Abstract
Purpose
To compare the efficacy and safety of high dose-intensity combination of methotrexate, vinblastine, adriamycin and cisplatin (HD MVAC) with gemcitabine plus cisplatin (GC) as a neoadjuvant chemotherapy (NAC) in muscle-invasive bladder cancer (MIBC) or locally advanced upper tract urothelial cancer (UTUC).
Patients and methods
A retrospective analysis was conducted for patients with UC (cT2-4aN0-1M0) who received NAC from January 2011 and December 2017 at Asan Medical Center. Pathologic complete response (pCR), down-staging (< ypT2 and no N upstaging), disease-free survival (DFS), OS and safety were compared for each regimen.
Results
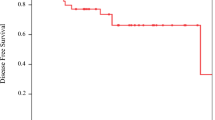
Out of a total of 277 patients, 176 patients received GC and 41 patients received HD MVAC. With the exception of age (patients receiving HD MVAC were younger; p = 0.002), other baseline characteristics were well balanced between groups. pCR rates were 27.0% for GC and 22.6% for HD MVAC (p = 0.62), and down-staging rate was 50.8% for GC and 58.1% for HD MVAC (p = 0.47). There were no differences in OS (72.1% vs 73.1% for GC vs HD MVAC; p = 0.58) and DFS (54.9% vs 63.3% for GC vs HD MVAC; p = 0.21) at 3 years. HD MVAC with prophylactic G-CSF was associated with a higher incidence of febrile neutropenia (p < 0.001) than GC. The NAC regimen was not an independent prognostic factor for OS.
Conclusion
Oncologic outcomes were not significantly different between the GC and HD MVAC when used as NAC in MIBC/UTUC.

Similar content being viewed by others
References
Advanced Bladder Cancer Meta-analysis Collaboration (2003) Neoadjuvant chemotherapy in invasive bladder cancer: a systematic review and meta-analysis. Lancet 361:1927–1934. https://doi.org/10.1016/s0140-6736(03)13580-5
Alfred Witjes J et al (2017) Updated EAU guidelines on muscle-invasive and metastatic bladder cancer. Eur Urol 71:462–475. https://doi.org/10.1016/j.eururo.2016.06.020
Birtle A et al (2020) Adjuvant chemotherapy in upper tract urothelial carcinoma (the POUT trial): a phase 3, open-label, randomised controlled trial. Lancet 395:1268–1277. https://doi.org/10.1016/S0140-6736(20)30415-3
Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A (2018) Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 68:394–424. https://doi.org/10.3322/caac.21492
Chakiryan N, Martinez A, Gao L, Liu JJ, Amling C, Garzotto M, Kopp RP (2019) Optimizing the sequence of chemotherapy for upper tract urothelial carcinoma with clinically positive regional lymph nodes. J Urol 202:76–82. https://doi.org/10.1097/JU.0000000000000172
Chang SS et al (2017) Treatment of non-metastatic muscle-invasive bladder cancer: AUA/ASCO/ASTRO/SUO guideline. J Urol 198:552–559. https://doi.org/10.1016/j.juro.2017.04.086
Choueiri TK et al (2014) Neoadjuvant dose-dense methotrexate, vinblastine, doxorubicin, and cisplatin with pegfilgrastim support in muscle-invasive urothelial cancer: pathologic, radiologic, and biomarker correlates. J Clin Oncol 32:1889–1894. https://doi.org/10.1200/jco.2013.52.4785
Flaig TW et al (2019) SWOG S1314: A randomized phase II study of co-expression extrapolation (COXEN) with neoadjuvant chemotherapy for localized, muscle-invasive bladder cancer. J Clin Oncol 37:4506–4506. https://doi.org/10.1200/JCO.2019.37.15_suppl.4506
Grossman HB et al (2003) Neoadjuvant chemotherapy plus cystectomy compared with cystectomy alone for locally advanced bladder cancer. N Engl J Med 349:859–866. https://doi.org/10.1056/NEJMoa022148
Leow JJ, Martin-Doyle W, Fay AP, Choueiri TK, Chang SL, Bellmunt J (2014) A systematic review and meta-analysis of adjuvant and neoadjuvant chemotherapy for upper tract urothelial carcinoma. Eur Urol 66:529–541. https://doi.org/10.1016/j.eururo.2014.03.003
