Abstract
Introduction
Proper management of the clinically involved neck in OSCC patients continues to be a matter of debate. Our aim was to analyze the accuracy of computerized tomography (CT) and ultrasound (US) in anticipating the exact location of lymph node (LN) metastases of OSCC patients across the AAO-HNS (American Academy of Otolaryngology—Head and Neck Surgery) levels ipsi- and contralaterally. Furthermore, we wanted to assess the suitability of therapeutic selective neck dissection (SND) in patients with one or two ipsilateral positive nodes upon clinical staging (cN1/cN2a and cN2b(2/x) patients).
Methods
We prospectively analyzed the LN status of patients with primary OSCC using CT and US from 2007 to 2013. LNs were individually assigned to a map containing the AAO-HNS levels; patients bearing a single or just two ipsilateral positive nodes (designated cN1/cN2a or cN2b(2/x) patients either by CT (CT group) or US alone (US group) or in a group combining findings of CT and US (CTUS group)) received an ipsi-ND (I–V) and a contra-ND (I–IV). 78% of the LNs were sent individually for routine histopathological examination; the remaining were dissected and analyzed per neck level.
Results
Upon the analysis of 1.670 LNs of 57 patients, the exact location of pathology proven LN metastases in cN1 patients was more precisely predicted by US compared to CT with confirmed findings only in levels IA, IB und IIA. Clearly decreasing the number of missed lesions, the findings in the CTUS group nearly kept the spatial reliability of the US group. The same analysis for patients with exactly two supposed ipsilateral lesions (cN2b(2/x)) yielded confirmed metastases from levels I to V for both methods individually and in combination and, therefore, render SND insufficient for these cases.
Conclusion
Our findings stress the importance of conducting both, CT and US, in patients with primary OSCC. Only the combination of their findings warrants the application of therapeutic SND in patients with a single ipsilateral LN metastasis (cN1/cN2a patients) but not in patients with more than one lesion upon clinical staging (≥ cN2b).






Similar content being viewed by others
Notes
The 2 indicates the 2 positive LNs and the x stands for the total number of LN seen per examination and varies from patient to patient and between the modalities, but was not further analyzed here.
References
Bessell A et al (2011) Interventions for the treatment of oral and oropharyngeal cancers: surgical treatment. Cochrane Database Syst Rev. https://doi.org/10.1002/14651858.CD006205.pub3
Chow TL, Chow TK, Chan TT, Yu NF, Fung SC, Lam SH (2004) Contralateral neck recurrence of squamous cell carcinoma of oral cavity and oropharynx. J Oral MaxillofacSurg 62:1225–1228
D’Cruz AK et al (2015) Elective versus therapeutic neck dissection in node-negative oral cancer. N Engl J Med 373:521–529. https://doi.org/10.1056/NEJMoa1506007
de Bree R et al (2015) Advances in diagnostic modalities to detect occult lymph node metastases in head and neck squamous cell carcinoma. Head Neck 37:1829–1839. https://doi.org/10.1002/hed.23814
Dziegielewski PT, O’Connell DA, Szudek J, Barber B, Joshi A, Harris JR, Seikaly H (2013) Neck metastases in oropharyngeal cancer: necessity and extent of bilateral treatment. Head Neck 35:1461–1467. https://doi.org/10.1002/hed.23172
Feng Z, Li JN, Niu LX, Guo CB (2014) Supraomohyoid neck dissection in the management of oral squamous cell carcinoma: special consideration for skip metastases at level IV or V. J Oral MaxillofacSurg 72:1203–1211. https://doi.org/10.1016/j.joms.2013.12.008
Ferlito A et al (2011) Proposal for a rational classification of neck dissections. Head Neck 33:445–450. https://doi.org/10.1002/hed.21614
