Abstract
Purpose
To evaluate the ability of the new Gleason grade groups (GGGs) to stratify risk in prostate cancer patients with locally adverse pathologic features after radical prostatectomy (RP) thereby allowing more accurate assessment for planning eventual adjuvant therapy.
Patients and methods
Data on 172 patients with locally adverse pathologic features (including seminal vesicle invasion, extracapsular extension, or positive surgical margins) who had been treated with wait and see policy after RP were retrospectively analyzed for biochemical recurrence (BCR)-free survival. Kaplan–Meier survival analysis and Cox proportional hazard regression models were used to test the association between the GGGs and BCR. Finally, concordance indices of different grading classifications were calculated to evaluate the predictive accuracy for biochemical failure after RP.
Results
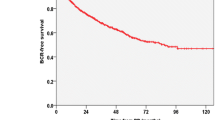
The five-year BCR-free survival rates were 71.2, 66.9, 25.7, 17.4, and 8.3 % for GGG 1–5 assessed on surgical specimens (p < 0.001, log-rank test). In the two-way log-rank test, men with prostatectomy GGG 2 had a lower progression risk relative to GGG 3 (p = 0.001), though similar risk as GGG 1 (p = 0.105). In multivariate Cox regression analysis, specimen GGG ≥3 and early postoperative PSA ≥0.1 ng/ml were independent risk factors for biochemical failure (p < 0.001). In addition, GGGs had higher predictive accuracy compared with the alternate classification system (improvement in concordance index by 0.036–0.141).
Conclusions
For the appropriate patient, depending on age, physical condition, early postoperative PSA, patient desire, etc., could be a candidate for wait and see policy with specimen GGG 2 disease, so to distinguish this from GGG 3 may facilitate discussions at the point of treatment decision making.

Similar content being viewed by others
References
Audenet F, Seringe E, Drouin SJ et al (2012) Persistently elevated prostate-specific antigen at six weeks after radical prostatectomy helps in early identification of patients who are likely to recur. World J Urol 30:239–244
Bolla M, van Poppel H, Tombal B et al (2012) Postoperative radiotherapy after radical prostatectomy for high-risk prostate cancer: long-term results of a randomised controlled trial (EORTC trial 22911). Lancet (London, England) 380:2018–2027
Chan TY, Partin AW, Walsh PC, Epstein JI (2000) Prognostic significance of Gleason score 3 + 4 versus Gleason score 4 + 3 tumor at radical prostatectomy. Urology 1(56):823–827
de Boo L, Pintilie M, Yip P, Baniel J, Fleshner N, Margel D (2015) Time from first detectable PSA following radical prostatectomy to biochemical recurrence: a competing risk analysis. Can Urol Assoc J 9:E14–E21
Dillioglugil O, Leibman BD, Kattan MW, Seale-Hawkins C, Wheeler TM, Scardino PT (1997) Hazard rates for progression after radical prostatectomy for clinically localized prostate cancer. Urology 50:93–99
Epstein JI, Carmichael M, Partin AW, Walsh PC (1993) Is tumor volume an independent predictor of progression following radical prostatectomy? A multivariate analysis of 185 clinical stage B adenocarcinomas of the prostate with 5 years of followup. J Urol 149:1478–1481
Epstein JI, Feng Z, Trock BJ, Pierorazio PM (2012) Upgrading and downgrading of prostate cancer from biopsy to radical prostatectomy: incidence and predictive factors using the modified Gleason grading system and factoring in tertiary grades. Eur Urol 61:1019–1024
Epstein JI, Zelefsky MJ, Sjoberg DD et al (2016a) A contemporary prostate cancer grading system: a validated alternative to the Gleason score. Eur Urol 69:428–435
Epstein JI, Egevad L, Amin MB, Delahunt B, Srigley JR, Humphrey PA (2016b) The 2014 International Society of Urological Pathology (ISUP) Consensus Conference on Gleason Grading of Prostatic Carcinoma: definition of grading patterns and proposal for a new grading system. Am J Surg Pathol 40:244–252
Fontenot PA, Mansour AM (2013) Reporting positive surgical margins after radical prostatectomy: time for standardization. BJU Int 111:E290–E299
Forgues A, Rozet F, Audenet F et al (2014) Oncologic outcomes after minimally invasive radical prostatectomy in patients with seminal vesicle invasion (pT3b) without adjuvant therapy. World J Urol 32:519–524
Grossfeld GD, Chang JJ, Broering JM et al (2000) Impact of positive surgical margins on prostate cancer recurrence and the use of secondary cancer treatment: data from the CaPSURE database. J Urol 163:1171–1177 (quiz 295)