National Cancer Institute (2010) Common Terminology Criteria for Adverse Events (CTCAE):Version 4.03; 2020. https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/CTCAE_4.03.xlsx. Accessed 1 July 2020
National Comprehensive Cancer Network (2020) Bladder Cancer (version 6.2020); 2021. https://www.nccn.org/professionals/physician_gls/pdf/bladder.pdf. Accessed 15 Jan 2021
Peyton CC et al (2018) Downstaging and survival outcomes associated with neoadjuvant chemotherapy regimens among patients treated with cystectomy for muscle-invasive bladder cancer. JAMA Oncol 4:1535–1542. https://doi.org/10.1001/jamaoncol.2018.3542
Pfister C et al (2021) Randomized phase III trial of dose-dense methotrexate, vinblastine, doxorubicin, and cisplatin, or gemcitabine and cisplatin as perioperative chemotherapy for patients with muscle-invasive bladder cancer. Analysis of the GETUG/AFU V05 VESPER trial secondary endpoints: chemotherapy toxicity and pathological responses. Eur Urol 79:214–221. https://doi.org/10.1016/j.eururo.2020.08.024
Sternberg CN et al (2001) Randomized phase III trial of high-dose-intensity methotrexate, vinblastine, doxorubicin, and cisplatin (MVAC) chemotherapy and recombinant human granulocyte colony-stimulating factor versus classic MVAC in advanced urothelial tract tumors: European Organization for Research and Treatment of Cancer Protocol no. 30924. J Clin Oncol 19:2638–2646. https://doi.org/10.1200/jco.2001.19.10.2638
Sternberg CN et al (2006) Seven year update of an EORTC phase III trial of high-dose intensity M-VAC chemotherapy and G-CSF versus classic M-VAC in advanced urothelial tract tumours. Eur J Cancer 42:50–54. https://doi.org/10.1016/j.ejca.2005.08.032
van de Putte EE et al (2016) Neoadjuvant induction dose-dense MVAC for muscle invasive bladder cancer: efficacy and safety compared with classic MVAC and gemcitabine/cisplatin. World J Urol 34:157–162. https://doi.org/10.1007/s00345-015-1636-y
von der Maase H et al (2000) Gemcitabine and cisplatin versus methotrexate, vinblastine, doxorubicin, and cisplatin in advanced or metastatic bladder cancer: results of a large, randomized, multinational, multicenter, phase III study. J Clin Oncol 18:3068–3077
Zargar H et al (2018) Neoadjuvant dose dense MVAC versus gemcitabine and cisplatin in patients with cT3–4aN0M0 bladder cancer treated with radical cystectomy. J Urol 199:1452–1458. https://doi.org/10.1016/j.juro.2017.12.062
Funding
Nothing to be declared.
Author information
Authors and Affiliations
Contributions
Dr. YJ Lee and JL Lee had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: YJ Lee, JL Lee. Acquisition, analysis, or interpretation of data: YJ Lee, JL Lee, YS Kim, BS Hong, YM Cho, JL Lee. Drafting of the manuscript: YJ Lee, JL Lee. Critical revision of the manuscript for important intellectual content: YJ Lee, JL Lee, YS Kim, BS Hong, YM Cho, JL Lee. Statistical analysis: YJ Lee, JL Lee. Administrative, technical, or material support: YJ Lee, JL Lee, YS Kim, BS Hong, YM Cho, JL Lee. Data analysis: YJ Lee, JL Lee.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no relevant conflict of interests.
Consent for publication
All authors consent to publish this study in J Cancer Res Clin Oncol.
Ethics approval
IRB of Asan Medical Center reviewed and approved this study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lee, Y., Kim, Y.S., Hong, B. et al. Comparison of clinical outcomes in patients with localized or locally advanced urothelial carcinoma treated with neoadjuvant chemotherapy involving gemcitabine–cisplatin and high dose-intensity MVAC. J Cancer Res Clin Oncol 147, 3421–3429 (2021). https://doi.org/10.1007/s00432-021-03582-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03582-x