Furukawa M, Dillon JK, Futran ND, Anzai Y (2014) The prevalence of lymph node metastases in clinically N0 necks with oral cavity squamous cell carcinoma: is CT good enough for nodal staging? Acta Radiologica (Stockholm, Sweden: 1987) 55:570–578. https://doi.org/10.1177/0284185113499326
Heusch P et al (2014) Diagnostic accuracy of ultrasound, (1)(8)F-FDG-PET/CT, and fused (1)(8)F-FDG-PET-MR images with DWI for the detection of cervical lymph node metastases of HNSCC. Clin Oral Investig 18:969–978. https://doi.org/10.1007/s00784-013-1050-z
Ho TY, Chao CH, Chin SC, Ng SH, Kang CJ, Tsang NM (2020) Classifying neck lymph nodes of head and neck squamous cell carcinoma in MRI images with radiomic features. J Digit Imaging 33:613–618. https://doi.org/10.1007/s10278-019-00309-w
Hohlweg-Majert B, Metzger MC, Voss PJ, Holzle F, Wolff KD, Schulze D (2009) Preoperative cervical lymph node size evaluation in patients with malignant head/neck tumors: comparison between ultrasound and computer tomography. J Cancer Res Clin Oncol 135:753–759. https://doi.org/10.1007/s00432-008-0487-y
Inoue H et al (2006) Quality of life after neck dissection. Arch Otolaryngol Head Neck Surg 132:662–666. https://doi.org/10.1001/archotol.132.6.662
Kohler HF, Kowalski LP (2012) Prognostic impact of the level of neck metastasis in oral cancer patients. Braz J Otorhinolaryngol 78:15–20
Kowalski LP, Carvalho AL (2002) Feasibility of supraomohyoid neck dissection in N1 and N2a oral cancer patients. Head Neck 24:921–924. https://doi.org/10.1002/hed.10127
Kuno H et al (2019) CT texture analysis of cervical lymph nodes on contrast-enhanced [(18)F] FDG-PET/CT images to differentiate nodal metastases from reactive lymphadenopathy in HIV-positive patients with head and neck squamous cell carcinoma. Am J Neuroradiol 40:543–550. https://doi.org/10.3174/ajnr.A5974
Kuntz AL, Weymuller EA Jr (1999) Impact of neck dissection on quality of life. Laryngoscope 109:1334–1338. https://doi.org/10.1097/00005537-199908000-00030
Laverick S, Lowe D, Brown JS, Vaughan ED, Rogers SN (2004) The impact of neck dissection on health-related quality of life. Arch Otolaryngol Head Neck Surg 130:149–154. https://doi.org/10.1001/archotol.130.2.149
Liang L, Zhang T, Kong Q, Liang J, Liao G (2015) A meta-analysis on selective versus comprehensive neck dissection in oral squamous cell carcinoma patients with clinically node-positive neck. Oral Oncol 51:1076–1081. https://doi.org/10.1016/j.oraloncology.2015.10.005
Lim YC, Koo BS, Lee JS, Choi EC (2006) Level V lymph node dissection in oral and oropharyngeal carcinoma patients with clinically node-positive neck: is it absolutely necessary? Laryngoscope 116:1232–1235. https://doi.org/10.1097/01.mlg.0000224363.04459.8b
McLean T, Kerr SJ, Giddings CEB (2017) Prophylactic dissection of level V in primary mucosal SCC in the clinically N positive neck: a systematic review. Laryngoscope 127:2074–2080. https://doi.org/10.1002/lary.26573
NCCN (2018) National comprehensive cancer network: head and neck cancers, Version 1.2018, NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®), https://www.nccn.org/professionals/physician_gls/pdf/head-and-neck.pdf
Ohtawa T, Katagiri M, Harada T (1998) A study of sternocleidomastoid muscular atrophy after modified neck dissection. Surg Today 28:46–58
Oikawa SI et al (2018) Association between contrast-enhanced ultrasonography and histopathological findings of the metastatic lymph nodes of patients with head and neck cancer: a preliminary study. Oncol Lett 15:4171–4176. https://doi.org/10.3892/ol.2018.7835
Pellitteri PK, Robbins KT, Neuman T (1997) Expanded application of selective neck dissection with regard to nodal status. Head Neck 19:260–265
