Abstract
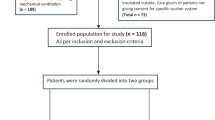
This study aimed to evaluate the effect of closed versus open endotracheal tube suction in reducing ventilator-associated pneumonia in mechanically ventilated neonates. In this open-label, parallel-group, randomized controlled trial with allocation concealment, ventilated neonates (≥ 28 weeks and ≥ 800 g) were either allocated to the closed-suction group (n = 41) or open-suction group (n = 39). The ventilator circuit of the babies enrolled in the closed-suction group was attached to the closed-suction catheter on the requirement of their first suction, and it was changed after every 48 h or earlier if visibly soiled whereas babies enrolled in the open-suction group were suctioned with a new suction catheter each time they require suction. The primary outcome was the incidence of VAP per 1000 days. Baseline maternal and neonatal characteristics were comparable between the two groups. The proportion of neonates with VAP in the closed-suction group was 3 (7.3%) and 1 (2.6%) in the open-suction group with an RR of 2.8 (95% CI: 0.30–26.28) and a p-value of 0.35. The incidence of VAP in the closed-suction group was 3.9 per 1000 ventilator days and 1.3 per 1000 ventilator days in the open-suction group. The incidence of clinical VAP/1000 ventilator days was 33.63 ± 22.96 in the closed-suction group and 28.67 ± 12.32 in the open-suction group with a mean difference of 5 (95% CI: − 3.26 to 13.26) and p-value of 0.24.
Conclusion: In a unit with a low incidence of VAP, the effect of the endotracheal suction method alone did not impact the occurrence of VAP in the study population.
Clinical trial registration: CTRI/2020/03/023679; Date: 17.02.2020.
What is Known: • Better physiological stability of the closed-suctioning method on short-term measures including noticeably fewer episodes of hypoxia, a smaller drop in TcPO2 levels, and less variability in heart rate and bradycardia episodes. | |
What is New: • In a unit with a low incidence of VAP in the neonates, randomized control trial studying the effect of the endotracheal suction method alone did not impact the occurrence of VAP amongst the study population. |


Similar content being viewed by others
Data availability
The data that support the findings of this study are not available publicly due to ethical reasons but are available with the authors and could be produced upon reasonable request.
Abbreviations
- BPD:
-
Bronchopulmonary dysplasia
- CDC:
-
Center for disease control and prevention
- IVH:
-
Intraventricular hemorrhage
- PVL:
-
Periventricular leukomalacia
- NICHD:
-
National institute of child health and human development
- NICU:
-
Neonatal intensive care unit
- NNIS:
-
National nosocomial infection surveillance
- RBSK:
-
Rashtriya Bal Swasthya Karyakram
- SNAPPE:
-
Score for neonatal acute physiology with perinatal extension
- VAP:
-
Ventilator-associated pneumonia
References
Beitler JR, Malhotra A, Thompson BT (2016) Ventilator-induced lung injury. Clin Chest Med 37:633–646
Garland JS (2010) Strategies to prevent ventilator-associated pneumonia in neonates. Clin Perinatol 37:629–643
Pneumonia (ventilator-associated [VAP] and non-ventilator-associated pneumonia [PNEU]) event. Available online at https://www.cdc.gov/nhsn/pdfs/pscmanual/6pscvapcurrent.pdf. Last accessed 18 Jan 2022
Siempos II, Vardakas KZ, Falagas ME (2008) closed tracheal suction systems for prevention of ventilator-associated pneumonia. Br J Anaesth 100:299–306
Kuriyama A, Umakoshi N, Fujinaga J, Takada T (2015) Impact of closed versus open tracheal suctioning systems for mechanically ventilated adults: a systematic review and meta-analysis. Intensive Care Med 41:402–411
Tume LN, Copnell B (2015) Endotracheal suctioning of the critically ill child. J Pediatr Intensive Care 4:56–63
Tekin R, Dal T, Pirinccioglu H, Oygucu SE (2013) A 4-year surveillance of device-associated nosocomial infections in a neonatal intensive care unit. Pediatr Neonatol 54:303–308
National Nosocomial Infections Surveillance System (2004) National Nosocomial Infections Surveillance (NNIS) System report, data summary from January 1992 through June 2004, issued October 2004. Am J Infect Control 32:470–485
Cernada M, Brugada M, Golombek S, Vento M (2014) ventilator-associated pneumonia in neonatal patients: an update. Neonatology 105:98–107
Geslain G, Guellec I, Guedj R, Guilbert J, Jean S, Valentin C et al (2018) Incidence and risk factors of ventilator-associated pneumonia in neonatal intensive care unit: a first French study. Minerva Anestesiol 84:829–835
Taylor JE, Hawley G, Flenady V, Woodgate PG (2011) Tracheal suctioning without disconnection in intubated ventilated neonates. Cochrane Database Syst Rev 7:CD003065.
