Abstract
Purpose
To investigate the mRNA expression of B-MYB and MDM2 together with their p53 relatedness in clear cell renal cell carcinoma (ccRCC).
Methods
Genes were screened for their mRNA expression from 529 patients in a publicly available ccRCC cohort (TCGA). A cohort of 101 patients with ccRCC served as validation by qRT-PCR mRNA tissue expression analysis.
Results
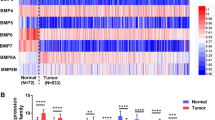
Expression: B-MYB expression was significantly higher in high-grade tumours (p < 0.0001 and p = 0.048) and in advanced stages (p = 0.005 and p = 0.037) in both cohorts.
Correlation: p53-B-MYB as well as MDM2-B-MYB showed significant correlations in local and low-grade ccRCCs, but not in high grade tumours or advanced stages (r < 0.3 and/or p > 0.05).
Survival: Multivariable Cox regression of the TCGA cohort revealed B-MYB upregulation and low MDM2 expression as predictors for an impaired overall survival (OS) (HR 1.97; p = 0.0003; HR 2.94, p < 0.0001) and progression-free survival (PFS) (HR 2.86; p = 0.0005; HR 1.58, p = 0.046). In the validation cohort, the results were confirmed for OS by univariable, but not multivariable regression: high B-MYB expression (HR = 3.05, p = 0.035) and low MDM2 expression (HR 3.81, p value 0.036).
Conclusion
In ccRCC patients with high-grade tumours and advanced stages, high B-MYB expression is common and is associated with poorer OS and PFS. These patients show a loss of their physiological B-MYB–p53 network correlation, suggesting an additional, alternative regulatory, oncogenic mechanism. Assuming further characterization of its signalling pathways, B-MYB could be a potential therapy target for ccRCC.




Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article and its Online Resource.
Code availability
Not applicable.
Abbreviations
- AJCC:
-
American Joint Committee on Cancer
- ccRCC:
-
Clear cell renal cell carcinoma
- FFPE:
-
Formaldehyde and embedded in paraffin
- HR:
-
Hazard ratio
- IQR:
-
Interquartile range
- OS:
-
Overall survival
- PFS:
-
Progression-free survival
- RCC:
-
Renal cell carcinoma
- STRAP:
-
Serine-threonine kinase receptor-associated protein
- UICC:
-
Union for International Cancer Control
- TCGA:
-
The Cancer Genome Atlas
References
Beckerman R, Prives C (2010) Transcriptional regulation by p53. Cold Spring Harb Perspect Biol 2:a000935. https://doi.org/10.1101/cshperspect.a000935
Bykov VJN, Eriksson SE, Bianchi J, Wiman KG (2018) Targeting mutant p53 for efficient cancer therapy. Nat Rev Cancer 18:89–102. https://doi.org/10.1038/nrc.2017.109
Calvisi DF et al (2011) Activation of v-Myb avian myeloblastosis viral oncogene homolog-like2 (MYBL2)-LIN9 complex contributes to human hepatocarcinogenesis and identifies a subset of hepatocellular carcinoma with mutant p53. Hepatology 53:1226–1236. https://doi.org/10.1002/hep.24174
Chen J, Chen X (2018) MYBL2 is targeted by miR-143–3p and regulates breast cancer cell proliferation and apoptosis. Oncol Res 26:913–922. https://doi.org/10.3727/096504017x15135941182107
Dalgliesh GL et al (2010) Systematic sequencing of renal carcinoma reveals inactivation of histone modifying genes. Nature 463:360–363. https://doi.org/10.1038/nature08672
Fan X et al (2018) B-myb mediates proliferation and migration of non-small-cell lung cancer via suppressing IGFBP3. Int J Mol Sci. https://doi.org/10.3390/ijms19051479
Freedman DA, Levine AJ (1998) Nuclear export is required for degradation of endogenous p53 by MDM2 and human papillomavirus E6. Mol Cell Biol 18:7288–7293. https://doi.org/10.1128/mcb.18.12.7288
Gu HY et al (2020) Risk score based on expression of five novel genes predicts survival in soft tissue sarcoma. Aging (Albany NY) 12(4):3807–3827. https://doi.org/10.18632/aging.102847
Gurova KV, Hill JE, Razorenova OV, Chumakov PM, Gudkov AV (2004) p53 pathway in renal cell carcinoma is repressed by a dominant mechanism. Cancer Res 64:1951–1958. https://doi.org/10.1158/0008-5472.can-03-1541
Honda R, Tanaka H, Yasuda H (1997) Oncoprotein MDM2 is a ubiquitin ligase E3 for tumor suppressor p53. FEBS Lett 420:25–27. https://doi.org/10.1016/s0014-5793(97)01480-4
Hsieh JJ et al (2017) Renal cell carcinoma. Nat Rev Dis Primers 3:17009. https://doi.org/10.1038/nrdp.2017.9
