol 33:773–781. https://doi.org/10.1200/JCO.2014.57.4756
Tumeh PC, Hellmann MD, Hamid O et al (2017) Liver metastasis and treatment outcome with anti-PD-1 monoclonal antibody in patients with melanoma and NSCLC. Cancer Immunol Res 5:417–424. https://doi.org/10.1158/2326-6066.CIR-16-0325
Zhao CY, Chou S, Liu RC, Fernandez-Peñas P (2018) Naevus lightening in melanoma patients under BRAF/MEK inhibitor combination therapy versus checkpoint immunotherapy: a histological and immunohistochemistry analysis. Pigment Cell Melanoma Res 31:341–344. https://doi.org/10.1111/pcmr.12669
Zitvogel L, Perreault C, Finn OJ, Kroemer G (2021) Beneficial autoimmunity improves cancer prognosis. Nat Rev Clin Oncol 18:591–602. https://doi.org/10.1038/s41571-021-00508-x
Acknowledgements
This project was funded, in part, with grants from the “Progetto Ricerca Corrente”, RC20_4.1, of the Italian Ministry of Health. MLC is the recipient of a Young Investigator Fellowship from Fondazione Umberto Veronesi. Authors would like to acknowledge the efforts and care of the Oncology Department nurse team, and the contribution and compliance of the patients.
Funding
This project was funded, in part, with grants from the “Progetto Ricerca Corrente”, RC20_4.1, of the Italian Ministry of Health. MLC is the recipient of a Young Investigator Fellowship from Fondazione Umberto Veronesi.
Author information
Authors and Affiliations
Contributions
Conceptualization: SD, FDG, CMF, and CF; data collection and management: SV, FRDP, SM, MLC, RM, and FMM; formal analysis: SV, FRDP, SM, and MLC; methodology: SV, FRDP, SM, MLC, and CF; validation: SM, CF; writing original draft: SV, FRDP, MLC, CMF, and CF; writing review and editing: DA, RM, FMM, SD, PM, FDG, CMF, and CF; funding acquisition: CMF, CF, DA, FDG, and PM.
Corresponding author
Ethics declarations
Conflicts of interest
No competing interests.
Ethics approval and consent to participate
The protocol was approved by the IDI-IRCCS Institutional Ethical Committee (n. 510/3, 2018).
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Verkhovskaia, S., Di Pietro, F.R., Mastroeni, S. et al. Vitiligo-like leukoderma as an indicator of clinical response to immune checkpoint inhibitors in late-stage melanoma patients. J Cancer Res Clin Oncol 148, 2529–2538 (2022). https://doi.org/10.1007/s00432-021-03811-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03811-3