Pfister DG et al (2015) Head and neck cancers, version 1.2015. J Natl ComprCancNetw 13:847–855 (quiz 856)
Robbins KT et al (2002) Neck dissection classification update: revisions proposed by the American Head and Neck Society and the American Academy of Otolaryngology-Head and Neck Surgery. Arch Otolaryngol Head Neck Surg 128:751–758
Robbins KT et al (2008) Consensus statement on the classification and terminology of neck dissection. Arch Otolaryngol Head Neck Surg 134:536–538. https://doi.org/10.1001/archotol.134.5.536
Rodrigo JP et al (2018) Selective neck dissection in surgically treated head and neck squamous cell carcinoma patients with a clinically positive neck: systematic review. Eur J Surg Oncol. https://doi.org/10.1016/j.ejso.2018.01.003
Seidler M et al (2019) Dual-energy CT texture analysis with machine learning for the evaluation and characterization of cervical lymphadenopathy. Comput Struct Biotechnol J 17:1009–1015. https://doi.org/10.1016/j.csbj.2019.07.004
Shah JP (1990) Patterns of cervical lymph node metastasis from squamous carcinomas of the upper aerodigestive tract. Am J Surg 160:405–409. https://doi.org/10.1016/s0002-9610(05)80554-9
Shin YS, Koh YW, Kim SH, Choi EC (2013) Selective neck dissection for clinically node-positive oral cavity squamous cell carcinoma. Yonsei Med J 54:139–144. https://doi.org/10.3349/ymj.2013.54.1.139
Simental AA Jr, Duvvuri U, Johnson JT, Myers EN (2006) Selective neck dissection in patients with upper aerodigestive tract cancer with clinically positive nodal disease. Ann OtolRhinolLaryngol 115:846–849. https://doi.org/10.1177/000348940611501109
Sobol S, Jensen C, Sawyer W 2nd, Costiloe P, Thong N (1985) Objective comparison of physical dysfunction after neck dissection. Am J Surg 150:503–509
Sobin L, Gospodarowicz L, Wittekind C (2011) TNM classification of malignant tumours, 7th edition edn. Wiley-Blackwell, New York
Som PM, Curtin HD, Mancuso AA (1999) An imaging-based classification for the cervical nodes designed as an adjunct to recent clinically based nodal classifications. Arch Otolaryngol Head Neck Surg 125:388–396
Som PM, Curtin HD, Mancuso AA (2000) Imaging-based nodal classification for evaluation of neck metastatic adenopathy. Am J Roentgenol 174:837–844. https://doi.org/10.2214/ajr.174.3.1740837
Sproll C et al (2017) Immunohistochemical detection of lymph node-DTCs in patients with node-negative HNSCC. Int J Cancer. https://doi.org/10.1002/ijc.30617
Stoeckli SJ, Haerle SK, Strobel K, Haile SR, Hany TF, Schuknecht B (2012) Initial staging of the neck in head and neck squamous cell carcinoma: a comparison of CT PET/CT, and ultrasound-guided fine-needle aspiration cytology. Head Neck 34:469–476. https://doi.org/10.1002/hed.21764
R Development Core Team (2015) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0. http://www.R-project.org
Thomsen JB, Sorensen JA, Grupe P, Karstoft J, Krogdahl A (2005) Staging N0 oral cancer: lymphoscintigraphy and conventional imaging. Acta Radiologica (Stockholm, Sweden: 1987) 46:492–496
Traynor SJ, Cohen JI, Gray J, Andersen PE, Everts EC (1996) Selective neck dissection and the management of the node-positive neck. Am J Surg 172:654–657
Tschammler A, Hahn D (1999) Multivariate analysis of the adjustment of the colour duplex unit for the differential diagnosis of lymph node alterations. EurRadiol 9:1445–1450. https://doi.org/10.1007/s003300050868
Uematsu T, Sano M, Homma K, Shiina M, Kobayashi S (2001) Three-dimensional helical CT of the breast: accuracy for measuring extent of breast cancer candidates for breast conserving surgery. Breast Cancer Res Treat 65:249–257