Hsieh JJ, Le V, Cao D, Cheng EH, Creighton CJ (2018) Genomic classifications of renal cell carcinoma: a critical step towards the future application of personalized kidney cancer care with pan-omics precision. J Pathol 244:525–537. https://doi.org/10.1002/path.5022
Kriegmair MC et al (2016) Expression of the p53 inhibitors MDM2 and MDM4 as outcome predictor in muscle-invasive bladder cancer. Anticancer Res 36:5205–5213. https://doi.org/10.21873/anticanres.11091
Kriegmair MC et al (2018) Prognostic value of molecular breast cancer subtypes based on Her2, ESR1, PGR and Ki67 mRNA-expression in muscle invasive bladder cancer. Transl Oncol 11:467–476. https://doi.org/10.1016/j.tranon.2018.02.001
Lamont JP, Nemunaitis J, Kuhn JA, Landers SA, McCarty TM (2000) A prospective phase II trial of ONYX-015 adenovirus and chemotherapy in recurrent squamous cell carcinoma of the head and neck (the baylor experience). Ann Surg Oncol 7:588–592. https://doi.org/10.1007/BF02725338
Lang G, Gombert WM, Gould HJ (2005) A transcriptional regulatory element in the coding sequence of the human Bcl-2 gene. Immunology 114:25–36. https://doi.org/10.1111/j.1365-2567.2004.02073.x
Li M et al (2019) Circ_0006332 promotes growth and progression of bladder cancer by modulating MYBL2 expression via miR-143. Aging (Albany NY) 11:10626–10643. https://doi.org/10.18632/aging.102481
Li W, Zhang B, Jia Y, Shi H, Wang H, Guo Q, Li H (2020a) LncRNA LOXL1-AS1 regulates the tumorigenesis and development of lung adenocarcinoma through sponging miR-423–5p and targeting MYBL2. Cancer Med 9:689–699. https://doi.org/10.1002/cam4.2641
Li X et al (2020b) miR-30a inhibits androgen-independent growth of prostate cancer via targeting MYBL2, FOXD1, and SOX4. Prostate. https://doi.org/10.1002/pros.23979
Linehan WM, Ricketts CJ (2019) The Cancer Genome Atlas of renal cell carcinoma: findings and clinical implications. Nat Rev Urol 16:539–552. https://doi.org/10.1038/s41585-019-0211-5
Liu F, Li N, Liu Y, Zhang J, Zhang J, Wang Z (2019) Homeodomain interacting protein kinase-2 phosphorylates FOXM1 and promotes FOXM1-mediated tumor growth in renal cell carcinoma. J Cell Biochem 120:10391–10401. https://doi.org/10.1002/jcb.28323
Mantovani F, Collavin L, Del Sal G (2019) Mutant p53 as a guardian of the cancer cell. Cell Death Differ 26:199–212. https://doi.org/10.1038/s41418-018-0246-9
Mitchell TJ, Rossi SH, Klatte T, Stewart GD (2018) Genomics and clinical correlates of renal cell carcinoma. World J Urol 36:1899–1911. https://doi.org/10.1007/s00345-018-2429-x
Moch H, Cubilla AL, Humphrey PA, Reuter VE, Ulbright TM (2016) The 2016 WHO classification of tumours of the urinary system and male genital organs-part A: renal penile, and testicular tumours. Eur Urol 70:93–105. https://doi.org/10.1016/j.eururo.2016.02.029
Momand J, Wu HH, Dasgupta G (2000) MDM2–master regulator of the p53 tumor suppressor protein. Gene 242:15–29. https://doi.org/10.1016/s0378-1119(99)00487-4
Musa J, Aynaud MM, Mirabeau O, Delattre O, Grunewald TG (2017) MYBL2 (B-Myb): a central regulator of cell proliferation, cell survival and differentiation involved in tumorigenesis. Cell Death Dis 8:e2895. https://doi.org/10.1038/cddis.2017.244
Musa J et al (2019) Cooperation of cancer drivers with regulatory germline variants shapes clinical outcomes. Nat Commun 10:4128. https://doi.org/10.1038/s41467-019-12071-2
Nag S, Qin J, Srivenugopal KS, Wang M, Zhang R (2013) The MDM2-p53 pathway revisited. J Biomed Res 27:254–271. https://doi.org/10.7555/jbr.27.20130030
Ness SA (1996) The Myb oncoprotein: regulating a regulator. Biochim Biophys Acta 1288:F123–139. https://doi.org/10.1016/s0304-419x(96)00027-3
CGAR Network (2013) Comprehensive molecular characterization of clear cell renal cell carcinoma. Nature 499:43–49. https://doi.org/10.1038/nature12222
Pfister NT, Prives C (2017) Transcriptional regulation by wild-type and cancer-related mutant forms of p53. Cold Spring Harb Perspect Med. https://doi.org/10.1101/cshperspect.a026054
Pietsch EC, Humbey O, Murphy ME (2006) Polymorphisms in the p53 pathway. Oncogene 25:1602–1611. https://doi.org/10.1038/sj.onc.1209367
Pitolli C, Wang Y, Mancini M, Shi Y, Melino G, Amelio I (2019) Do mutations turn p53 into an oncogene? Int J Mol Sci. https://doi.org/10.3390/ijms20246241
