Abstract
Newborns are at relatively high risk for developing hypoglycaemia in the first 24 h after birth. Well-known risk factors are prematurity, small for gestational age (SGA) or large for gestational age (LGA), and maternal pre-existent or gestational diabetes mellitus. Prolonged hypoglycaemia is associated with poor neurodevelopmental outcomes; hence, prevention through proper monitoring and treatment is important. Given the ongoing debate concerning frequency and duration of screening for neonatal hypoglycaemia, therefore, we investigated the frequency and duration of glucose monitoring safe to discover neonatal hypoglycaemia in different risk groups. Data of newborns at risk for hypoglycaemia were retrospectively collected and analysed. Blood glucose concentrations were measured 1, 3, 6, 12, and 24 h after birth. Moderate hypoglycaemia was defined as a blood glucose concentration of < 2.2 mM and severe hypoglycaemia as a concentration of < 1.5 mM. Of 1570 newborns, 762 (48.5%) had at least one episode of hypoglycaemia in the first 24 h after birth; 30.6% of them had severe hypoglycaemia (all in the first 9 h after birth). Only three SGA and two LGA newborns had a first moderate asymptomatic hypoglycaemic episode beyond 12 h after birth. The incidence of hypoglycaemia increased with accumulation of multiple risk factors.
Conclusion: Safety of limiting the monitoring to 12 h still has to be carefully evaluated in the presence of SGA or LGA newborns; however, our results suggest that 12 h is enough for late preterm newborns (> 34 weeks) and maternal diabetes.
What is Known: • Newborns are at relatively high risk for developing hypoglycaemia and such hypoglycaemia is associated with adverse neurodevelopmental outcomes. • Proper glucose monitoring and prompt treatment in case of neonatal hypoglycaemia are necessary. | |
What is New: • Glucose monitoring 12 h after birth is proficient for most newborns at risk. • Maternal diabetes leads to the highest risk of early neonatal hypoglycaemia and newborns with more than one risk factor are at increased risk of hypoglycaemia. |
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
To date, there is still little evidence and consensus regarding definition, indication, and frequency of screening for neonatal hypoglycaemia, one of the most frequently encountered problems in neonatology. Newborns are at relatively high risk for developing hypoglycaemia in the first 24 h after birth [10]. The incidence of hypoglycaemia in apparently healthy newborns varies widely (between 4 and 40%) depending amongst others on definition of hypoglycaemia, timing of glucose monitoring after delivery, and population studied [16, 21].
Risk factors for neonatal hypoglycaemia are prematurity (< 37 weeks of gestation), being small for gestational age (SGA) (birth weight ≤ 10th percentile for GA), large for gestational age (LGA) (birth weight ≥ 90th percentile for GA), and pre-existent maternal diabetes (diabetes mellitus type 1, type 2 (T1DM, T2DM)) or gestational diabetes mellitus (GDM) [2, 3, 11, 26].
In apparently healthy infants, glucose concentrations tend to decrease after birth, reach a nadir 1 h after birth, increase again, and stabilise after one day [2, 16]. The majority of first hypoglycaemic episodes occur in the first 24 h after birth (81%), from which 48% within 6 h [12].
Recurrent severe hypoglycaemia is associated with poor neurodevelopmental outcomes; however, duration and severity of hypoglycaemia to cause injury are unknown. The management of the asymptomatic infant at risk for low glucose remains controversial. Despite this, prevention of (prolonged) hypoglycaemia by screening and treatment seems rational [3, 13, 19].
Existing guidelines for screening and treatment recommend glucose monitoring in newborns at risk but vary in (1) definition of hypoglycaemia (between 2.2 and 2.6 mM), (2) total monitoring time (12–48 h after birth), and (3) time intervals (between 1 and 8 h). This is illustrating the lack of consensus regarding the frequency and duration of glucose monitoring deemed necessary and safe to discover neonatal hypoglycaemia [2, 7, 12, 15, 18]. Furthermore, different risk factors may prompt a different strategy. Therefore, we studied the onset of the first hypoglycaemic episode in newborns at risk according to their risk factors.
