Abstract
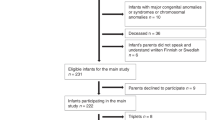
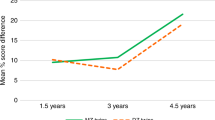
Our aims were to study the effect of birthweight growth discordance (≥20 %) on neuro-developmental outcome of monochorionic and dichorionic twins and to compare the relative effects of foetal growth discordance and prematurity on cognitive outcome. We performed a cross-sectional multicentre prospective follow-up study from a cohort of 948 twin pregnancies. One hundred nineteen birthweight-discordant twin pairs were examined (24 monochorionic pairs) and were matched for gestational age at delivery with 111 concordant control pairs. Participants were assessed with the Bayley Scales between 24 and 42 months of age. Analysis was by paired t test for intra-twin pair differences and by multiple linear regression. Compared to the larger twin of a discordant pair, the smaller twin performed significantly worse in cognition (mean composite cognitive score difference = −1.7, 95 % confidence interval (CI) = 0.3–3.1, p = 0.01) and also in language and motor skills. Prematurity prior to 33 weeks’ gestation, however, had a far greater impact on cognitive outcomes (mean cognitive composite score difference = −5.8, 95 % CI = 1.2–10.5, p = 0.008).
Conclusion: Birthweight growth discordance of ≥20 % confers an independent adverse effect on long-term neuro-development of the smaller twin. However, prior to 33 weeks’ gestation, gestational age at birth adversely affects cognitive development to a greater extent than foetal growth discordance.
What is known: • Growth discordance is a common problem encountered in monochorionic and dichorionic twin pregnancies. • Previous studies have demonstrated adverse developmental outcomes in one or two areas of development. |
What is new: • Growth discordance has a negative impact on all three areas of development: cognition, language and motor skills. • The current study is amongst the first to compare the impact of growth discordance and prematurity on cognitive outcomes. |



Similar content being viewed by others
Abbreviations
- AGA:
-
Appropriately grown for gestational age
- BW:
-
Birthweight
- CI:
-
Confidence interval
- DC:
-
Dichorionic
- ESPRiT (Study):
-
Evaluation of Sonographic Predictors of Restricted Growth in Twins
- MC:
-
Monochorionic
- NOTES (Study):
-
Neuro-Developmental Outcome for Twins of the ESPRiT Study
- SGA:
-
Small for gestational age
- TTTS:
-
Twin-twin transfusion syndrome
- VLBW:
-
Very low birthweight
References
Achenbach T, Rescorla L (2000) The child behaviour checklist, ages 18 months to 5 years
Acton BR, Biggs WS, Creighton DE, Penner KA, Switzer HN, Thomas JH, Joffe AR, Robertson CM (2011) Overestimating neurodevelopment using the Bayley-111 after complex cardiac surgery. J Pediatr 128(4):e794–e800
Bayley N (2006) The Bayley Scales of Infant and Toddler Development, third edition
Blickstein I, Friedman A, Caspi B et al (1989) Ultrasonic prediction of growth discordancy by intertwin difference in abdominal circumference. Int J Gynaecol Obstet 29:121–124
Blickstein I, Lancet M (1988) The growth discordant twin. Obstet Gynecol Surv 43(9):509–515
Breathnach FM, McAuliffe FM, Geary M, Daly S, Higgins JR, Dornan J, Morrison JJ, Burke G, Higgins S, Dicker P, Manning F, Mahony R, Malone FD (2011) Definition of intertwin birth weight discordance. Obstet Gynecol 118(1):94–103
Breathnach FM, McAuliffe FM, Geary M, Daly S, Higgins JR, Dornan J, Morrison JJ, Burke G, Higgins S, Dicker P, Manning F, Carroll S, Malone FD (2012) Optimum timing for planned delivery of uncomplicated monochorionic and dichorionic twin pregnancies. Obstet Gynecol 119(1):50–59
Chyi LJ, Lee HC, Hintz SR, Gould JB, Sutcliffe TL (2008) School outcomes of late preterm infants: special needs and challenges for infants born at 32 to 36 weeks gestation. J Pediatr 153(1):5–6
Datar A, Jacknowitz A (2009) Birth weight effects on children’s mental, motor and physical development: evidence from twins data. Matern Child Health J 13(6):780–794
Edmonds CJ, Isaacs EB, Cole TJ, Rogers MH, Lanigan J, Singhal A, Birbara T, Gringras P, Denton J, Lucas A (2010) The effect of intrauterine growth on verbal IQ scores in childhood: a study of monozygotic twins. Pediatrics 126:e1095–e1101
Fortin A, Rajguru M, Madalenat P, Mahieu-Caputo D (2005) Neurological outcome of children from twin pregnancies. Gynecol Obstet Fertil 33(9):563–569
Goyen TA, Veddovi M, Lui K (2003) Developmental outcome of discordant premature twins at 3 years. Early Hum Dev 73(1):27–37
Hack KEA, Derks JB, Elias SG, Franx A, Roos EJ, Voerman SK, Bode CL, Koopman-Esseboom C, Visser GH (2008) Increased perinatal mortality and morbidity in monochorionic versus dichorionic twin pregnancies: clinical implications of a large Dutch cohort. BJOG 115:58–67
Hack KEA, Koomam-Esseboom C, Derks JB, Elias SG, de Kleine M, Baerts W, Go AT, Schaap AH, van der Hoeven MA, Eggink AJ, Sollie KM, Weisglass-Kuperus N, Visser GH A (2009) Long-term neurodevelopmental outcome of monochorionic and matched dichorionic twins. PLoS One 4(8):e6815
Hagihara S, Matsubara S, Kuwata T, Baba Y, Schimada K, Yamanaka H, Watanabe T, Suzuki M (2003) Discordant Twin: marked vascular communication between separated dichorionic diamniotic placentas. Twin Res 6(4):267–269
Luu TM, Vohr B (2009) Twinning on the brain: the effect on neurodevelopmental outcomes. Am J Med Genet C: Semin Med Genet 151C(2):142–147