Weiss MH, Harrison LB, Isaacs RS (1994) Use of decision analysis in planning a management strategy for the stage N0 neck. Arch Otolaryngol Head Neck Surg 120:699–702
Wolff KD, Follmann M, Nast A (2012) The diagnosis and treatment of oral cavity cancer. DtscArztebl Int 109:829–835. https://doi.org/10.3238/arztebl.2012.0829
Wolff K.-D. A-NB, Al-Sharif U, Beck J, Bikowski K, Bissinger O, Böhme P, Bönte-Hieronymus I, Bootz F, Bozzato A, Budach W, Burkhardt A, Danker H, Eberhardt W, Engers K, Fietkau R, Frerich B, Gauler T, Gehrmann-Weide K, Germann G, Giannakopoulos N, Gittler-Hebestreit N, Grötz K, Hertrampf K, Hoffmann J, Horch R, Ihrler S, Kaufmann R, Kehrer A, Keilholz U, Klußmann P, Kolk A, Lell M, Lübbe A, Mantey W, Mischkowski R, Moll R, Nieberler M, Nusser-Müller-Busch R, Pistner H, Paradies K, Rau A, Reichert T, Reinert S, Schilling B, Schliephake H, Schmidt K, Schmitter M, Singer S, Terheyden H, Troost E, Waterboer T, Westhofen M, Weitz J, Wirz S, Wittlinger M, Zöphel K (2019) S3-Leitlinie Diagnostik und Therapie des Mundhöhlenkarzinoms, Langversion 3.01 (Konsultationsfassung), AWMF Register nummer: 007/100OL. Accessed 5 May 2020
Funding
No specific funding was provided for this work.
Author information
Authors and Affiliations
Contributions
KCS, HH and CT wrote this publication based on the data that have been taken prospectively. JH was mainly responsible for the study design. KCS performed the ultrasound examinations. JA and PK did the CT evaluations. KCS and JH provided the lymph node samples. CT reviewed the histopathological diagnoses. SL and KCS took the patient data prospectively. CS was the principle investigator of this study. He corrected and approved the manuscript. WK, HH and CS performed the statistical analyses. JH, HH, JL and LKS corrected and approved this manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors does have any commercial interest to disclose.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
432_2021_3523_MOESM1_ESM.pptx
Flowchart of patients included. All patients were examined with both procedures, CT and US. Depending on the number of suspected metastases (0, 1, 2 or more than 2 ipsilateral or contralateral), they were divided into the groups shown. Patients can be assigned to several groups; hence, the higher total number of patients than were included at all (PPTX 39 KB)
432_2021_3523_MOESM2_ESM.pptx
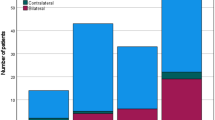
Comparison of the number of suspected metastases and histopathologically confirmed metastases per procedure and patient. x-axis: number of histopathologically confirmed metastases. y-axis: number of individual patients. a Patients in whom the imaging shows exactly one ipsilateral metastasis in CT = blue bars (CT cN1 group) or in ultrasound = green bars (US cN1 group) or in a combination of both methods = yellow bars (CTUS cN1 group). b Patients in whom the imaging shows exactly two ipsilateral metastases on CT = blue bars (CT cN2b(2/x) group) or on ultrasound = green bars (US cN2b(2/x) group) or in a combination of both procedures = yellow bars (CTUS cN2b(2/x) group) a maximum of two ipsilateral metastases was seen. The black boxes frame those patients for whom the imaging findings and the histopathological assessment are matching (PPTX 62 KB)
Rights and permissions
About this article
Cite this article
Sproll, K.C., Leydag, S., Holtmann, H. et al. Is the prediction of one or two ipsilateral positive lymph nodes by computerized tomography and ultrasound reliable enough to restrict therapeutic neck dissection in oral squamous cell carcinoma (OSCC) patients?. J Cancer Res Clin Oncol 147, 2421–2433 (2021). https://doi.org/10.1007/s00432-021-03523-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-021-03523-8