Abstract
Purpose
Osteosarcoma is the most common bone tumor, mainly affecting adolescents and young adults, and metastatic disease has poor outcomes with a dismal overall survival. Currently, chemotherapy is the standard of care with limited results, finding that new therapies could improve these outcomes. Preclinical and clinical studies have suggested a possible important role of ErbB pathway aberrations in osteosarcoma etiology. The present study shows the effect of afatinib, an irreversible ErbB family blocker in osteosarcoma cell lines.
Methods
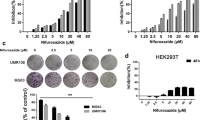
Within a panel of human osteosarcoma cell lines, we addressed cell viability assay using afatinib at increasing concentrations. Motility was measured in wound-healing assays and invasion capacity was assessed in Transwell chamber assays. Finally, to monitor ErbB pathway modulation by afatinib and related compounds, we used Western blot analyses.
Results
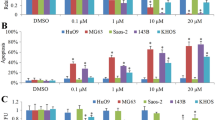
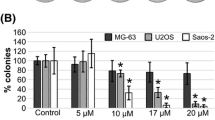
Cell viability inhibition, as well as a reduction of motility and migration of osteosarcoma cell line were observed after treatment with afatinib. Likewise, in the HOS cell line, afatinib decreased phosphorylation of key components in the ErbB signaling pathway.
Conclusions
Afatinib shows relevant antitumor effect in several osteosarcoma cell lines, as it causes a significant impact on cell viability, motility, and migration with a significant decrease in the activation of ErbB pathway activity.




Similar content being viewed by others
Change history
19 August 2020
The title of the article is incorrectly published in the original article. The correct article title is ���Afatinib is active in osteosarcoma cell lines���.
References
Allison DC, Carney SC, Ahlmann ER et al (2012) A meta-analysis of osteosarcoma outcomes in the modern medical era. Sarcoma. https://doi.org/10.1155/2012/704872
Bacci G, Briccoli A, Rocca M et al (2003) Neoadjuvant chemotherapy for osteosarcoma of the extremities with metastases at presentation: recent experience at the Rizzoli Institute in 57 patients treated with cisplatin, doxorubicin, and a high dose of methotrexate and ifosfamide. Ann Oncol 14(7):1126–1134. https://doi.org/10.1093/annonc/mdg286
Citri A, Yarden Y (2006) EGF-ERBB signalling: towards the systems level. Nat Rev Mol Cell Biol 7(7):505–516. https://doi.org/10.1038/nrm1962
Flick DA, Gifford GE (1984) Comparison of in vitro cell cytotoxic assays for tumor necrosis factor. J Immunol Methods 68(1–2):167–175. https://doi.org/10.1016/0022-1759(84)90147-9
Gvozdenovic A, Boro A, Born W et al (2017) A bispecific antibody targeting IGF-IR and EGFR has tumor and metastasis suppressive activity in an orthotopic xenograft osteosarcoma mouse model. Am J Cancer Res 7(7):1435–1449
Hughes DPM, Thomas DG, Giordano TJ et al (2004) Cell surface expression of epidermal growth factor receptor and Her-2 with nuclear expression of Her-4 in primary osteosarcoma. Cancer Res 64(6):2047–2053. https://doi.org/10.1158/0008-5472.can-03-3096
Hughes DPM, Thomas DG, Giordano TJ et al (2006) Essential erbB family phosphorylation in osteosarcoma as a target for CI-1033 inhibition. Pediatr Blood Cancer 46(5):614–623. https://doi.org/10.1002/pbc.20454
Jullien N, Dieudonné FX, Habel N et al (2013) ErbB3 silencing reduces osteosarcoma cell proliferation and tumor growth in vivo. Gene 521(1):55–61. https://doi.org/10.1016/j.gene.2013.03.031
Kager L, Zoubek A, Pötschger U et al (2003) Primary metastatic osteosarcoma: presentation and outcome of patients treated on neoadjuvant Cooperative Osteosarcoma Study Group protocols. J Clin Oncol 21(10):2011–2018. https://doi.org/10.1200/JCO.2003.08.132
Kansara M, Teng MW, Smyth MJ, Thomas DM (2014) Translational biology of osteosarcoma. Nat Rev Cancer 14(11):722–735. https://doi.org/10.1038/nrc3838
Kersting C, Gebert C, Agelopoulos K et al (2007) Epidermal growth factor receptor expression in high-grade osteosarcomas is associated with a good clinical outcome. Clin Cancer Res 13(10):2998–3005. https://doi.org/10.1158/1078-0432.CCR-06-2432
Kubo T, Piperdi S, Rosenblum J et al (2008) Platelet-derived growth factor receptor as a prognostic marker and a therapeutic target for imatinib mesylate therapy in osteosarcoma. Cancer 112(10):2119–2129. https://doi.org/10.1002/cncr.23437
Lee JA, Ko Y, Kim DH et al (2012) Epidermal growth factor receptor: Is it a feasible target for the treatment of Osteosarcoma? Cancer Res Treat 44(3):202–209. https://doi.org/10.4143/crt.2012.44.3.202
Liu J, Wu J, Zhou L et al (2015) ZD6474, a new treatment strategy for human osteosarcoma, and its potential synergistic effect with celecoxib. Oncotarget 6(25):21341–21352. https://doi.org/10.18632/oncotarget.4179
MacEwen EG, Pastor J, Kutzke J et al (2004) IGF-1 receptor contributes to the malignant phenotype in human and canine osteosarcoma. J Cell Biochem 92(1):77–91. https://doi.org/10.1002/jcb.20046
