Abstract
Background
Checkpoint inhibitor therapy (CPI) has significantly changed therapy in non-small cell lung cancer (NSCLC) in recent years. There are some data that the effect of CPI therapy is influenced by the microbiome. Little is known about the influence and timing of antimicrobial therapy (AMT) on the microbiome-mediated effect on CPI therapy.
Patients and methods
We retrospectively analysed 70 patients (age 68 ± 9.2 years) with NSCLC stage IV. Patients were treated according to the guidelines with either CPI alone (pembrolizumab, nivolumab, atezolizumab) or chemotherapy (platin doublet or docetaxel/nintedanib or pemetrexed). We registered patient’s characteristics including presence and timing of AMT. Group 1 consisted of 27 patients with AMT in the month before CPI- or chemotherapy, group 2 was 30 patients with AMT during CPI- or chemotherapy, and group 3 was 43 patients without AMT.
Results
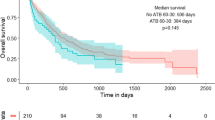
Groups 1–3 showed comparable patient characteristics. Using cox-regression analysis, we found that AMT in the month before CPI resulted in a decreased progression-free survival (PFS) compared to patients with CPI and no AMT (14 ± 1.56 vs. 5 ± 0.99, p = 0.005, 95% CI: 0.13–0.67). In patients, who were treated with chemotherapy alone, there was no difference in PFS in those with or without AMT in the month before therapy (5 ± 0.99 vs. 6 ± 0.81 months, p = 0.3). Interestingly, AMT during chemotherapy or CPI therapy showed no effect on PFS.
Conclusions
In a real-life setting, we found that AMT reduces PFS when given in the month before CPI therapy. AMT before chemotherapy and during CPI and chemotherapy seems not to influence PFS. The best PFS was seen in patients without AMT before CPI therapy. This implies the need for an even more restrictive use of AMT in the context of patients with NSCLC stage IV disease.

Similar content being viewed by others
References
Chalabi M, Cardona A, Nagarkar DR et al (2020) Efficacy of chemotherapy and atezolizumab in patients with non-small-cell lung cancer receiving antibiotics and proton pump inhibitors: pooled post hoc analyses of the OAK and POPLAR trials. Ann Oncol 31(4):525–531
Hekmatshoar Y, Rahbar Saadat Y, Hosseiniyan Khatibi SM et al (2019) The impact of tumor and gut microbiotas on cancer therapy: beneficial or detrimental? Life Sci 233:116680
Moreno-Indias I, Cardona F, Tinahones FJ et al (2014) Impact of the gut microbiota on the development of obesity and type 2 diabetes mellitus. Front Microbiol 5:190
Pflug N, Kluth S, Vehreschild JJ et al (2016) Efficacy of antineoplastic treatment is associated with the use of antibiotics that modulate intestinal microbiota. Oncoimmunology 5(6):e1150399
Vetizou M, Pitt JM, Daillere R et al (2015) Anticancer immunotherapy by CTLA-4 blockade relies on the gut microbiota. Science 350(6264):1079–1084
Funding
There was no funding.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors have read the manuscript and agreed to publication. No author has got conflicts of interest.
Ethical approval
According to an ethics committee, approval is not needed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Uhlenbruch, M., Krüger, S. Effect of antimicrobial therapy on progression-free survival of patients with non-small cell lung cancer receiving checkpoint inhibitor- and chemotherapy. J Cancer Res Clin Oncol 148, 2079–2082 (2022). https://doi.org/10.1007/s00432-021-03734-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03734-z