Materials and methods
This is a retrospective observational cohort study. We enrolled all eligible newborns born and admitted at Radboudumc Amalia Children’s Hospital Nijmegen, the Netherlands, from a four-year period (2010–2013). Inclusion criteria were gestational age (GA) ≥ 34 weeks and screening indication according to our local guideline: prematurity (< 37 weeks of gestation), SGA, LGA, and maternal diabetes of any kind (identical to recommendations by the American Academy of Pediatrics (AAP)) [3, 4]. Exclusion criteria were severe asphyxia (Apgar 5 min after birth ≤ 3) [1] and death within the first 24 h after birth. In the first 24 h after birth, moderate hypoglycaemia and severe hypoglycaemia were defined as a blood glucose concentration (GC) between 1.5–2.1 mM and below 1.5 mM, respectively, moderate and severe reflecting therapy in this case. Blood glucose concentrations were measured on capillary blood samples taken by several point-of-care (POC) glucose meters during this period (HemoCue Glu201DM, Nova StatStrip GluCard memory PC, Roche OMNI-56 blood gas analyser and Siemens Rapid lab 1265). In case of hypoglycaemia (GC < 2.2 mM), capillary blood was sent to the laboratory for confirmation.
Blood glucose samples were taken 1, 3, 6, 12, and 24 h after birth according to local guidelines [9]. In case of any clinical signs of hypoglycaemia, such as twitching and drowsiness, additional glucose samples were taken.
Feeding was initiated in the first hour after birth. Breastfeeding was encouraged throughout pregnancy, but the ultimate choice between breastfeeding and formula feeding was made by the parents. In our hospital (as in the Netherlands), approximately 80% of women start with exclusive breastfeeding post-delivery [22]. Feeding is continued at least every 3 h, with breastfeeding sometimes more often. Newborns with moderate hypoglycaemia were treated with increased enteral carbohydrate intake, either through more frequent breast feedings or (additional) formula feeding. In case of repeated moderate hypoglycaemia (≥ 3×), continuous intravenous glucose 10% was started after administration of 2 ml/kg bolus. Newborns with severe hypoglycaemia were immediately treated with continuous intravenous glucose 10% and a bolus.
Birth percentiles, SGA (< 10th percentile) or LGA (> 90th percentile), were calculated using Dutch birth weight curves (https://www.perined.nl/producten/geboortegewichtcurven).
Relevant maternal and neonatal information and GC were extracted from patient files. Our primary outcome was time of first hypoglycaemia, which we studied in all risk factors separately. When hypoglycaemia was described in discharge correspondence but data on timing of blood glucose concentrations were missing of more than two standardised sample moments, cases were only used for overall analysis.
For statistical analysis, SPSS software version 20 was used. Normally, distributed data were expressed as percentage and mean (SD). Median (range) was used for skewed distributed data. For 2 × 2 tables, chi-square test was performed. Multivariate logistic regression analyses were used to examine the association between (severe) hypoglycaemia and possible risk factors, including screening indications, background information, and other complications. A p value < 0.05 was considered statistically significant.
This study was exempt from Regional Ethics Review Board approval, under the legal requirements for clinical research in the Netherlands.
Results
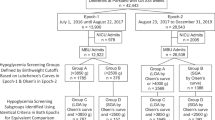
We enrolled 1628 newborns with risk factors for hypoglycaemia of which 1573 met our inclusion and exclusion criteria. Data of three newborns were unavailable. The final analysis was performed on data from 1570 newborns. In 327 cases, data of more than two sample moments were missing. These cases were only used for the overall analysis (hypoglycaemia 125/327). Newborns at risk were premature (33.5%), SGA (32.4%), LGA (27.6%), or born to a diabetic mother (19.1%) (Table 1). Most newborns had one risk factor (87.1%), 12.1% two risk factors, and 0.7% three risk factors (Fig. 1). In the first hour after birth, GCs for this cohort reach their lowest values. After that, blood GCs remain stable at a value of 3.1–3.4 mM (Fig. 2).
Of 1570 patients at risk, 762 (48.5%) suffered from at least one episode of hypoglycaemia. One-third of hypoglycaemic episodes were severe (< 1.5 mM), with maternal pre-existing diabetes as the main risk factor (54.5%) (Table 1). The prevalence of hypoglycaemia is similar in different birth weight groups (p > 0.05) (Table 1). Pre-existent maternal diabetes led to early hypoglycaemia (first hour after birth) in 21 cases (75%) (Table 2).
In GDM, the majority of newborns (100/113; 88.5%) had a hypoglycaemia within 3 h after birth. Hypoglycaemia occurring more than 12 h after birth for the first time was seen in only five newborns (Table 2). One SGA newborn had a GC of 2.1 mM 15 h after birth, and three LGA newborns and one SGA newborn had moderate hypoglycaemias (GC 1.7, 1.9, 2.1, and 2.1 mM respectively) 24 h after birth. These newborns were all treated with additional feedings only.