Khan MA, Partin AW, Mangold LA, Epstein JI, Walsh PC (2003) Probability of biochemical recurrence by analysis of pathologic stage, Gleason score, and margin status for localized prostate cancer. Urology 62:866–871
Loeb S, Folkvaljon Y, Robinson D, Lissbrant IF, Egevad L, Stattin P (2015) Evaluation of the 2015 Gleason grade groups in a nationwide population-based cohort. Eur Urol 69:1135–1141
Pierorazio PM, Walsh PC, Partin AW, Epstein JI (2013) Prognostic Gleason grade grouping: data based on the modified Gleason scoring system. BJU Int 111:753–760
Sartor O, Loriaux DL (2006) The emotional burden of low-risk prostate cancer: proposal for a change in nomenclature. Clin Genitourin Cancer 5:16–17
Secin FP, Bianco FJ Jr, Vickers AJ et al (2006) Cancer-specific survival and predictors of prostate-specific antigen recurrence and survival in patients with seminal vesicle invasion after radical prostatectomy. Cancer 1(106):2369–2375
Shapiro RH, Johnstone PA (2012) Risk of Gleason grade inaccuracies in prostate cancer patients eligible for active surveillance. Urology 80:661–666
Siddiqui SA, Boorjian SA, Blute ML et al (2011) Impact of adjuvant androgen deprivation therapy after radical prostatectomy on the survival of patients with pathological T3b prostate cancer. BJU Int 107:383–388
Stark JR, Perner S, Stampfer MJ et al (2009) Gleason score and lethal prostate cancer: does 3 + 4 = 4 + 3? J Clin Oncol 20(27):3459–3464
Stephenson AJ, Kattan MW, Eastham JA et al (2009) Prostate cancer-specific mortality after radical prostatectomy for patients treated in the prostate-specific antigen era. J Clin Oncol 10(27):4300–4305
Swindle P, Eastham JA, Ohori M et al (2005) Do margins matter? The prognostic significance of positive surgical margins in radical prostatectomy specimens. J Urol 174:903–907
Thompson IM Jr, Tangen CM, Paradelo J et al (2006) Adjuvant radiotherapy for pathologically advanced prostate cancer: a randomized clinical trial. JAMA 15(296):2329–2335
Thompson IM, Tangen CM, Paradelo J et al (2009) Adjuvant radiotherapy for pathological T3N0M0 prostate cancer significantly reduces risk of metastases and improves survival: long-term followup of a randomized clinical trial. J Urol 181:956–962
Vellekoop A, Loeb S, Folkvaljon Y, Stattin P (2014) Population based study of predictors of adverse pathology among candidates for active surveillance with Gleason 6 prostate cancer. J Urol 191:350–357
Vesely S, Jarolim L, Duskova K, Schmidt M, Dusek P, Babjuk M (2014) The use of early postoperative prostate-specific antigen to stratify risk in patients with positive surgical margins after radical prostatectomy. BMC Urol 14:79
Wiegel T, Bottke D, Steiner U et al (2009) Phase III postoperative adjuvant radiotherapy after radical prostatectomy compared with radical prostatectomy alone in pT3 prostate cancer with postoperative undetectable prostate-specific antigen: ARO 96-02/AUO AP 09/95. J Clin Oncol 20(27):2924–2930
Wiegel T, Bartkowiak D, Bottke D et al (2015) Prostate-specific antigen persistence after radical prostatectomy as a predictive factor of clinical relapse-free survival and overall survival: 10-year data of the ARO 96-02 trial. Int J Radiat Oncol Biol Phys 1(91):288–294
Acknowledgments
This study was supported by National Natural Science Foundation of China (91129725, 81572536), Science and Technology Commission of Shanghai Municipality (14140901700), Shanghai Municipal Education Commission-Gaofeng Clinical Medicine Grant Support (20152215), and the Key Disciplines Group Construction Project of Pudong Health Bureau of Shanghai (PWZxq2014-05). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflict of interest to declare.
Human rights
This study was approved by the Ethics Committee at the Renji Hospital, Shanghai Jiao Tong University School of Medicine. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Xun Shangguan, Baijun Dong and Yanqing Wang have contributed equally to this work.
Jiahua Pan and Wei Xue co-supervised this work.
Rights and permissions
About this article
Cite this article
Shangguan, X., Dong, B., Wang, Y. et al. Management of prostate cancer patients with locally adverse pathologic features after radical prostatectomy: feasibility of active surveillance for cases with Gleason grade 3 + 4 = 7. J Cancer Res Clin Oncol 143, 123–129 (2017). https://doi.org/10.1007/s00432-016-2262-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00432-016-2262-9