Machiels JPH, Haddad RI, Fayette J et al (2015) Afatinib versus methotrexate as second-line treatment in patients with recurrent or metastatic squamous-cell carcinoma of the head and neck progressing on or after platinum-based therapy (LUX-Head & Neck 1): an open-label, randomised phase 3 trial. Lancet Oncol 16(5):583–594. https://doi.org/10.1016/S1470-2045(15)70124-5
Mirabello L, Troisi RJ, Savage SA (2009) Osteosarcoma incidence and survival rates from 1973 to 2004: data from the surveillance, epidemiology, and end results program. Cancer 115(7):1531–1543. https://doi.org/10.1002/cncr.24121
Normanno N, De Luca A, Bianco C et al (2006) Epidermal growth factor receptor (EGFR) signaling in cancer. Gene 366(1):2–16. https://doi.org/10.1016/j.gene.2005.10.018
Ottaviani G, Jaffe N (2009) The epidemiology of osteosarcoma. Cancer Treat Res 152:3–13. https://doi.org/10.1007/978-1-4419-0284-9_1
Scotlandi K, Manara MC, Hattinger CM et al (2005) Prognostic and therapeutic relevance of HER2 expression in osteosarcoma and Ewing’s sarcoma. Eur J Cancer 41(9):1349–1361. https://doi.org/10.1016/j.ejca.2005.03.015
Sevelda F, Mayr L, Kubista B et al (2015) EGFR is not a major driver for osteosarcoma cell growth in vitro but contributes to starvation and chemotherapy resistance. J Exp Clin Cancer Res 34(1):134. https://doi.org/10.1186/s13046-015-0251-5
Solca F, Dahl G, Zoephel A et al (2012) Target binding properties and cellular activity of afatinib (BIBW 2992), an irreversible ErbB family blocker. J Pharmacol Exp Ther 343(2):342–350. https://doi.org/10.1124/jpet.112.197756
Soria JC, Felip E, Cobo M et al (2015) Afatinib versus erlotinib as second-line treatment of patients with advanced squamous cell carcinoma of the lung (LUX-Lung 8): an open-label randomised controlled phase 3 trial. Lancet Oncol 16(8):897–907. https://doi.org/10.1016/S1470-2045(15)00006-6
Wen YH, Koeppen H, Garcia R et al (2007) Epidermal growth factor receptor in osteosarcoma: expression and mutational analysis. Hum Pathol 38(8):1184–1191. https://doi.org/10.1016/j.humpath.2007.01.002
Wu PK, Chen WM, Chen CF et al (2009) Primary osteogenic sarcoma with pulmonary metastasis: clinical results and prognostic factors in 91 patients. Jpn J Clin Oncol 39(8):514–522. https://doi.org/10.1093/jjco/hyp057
Yang JCH, Wu YL, Schuler M et al (2015) Afatinib versus cisplatin-based chemotherapy for EGFR mutation-positive lung adenocarcinoma (LUX-Lung 3 and LUX-Lung 6): analysis of overall survival data from two randomised, phase 3 trials. Lancet Oncol 16(2):141–151. https://doi.org/10.1016/S1470-2045(14)71173-8
Young NR, Soneru C, Liu J et al (2015) Afatinib efficacy against squamous cell carcinoma of the head and neck cell lines in vitro and in vivo. Target Oncol 10(4):501–508. https://doi.org/10.1007/s11523-014-0353-6
Acknowledgements
Boehringer Ingelheim RCV GmbH & Co KG provided afatinb and financial support for some supplies of this work.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
432_2020_3220_MOESM1_ESM.tif
Supplementary file1 Supplementary Fig. 1 Cell Growth Inhibition. Percentage of inhibition of (a) HOS, (b) SAOS-2, (c) SJSA-1, (d) MNNG, (e) U2OS cell lines. This experiment was done four times. Data are presented as mean ± standard deviation (n=4). *P<0.05 compared to the control group (TIF 489 kb)
432_2020_3220_MOESM2_ESM.tif
Supplementary file2 Supplementary Fig. 2 Effect of afatinib in SAOS-2, SJSA-1 and U2OS cell migration. Representative images of non-metastatic osteosarcoma cell lines treated with afatinib, (a) SAOS-2 (b) SJSA-1 (c) U2OS. Each osteosarcoma cell line was treated with 0μM and IC50 dose. The monolayer wound was evaluated at basal time, at 12h and 24h, (n=3). The wounds were photographed using an inverted microscope (Evos, Life Technologies) at the 0h, 12h and 24h (TIF 1173 kb)
432_2020_3220_MOESM3_ESM.tif
Supplementary file3 Supplementary Fig. 3 Migration Assay. Distribution of the wound separation line caused by afatinib concentration and exposure time on the MNNG (a), HOS (b), SAOS-2 (c), SJSA-1 (d) and U2OS (e) cell lines. * stands for a significant p-value for Kruskal-Wallis test adjusted by multiple comparisons (p<0.05) (TIF 1088 kb)
432_2020_3220_MOESM4_ESM.docx
Supplementary file4 Supplementary Table 1. Width reduction median of the separating line since basal exposure time by Afatinib concentration and exposure time Supplementary Table 2. Estimated mean difference in the separating line width by Afatinib concentration stratified by exposure time (DOCX 14 kb)
Rights and permissions
About this article
Cite this article
Cruz-Ramos, M., Zamudio-Cuevas, Y., Medina-Luna, D. et al. Afatinib is active in osteosarcoma in osteosarcoma cell lines. J Cancer Res Clin Oncol 146, 1693–1700 (2020). https://doi.org/10.1007/s00432-020-03220-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-020-03220-y