In the group with other complications, e.g. congenital abnormalities, respiratory distress, or (risk for) infection, significantly less newborns developed a hypoglycaemic episode (p = 0.04).
The overall prevalence of hypoglycaemia increased with increasing number of risk factors (one risk factor (43–55%), comparing to the group with two risk factors (51–84%, p = 0.011) and the group with three risk factors (50–89%, p = 0.004)) (Fig. 1).
Multivariate logistic regression analysis including all potential risk factors, sex, single/multiple birth, and other complications (congenital abnormalities, respiratory distress and (risk for) infection) showed that hypoglycaemia was significant associated with prematurity (OR = 1.545, 95% CI 1.00–2.02, p < 0.05), pre-existent DM (T1DM and T2DM combined) (OR = 4.23, 95% CI 1.81–10.32, p = 0.001), and other complications (OR = 0.70, 95% CI 0.53–0.94, p = 0.018). Also, in case of severe hypoglycaemia, significant association was found with newborns born to mothers with pre-existent DM (OR = 4.74, 95% CI 2.44–9.20, p < 0.001) and prematurity (OR = 1.84, 95% CI 1.24–2.73, p = 0.002).
Discussion
The overall incidence of hypoglycaemia in our cohort of neonates at risk was 48.5%. For most newborns at risk, it is safe to stop monitoring GCs 12 h after birth. The highest incidence of hypoglycaemia was related to maternal diabetes and there is an increasing risk of hypoglycaemia with increasing number of risk factors. The majority of first hypoglycaemias occurred within 3 h after birth, comparable to other studies [14, 16].
The overall incidence of hypoglycaemia in our cohort is rather high; other studies report 6.8–51% [6, 12, 20, 23]. This might be due to our selection criteria for newborns at risk for hypoglycaemia although our screening indications are identical to those recommended by the AAP [3]. Other possible explanations are our standardised and frequent monitoring and difference in GC cut-off point to define hypoglycaemia. Hypoglycaemia was defined as a GC < 2.6 mM by Harris et al. [12] (overall incidence 51%) and a GC < 1.6 mM by Schaefer-Graf et al. [23] (overall incidence 25.2%); by means of this, incidence of hypoglycaemia might be influenced. Our lowest level of action is 2.2 mM in the first 24 h which is comparable to the actionable range recommended by the AAP (4–24 h after birth, 1.9–2.5 mM) [3].
A wide range of policies regarding duration of glucose concentration monitoring exists [2, 7, 12, 15]. Some authors state that the same screening strategy should be applied to each risk group while others suggest to differentiate between risk groups [12]. Different studies suggest that LGA newborns and newborns of diabetic mothers should have their monitoring discontinued 12 h after birth (if GC is ≥ 2.6 mM) [2, 7] and SGA newborns and late preterm newborns after 24 h [2], 36 h [7], or 48 h [15].
The high incidence of hypoglycaemia in offspring of mothers with pre-existent DM (81%) was comparable to the incidence seen in the study of Maayan Metzger et al. (54% GC < 2.6 mM and 83% GC < 2.2 mM) [18] but rather high compared to the study of Harris et al. (49%) [12]. However, in the latter study, no distinction between maternal pre-existent DM and GDM was defined.
More than one risk factor resulted in higher incidence (Fig. 2) which is in contrast to the study of Harris et al. in which no significant difference was found [12]. This might be due to the relative small sample size in their study.
The nadir 1–2 h after birth in blood GC is also observed in apparently health newborns and is considered part of normal adaptation to postnatal life by several authors, although it is not consistently described [2, 14, 16].
Our data illustrate that 96.7% (710/734) of all newborns who developed hypoglycaemia had the first hypoglycaemia within 6 h after birth. Since there were only five cases of mild asymptomatic hypoglycaemia beyond 12 h, all treated with additional feeding only, one could argue to stop glucose monitoring after 12 h for all risk groups. Safety of limiting the monitoring to 12 h still has to be carefully evaluated in the presence of SGA or LGA newborns.