Qin H, Li Y, Zhang H, Wang F, He H, Bai X, Li S (2019) Prognostic implications and oncogenic roles of MYBL2 protein expression in esophageal squamous-cell carcinoma. Onco Targets Ther 12:1917–1927. https://doi.org/10.2147/ott.S190145
Quaas M, Müller GA, Engeland K (2012) p53 can repress transcription of cell cycle genes through a p21(WAF1/CIP1)-dependent switch from MMB to DREAM protein complex binding at CHR promoter elements. Cell Cycle 11:4661–4672. https://doi.org/10.4161/cc.22917
Ren F et al (2015) MYBL2 is an independent prognostic marker that has tumor-promoting functions in colorectal cancer. Am J Cancer Res 5:1542–1552
Sadasivam S, DeCaprio JA (2013) The DREAM complex: master coordinator of cell cycle-dependent gene expression. Nat Rev Cancer 13:585–595. https://doi.org/10.1038/nrc3556
Sato Y et al (2013) Integrated molecular analysis of clear-cell renal cell carcinoma. Nat Genet 45:860–867. https://doi.org/10.1038/ng.2699
Seong HA, Manoharan R, Ha H (2011) B-MYB positively regulates serine-threonine kinase receptor-associated protein (STRAP) activity through direct interaction. J Biol Chem 286:7439–7456. https://doi.org/10.1074/jbc.M110.184382
Sobin LH, Gospodarowicz MK, Wittekind C (2011) TNM Classification of Malignant Tumours, 7th Edition. UICC, vol 7th. Edition. Wiley-Blackwell
Sun SS, Fu Y, Lin JY (2020) Upregulation of MYBL2 independently predicts a poorer prognosis in patients with clear cell renal cell carcinoma. Oncol Lett 19:2765–2772. https://doi.org/10.3892/ol.2020.11408
Syed JS, Brito J, Pooli A, Boutros PC, Shuch B (2020) Transcriptomics in RCC. Urol Oncol. https://doi.org/10.1016/j.urolonc.2019.12.003
Toledo F, Wahl GM (2006) Regulating the p53 pathway: in vitro hypotheses, in vivo veritas. Nat Rev Cancer 6:909–923. https://doi.org/10.1038/nrc2012
Vieler M, Sanyal S (2018) p53 isoforms and their implications in cancer. Cancers (Basel). https://doi.org/10.3390/cancers10090288
Vogelstein B, Lane D, Levine AJ (2000) Surfing the p53 network. Nature 408:307–310. https://doi.org/10.1038/35042675
Vousden KH, Lane DP (2007) p53 in health and disease. Nat Rev Mol Cell Biol 8:275–283. https://doi.org/10.1038/nrm2147
Wade M, Li YC, Wahl GM (2013) MDM2, MDMX and p53 in oncogenesis and cancer therapy. Nat Rev Cancer 13:83–96. https://doi.org/10.1038/nrc3430
Warburton HE, Brady M, Vlatković N, Linehan WM, Parsons K, Boyd MT (2005) p53 regulation and function in renal cell carcinoma. Cancer Res 65:6498–6503. https://doi.org/10.1158/0008-5472.Can-05-0017
Wu X, Bayle JH, Olson D, Levine AJ (1993) The p53-mdm-2 autoregulatory feedback loop. Genes Dev 7:1126–1132. https://doi.org/10.1101/gad.7.7a.1126
Yang F et al (2018) LncRNA LOC653786 promotes growth of RCC cells via upregulating FOXM1. Oncotarget 9:12101–12111. https://doi.org/10.18632/oncotarget.24027
Zhang X, Lv QL, Huang YT, Zhang LH, Zhou HH (2017) Akt/FoxM1 signaling pathway-mediated upregulation of MYBL2 promotes progression of human glioma. J Exp Clin Cancer Res 36:105. https://doi.org/10.1186/s13046-017-0573-6
Acknowledgements
We would like to thank the entire team of the Urological Research Laboratory at the University Hospital Mannheim, who provided insights and expertise that greatly assisted in the realization of the experiments.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
MN: guarantor of integrity of the entire study, statistical analysis, manuscript preparation; KM: experimental studies/data analysis; KN and AS experimental guidance; ZVP and SP pathological validation; PE, JM, and FW: manuscript editing; MCK: study concepts and design, guarantor of integrity of the entire study, manuscript editing.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethical approval
The authors declare that no competing financial interests exist. This study was performed in adherence to the Declaration of Helsinki, and all patients gave approval to participate in this study.
Consent to participate
All participants have given their consent to participate in the study freely and after being informed.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nientiedt, M., Müller, K., Nitschke, K. et al. B-MYB—p53-related relevant regulator for the progression of clear cell renal cell carcinoma. J Cancer Res Clin Oncol 147, 129–138 (2021). https://doi.org/10.1007/s00432-020-03392-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03392-7