Herting E, Gefeller O, Land M, van Sonderen L, Harms K, Robertson B et al (2000) Surfactant treatment of neonates with respiratory failure and group B streptococcal infection. Pediatrics 106:957–964
van der Zwet WC, Kaiser AM, van Elburg RM, Berkhof J, Fetter WP, Parlevliet GA et al (2005) Nosocomial infections in a Dutch neonatal intensive care unit: surveillance study with definitions for infection specifically adapted for neonates. J Hosp Infect 61:300–311
Volpe JJ (2008) Intracranial hemorrhage: Neurology of the neonate. Saunders Elsevier, Philadelphia, PA, pp 481–588
de Vries LS, Eken P, Dubowitz LM (1992) The spectrum of leukomalacia using cranial ultrasound. Behav Brain Res 49:1–6
International Classification of Diseases (ICD)-WHO. https://icd.who.int/browse10/Content/statichtml/ICD10Volume2_en_2010.pdf. Last accessed 20 Jan 2022
Jobe AH, Bancalari E (2001) Bronchopulmonary dysplasia. Am J Respir Crit Care Med 163:1723–1729
Universal eye screening-National Health Mission. Available at https://www.nhm.gov.in/images/pdf/programmes/RBSK/Resource_Documents/Revised_ROP_Guidelines-Web_Optimized.pdf. Last accessed 19 Jan 2022
Ritz R, Scott LR, Coyle MB, Pierson DJ (1986) Contamination of a multiple-use suction catheter in a closed-circuit system compared to contamination of a disposable, single-use suction catheter. Respir Care 31:1086–1091
Morrow BM, Mowzer R, Pitcher R, Argent AC (2012) Investigation into the effect of closed-system suctioning on the frequency of pediatric ventilator-associated pneumonia in a developing country. Pediatr Crit Care Med 13:25–32
Tullu MS, Deshmukh CT, Baveja SM (2000) Bacterial nosocomial pneumonia in Paediatric Intensive Care Unit. J Postgrad Med 46:18–22
Srinivasan R, Asselin J, Gildengorin G, Wiener-Kronish J, Flori HR (2009) A prospective study of ventilator-associated pneumonia in children. Pediatrics 123:1108–1115
Principi N, Esposito S (2007) Ventilator-associated pneumonia (VAP) in pediatric intensive care units. Pediatr Infect Dis J 26:841–843
Dell’Orto V, Raschetti R, Centorrino R, Montane A, Tissieres P, Yousef N et al (2019) Short- and long-term respiratory outcomes in neonates with ventilator-associated pneumonia. Pediatr Pulmonol 54:1982–1988
Cordero L, Sananes M, Ayers LW (2000) Comparison of a closed (Trach Care MAC) with an open endotracheal suction system in small premature infants. J Perinatol 20:151–156
Elward AM, Warren DK, Fraser VJ (2002) Ventilator-associated pneumonia in pediatric intensive care unit patients: risk factors and outcomes. Pediatrics 109:758–764
Almuneef M, Memish ZA, Balkhy HH, Alalem H, Abutaleb A (2004) Ventilator-associated pneumonia in a pediatric intensive care unit in Saudi Arabia: a 30-month prospective surveillance. Infect Control Hosp Epidemiol 25:753–758
Author information
Authors and Affiliations
Contributions
All authors contributed to the study’s conception and design. Material preparation, data collection, and analysis were performed by Dr. Ajaya Kumar Gahan, Dr. Suksham Jain, Dr. Supreet Khurana, and Dr. Deepak Chawla. The first draft of the manuscript was written by Dr. Ajaya Kumar Gahan, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval
The study was approved by the ethics committee of GMCH (no. GMCH/IEC/2019/236, dt. 30/12/2019).
Consent to participate
Written informed consent was obtained from the parents of all participants.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Daniele De Luca
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Article summary: Closed-suction method as compared to the open-suction method should decrease VAP in mechanically ventilated neonates. The current study was done to assess the same.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gahan, A.K., Jain, S., Khurana, S. et al. Closed versus open endotracheal tube suction in mechanically ventilated neonates: a randomized controlled trial. Eur J Pediatr 182, 785–793 (2023). https://doi.org/10.1007/s00431-022-04726-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-022-04726-y