Some limitations of this study should be addressed. Due to the retrospective design, it was not possible to obtain the exact glucose concentrations in 327 cases. Since the introduction of the electronic patient files, values of blood glucose concentrations are directly stored in these files. Furthermore, not all samples were obtained at the exact prescribed time points, which was mainly due to breastfeeding on demand and sampling just prior to intake (recommended clinical practice). Also, in this time period, several blood glucose meters and analysers were used. It is known that results of POC glucose meters in the critical range may be unreliable [24]. To minimise misdiagnosis, blood samples, which showed a GC below 2.2 mM, were retested in the clinical laboratory. Moreover, variance in reliability of POC glucose meters is depending on variability in instrument analytical performance [17] and interaction between users and POC glucose meters [8]. Continuous glucose monitoring (CGM) has been studied in neonates and results are promising. CGM could potentially decrease number of blood samples and the exposure to hypoglycaemia. Furthermore, it is feasible, calibration can be as low as 12 hourly, and it has limited side effects even in premature newborns (birth weight < 1500 g) and is well tolerated [5]. However, no association with improved clinical outcomes has been confirmed yet. There are technical issues to be improved as well, for example the provided range of 2.2 to 22.0 mM, which is insufficient to detect neonatal hypoglycaemia [25]. Randomised trials should demonstrate long-term outcome data using CGM [5, 25]. In our centre, ~ 85% is Caucasian; therefore, our results may need to be confirmed in other ethnic populations.
To our knowledge, this is the largest published study that specifically investigated the length of time that is necessary in neonatal glucose monitoring. The newborns were selected based on strict inclusion criteria for each separate risk factor and GC were measured using a standardised protocol.
In conclusion, the onset of first hypoglycaemia did not occur beyond 12 h of glucose monitoring after birth in prematurity and maternal diabetes in the observed time frame. Multiple risk factors and an inappropriate birth weight for gestational age may increase the risk of hypoglycaemic episode.
Abbreviations
- CGM:
-
Continuous glucose monitoring
- DM:
-
Diabetes mellitus
- GC:
-
Glucose concentration
- GDM:
-
Gestational diabetes mellitus
- LGA:
-
Large for gestational age
- mM:
-
Millimolar (1 mmol/litre)
- POC:
-
Point-of-care
- SD:
-
Standard deviation
- SGA:
-
Large for gestational age
- T1DM:
-
Diabetes mellitus type 1
- T2DM:
-
Diabetes mellitus type 2
References
ACOG Committee Opinion (2006) The Apgar score. Obstet Gynecol 107:1209–1212
Adamkin DH (2011) Postnatal glucose homeostasis in late-preterm and term infants. Pediatrics 127:575–579. https://doi.org/10.1542/peds.2010-3851
Adamkin DH (2017) Neonatal hypoglycemia. Semin Fetal Neonatal Med 22:36–41. https://doi.org/10.1016/j.siny.2016.08.007
Adamkin DH, Committe on Fetus and Newborn (2011) Postnatal glucose homeostasis in late-preterm and term infants. Pediatrics 127:575–579. https://doi.org/10.1542/peds.2010-3851
Beardsall K, Vanhaesebrouck S, Ogilvy-Stuart AL, Vanhole C, vanWeissenbruch M, Midgley P, Thio M, Cornette L, Ossuetta I, Palmer CR, Iglesias I, de Jong M, Gill B, de Zegher F, Dunger DB (2013) Validation of the continuous glucose monitoring sensor in preterm infants. Arch Dis Child Fetal Neonatal Ed 98:136–141. https://doi.org/10.1136/archdischild-2012-301661
Bhat MA, Kumar P, Bhansali A, Majumdar S, Narang A (2000) Hypoglycemia in small for gestational age babies. Indian J Pediatr 67:423–427
Canadian Pediatric Society Fetal and Newborn Committee (2004) Screening guidelines for newborns at risk for low blood glucose. Paediatr Child Health 9:723–740
Cohen M, Boyle E, Delaney C, Shaw J (2006) A comparison of blood glucose meters in Australia. Diabetes Res Clin Pract 71:113–118. https://doi.org/10.1016/j.diabres.2005.05.013
Department of neonatology R (2013) Guideline hypoglycaemia and hyperglycaemia in neonates
Deshpande S, Ward Platt M (2005) The investigation and management of neonatal hypoglycaemia. Semin Fetal Neonatal Med 10:351–361. https://doi.org/10.1016/j.siny.2005.04.002