Marlow N, Wolke D, Bracewall MA, Samara M (2005) Neurological and developmental disability at 6 years of age after extremely preterm birth. N Engl J Med 353:9–19
Monset-Couchard M, De Bethmann O, Relier J (2004) Long term outcome of small versus appropriate size for gestational age twins/triplets. Arch Dis Child Fetal Neonatal Ed 89(4):F310–F314
Morse SB, Zheng H, Tang Y, Roth J (2009) Early school-age outcomes of late preterm infants. Pediatrics 123(4):e622–e699
Petrini JR, Dias T, McCormack MC, Massolo ML, Green NS, Escobar GJ (2009) Increased risk of adverse neurological development for late preterm infants. J Pediatr 154(2):169–176
Ross GS, Krauss AN, Perlman JM (2012) Physical growth and cognitive abilities in concordant versus discordant birth weight twins at three years old. Early Hum Dev 88(9):753–756
Selman TR, Morris RK, Kilby MD (2011) Management of twin-to-twin transfusion syndrome. Arch Dis Child Fetal Neonatal Ed 96(5):318–320
Shah P, Kingdom J (2011) Long-term neurocognitive outcomes of SGA/IUGR infants. Obstet Gynaecol Reprod Med 21(5):142–146
Soc90 classification. Central Statistics Office. Dublin, Ireland. 2012
Vohr BR, Stephens BE, Higgins RD, Bann CM, Hintz SR, Das A, Newman JE, Peralta-Carcelen M, Yoton K, Dusick AM, Evans PW, Goldstein RF, Ehrenkranz RA, Pappas A, Adams-Chapman I, Wilson-Costello DE, Bauer CR, Bodnar A, Heyne RJ, Vaucher YE, Dillard RG, Acarregui MJ, McGowan EC, Myers GJ, Fuller J (2012) Are outcomes of extremely preterm infants improving? Impact of Bayley assessment on outcomes. J Pediatr 161(2):222–228
Yinon Y, Mazkerath R, Rosentzweig N, Jarus-hakak A, Schiff E, Simchen MJ (2005) Growth restriction as a determinant of outcome in preterm discordant twins. Obstet Gynecol 105(1):80–84
Acknowledgments
The authors wish to thank contributors from The Perinatal Ireland Research Consortium: Dr Michael P Geary, Dr Sean Daly, Dr John Higgins, Dr James Dornan, Dr Gerard Burke, Dr Shane Higgins and Dr Stephen Carroll.
The authors also wish to thank the paediatric collaborators: Dr Louise Gibson, Dr Sinead Harty, Dr Orla Flanagan, Dr John Murphy, Dr Martin White and Dr Siobhan Gallagher.
Finally, thank you to our research nurses and psychologists: Ms Niamh Pearson, Ms Mairead Diviney, Ms Caroline Rawdon, Ms Roisin Reid, Ms Patricia McCreesh and Ms Anne Marie McCarthy.
Author’s contributions
Cecilie Halling (first author): Dr Halling conceptualized and designed the study, coordinated all data collection, drafted the initial manuscript and wrote the final manuscript with contributions from other authors.
John David Corcoran (senior author): Dr Corcoran conceptualized and helped design the study, supervised the study, reviewed and revised the initial manuscript and approved the final manuscript as submitted.
Fergal Malone: Dr Malone conceptualized and helped design the study, reviewed and revised the initial manuscript and approved the final manuscript as submitted.
Fionnuala Breathnach: Dr Breathnach conceptualized and helped design the study, reviewed and revised the initial manuscript and approved the final manuscript as submitted.
Moira Stewart: Dr Stewart conceptualized and helped design the study, revised the initial manuscript and approved the final manuscript as submitted.
Fionnuala McAuliffe: Dr McAuliffe conceptualized and helped design the study, reviewed and revised the initial manuscript and approved the final manuscript as submitted.
John J Morrison: Dr Morrison conceptualized and helped design the study, reviewed and revised the initial manuscript and approved the final manuscript as submitted.
Fiona Manning: Ms Manning conceptualized and helped design the study, helped with the grant application process, reviewed and revised the initial manuscript and approved the final manuscript as submitted.
Patrick Dicker: Mr Dicker conceptualized and helped design the study, designed the data collection instruments, carried out all statistical analysis for the study and approved the final manuscript as submitted.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Funding
This study was funded by the National Children’s Research Centre, Dublin, Ireland, and the ESPRiT study was funded by the Health Research Board Ireland. The funding source had no involvement in the study design, in the collection, analysis and interpretation of the data, in the writing of the manuscript or in the decision to submit the paper.
Conflict of interest
Author A declares that she has no conflict of interest. Author B declares that he has no conflict of interest. Author C declares that she has no conflict of interest. Author D declares that she has no conflict of interest. Author E declares that she has no conflict of interest. Author F declares that he has no conflict of interest. Author G declares that he has no conflict of interest. Author H declares that she has no conflict of interest. Author I declares that he has no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Communicated by Peter de Winter
Rights and permissions
About this article
Cite this article
Halling, C., Malone, F.D., Breathnach, F.M. et al. Neuro-developmental outcome of a large cohort of growth discordant twins. Eur J Pediatr 175, 381–389 (2016). https://doi.org/10.1007/s00431-015-2648-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00431-015-2648-8