Flores-le Roux JA, Sagarra E, Benaiges D, Hernandez-Rivas E, Chillaron JJ, Puig de Dou J, Mur A, Lopez-Vilchez MA, Pedro-Botet J (2012) A prospective evaluation of neonatal hypoglycaemia in infants of women with gestational diabetes mellitus. Diabetes Res Clin Pract 97:217–222. https://doi.org/10.1016/j.diabres.2012.03.011
Harris DL, Weston PJ, Harding JE (2012) Incidence of neonatal hypoglycemia in babies identified as at risk. J Pediatr 161:787–791. https://doi.org/10.1016/j.jpeds.2012.05.022
Harris DL, Weston PJ, Signal M, Chase JG, Harding JE (2013) Dextrose gel for neonatal hypoglycaemia (the Sugar Babies Study): a randomised, double-blind, placebo-controlled trial. Lancet 382:2077–2083. https://doi.org/10.1016/S0140-6736(13)61645-1
Heck LJ, Erenberg A (1987) Serum glucose levels in term neonates during the first 48 hours of life. J Pediatr 110:119–122
Holtrop PC (1993) The frequency of hypoglycemia in full-term large and small for gestational age newborns. Am J Perinatol 10:150–154. https://doi.org/10.1055/s-2007-994649
Hoseth E, Joergensen A, Ebbesen F, Moeller M (2000) Blood glucose levels in a population of healthy, breast fed, term infants of appropriate size for gestational age. Arch Dis Child Fetal Neonatal Ed 83:F117–F119
Khan AI, Vasquez Y, Gray J, Wians FHJ, Kroll MH (2006) The variability of results between point-of-care testing glucose meters and the central laboratory analyzer. Arch Pathol Lab Med 130:1527–1532. https://doi.org/10.1043/1543-2165(2006)130[1527:TVORBP]2.0.CO;2
Maayan-Metzger A, Lubin D, Kuint J (2009) Hypoglycemia rates in the first days of life among term infants born to diabetic mothers. Neonatology 96:80–85. https://doi.org/10.1159/000203337
McKinlay CJD, Alsweiler JM, Ansell JM, Anstice NS, Chase JG, Gamble GD, Harris DL, Jacobs RJ, Jiang Y, Paudel N, Signal M, Thompson B, Wouldes TA, Yu TY, Harding JE, CHYLD Study Group (2015) Neonatal glycemia and neurodevelopmental outcomes at 2 years. N Engl J Med 373:1507–1518. https://doi.org/10.1056/NEJMoa1504909
Melamed N, Klinger G, Tenenbaum-Gavish K, Herscovici T, Linder N, Hod M, Yogev Y (2009) Short-term neonatal outcome in low-risk, spontaneous, singleton, late preterm deliveries. Obstet Gynecol 114:253–260. https://doi.org/10.1097/AOG.0b013e3181af6931
Nicholl R (2003) What is the normal range of blood glucose concentrations in healthy term newborns? Arch Dis Child 88:238–239
Peeters D, Lanting CI, van Wouwe JP (2015) Peiling melkvoeding van zuigelingen 2015. TNO, Leiden
Schaefer-Graf UM, Rossi R, Buhrer C, Siebert G, Kjos SL, Dudenhausen JW, Vetter K (2002) Rate and risk factors of hypoglycemia in large-for-gestational-age newborn infants of nondiabetic mothers. Am J Obstet Gynecol 187:913–917
Schifman RB, Howanitz PJ, Souers RJ (2016) Point-of-care glucose critical values: a Q-probes study involving 50 health care facilities and 2349 critical results. Arch Pathol Lab Med 140:119–124. https://doi.org/10.5858/arpa.2015-0058-CP
Shah R, McKinlay CJD, Harding JE (2018) Neonatal hypoglycemia: continuous glucose monitoring. Curr Opin Pediatr 30:1–208. https://doi.org/10.1097/MOP.0000000000000592
Woo HC, Tolosa L, El-Metwally D, Viscardi RM (2014) Glucose monitoring in neonates: need for accurate and non-invasive methods. Arch Dis Child Fetal Neonatal Ed 99:F153–F157. https://doi.org/10.1136/archdischild-2013-304682
Author information
Authors and Affiliations
Contributions
Celine Blank (CB), Jeroen van Dillen (JD), and Marije Hogeveen (MH) planned and implemented the study. CB collected the data. CB and MH did the statistical analyses. CB wrote the first draft of the manuscript. All authors participated to the discussing of the findings and revised the manuscript.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study, formal consent is not required.
Informed consent
This study is a retrospective evaluation of the already collected data; thus, formal consent is not required.
Additional information
Communicated by Patrick Van Reempts
Rights and permissions
Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (http://creativecommons.org/licenses/by/4.0/), which permits unrestricted use, distribution, and reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made.
About this article
Cite this article
Blank, C., van Dillen, J. & Hogeveen, M. Primum non nocere: earlier cessation of glucose monitoring is possible. Eur J Pediatr 177, 1239–1245 (2018). https://doi.org/10.1007/s00431-018-3169-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-018-3169-z