Abstract
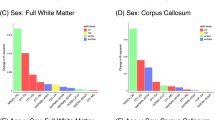
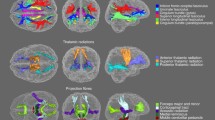
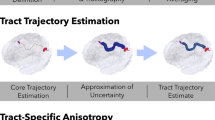
Quantifying the microstructural and macrostructural geometrical features of the human brain’s connections is necessary for understanding normal aging and disease. Here, we examine brain white matter diffusion magnetic resonance imaging data from one cross-sectional and two longitudinal data sets totaling in 1218 subjects and 2459 sessions of people aged 50–97 years. Data was drawn from well-established cohorts, including the Baltimore Longitudinal Study of Aging data set, Cambridge Centre for Ageing Neuroscience data set, and the Vanderbilt Memory & Aging Project. Quantifying 4 microstructural features and, for the first time, 11 macrostructure-based features of volume, area, and length across 120 white matter pathways, we apply linear mixed effect modeling to investigate changes in pathway-specific features over time, and document large age associations within white matter. Conventional diffusion tensor microstructure indices are the most age-sensitive measures, with positive age associations for diffusivities and negative age associations with anisotropies, with similar patterns observed across all pathways. Similarly, pathway shape measures also change with age, with negative age associations for most length, surface area, and volume-based features. A particularly novel finding of this study is that while trends were homogeneous throughout the brain for microstructure features, macrostructural features demonstrated heterogeneity across pathways, whereby several projection, thalamic, and commissural tracts exhibited more decline with age compared to association and limbic tracts. The findings from this large-scale study provide a comprehensive overview of the age-related decline in white matter and demonstrate that macrostructural features may be more sensitive to heterogeneous white matter decline. Therefore, leveraging macrostructural features may be useful for studying aging and could facilitate comparisons in a variety of diseases or abnormal conditions.






Similar content being viewed by others
Data availability
Derived microstructure and macrostructure features, for all pathways and subjects, along with demographic information, are made available at (link upon acceptance) for the CAMCAN data set. Data from VMAP are available by request at http://vmacdata.org/vmap/data-requests. Data from the BLSA are available on request from the BLSA website (http://blsa.nih.gov). All requests are reviewed by the BLSA Data Sharing Proposal Review Committee and may also be subject to approval from the NIH institutional review board.
References
Abe O, Yamasue H, Aoki S et al (2008) Aging in the CNS: comparison of gray/white matter volume and diffusion tensor data. Neurobiol Aging 29(1):102–116. https://doi.org/10.1016/j.neurobiolaging.2006.09.003
Ardekani S, Kumar A, Bartzokis G, Sinha U (2007) Exploratory voxel-based analysis of diffusion indices and hemispheric asymmetry in normal aging. Magn Reson Imaging 25(2):154–167. https://doi.org/10.1016/j.mri.2006.09.045
Beck D, de Lange AG, Maximov II et al (2021) White matter microstructure across the adult lifespan: a mixed longitudinal and cross-sectional study using advanced diffusion models and brain-age prediction. Neuroimage 224:117441. https://doi.org/10.1016/j.neuroimage.2020.117441
Bergfield KL, Hanson KD, Chen K et al (2010) Age-related networks of regional covariance in MRI gray matter: reproducible multivariate patterns in healthy aging. Neuroimage 49(2):1750–1759. https://doi.org/10.1016/j.neuroimage.2009.09.051
Bigham B, Zamanpour SA, Zare H (2022) Alzheimer’s disease neuroimaging I. Features of the superficial white matter as biomarkers for the detection of Alzheimer’s disease and mild cognitive impairment: a diffusion tensor imaging study. Heliyon 8(1):08725. https://doi.org/10.1016/j.heliyon.2022.e08725
Chamberland M, Tax CMW, Jones DK (2018) Meyer’s loop tractography for image-guided surgery depends on imaging protocol and hardware. Neuroimage Clin 20:458–465. https://doi.org/10.1016/j.nicl.2018.08.021
Chandio BQ, Risacher SL, Pestilli F et al (2020) Bundle analytics, a computational framework for investigating the shapes and profiles of brain pathways across populations. Sci Rep 10(1):17149. https://doi.org/10.1038/s41598-020-74054-4
Chang YS, Owen JP, Pojman NJ et al (2015) White matter changes of neurite density and fiber orientation dispersion during human brain maturation. PLoS One 10(6):e0123656. https://doi.org/10.1371/journal.pone.0123656
Coutu JP, Chen JJ, Rosas HD, Salat DH (2014) Non-Gaussian water diffusion in aging white matter. Neurobiol Aging 35(6):1412–1421. https://doi.org/10.1016/j.neurobiolaging.2013.12.001
Cox SR, Ritchie SJ, Tucker-Drob EM et al (2016) Ageing and brain white matter structure in 3513 UK Biobank participants. Nat Commun 7:13629. https://doi.org/10.1038/ncomms13629
Daducci A, Canales-Rodríguez EJ, Descoteaux M et al (2014) Quantitative comparison of reconstruction methods for intra-voxel fiber recovery from diffusion MRI. IEEE Trans Med Imaging 33(2):384–399. https://doi.org/10.1109/TMI.2013.2285500
de Groot M, Ikram MA, Akoudad S et al (2015) Tract-specific white matter degeneration in aging: the Rotterdam study. Alzheimers Dement 11(3):321–330. https://doi.org/10.1016/j.jalz.2014.06.011
Dong JW, Jelescu IO, Ades-Aron B et al (2020) Diffusion MRI biomarkers of white matter microstructure vary nonmonotonically with increasing cerebral amyloid deposition. Neurobiol Aging 89:118–128. https://doi.org/10.1016/j.neurobiolaging.2020.01.009
Fan Q, Tian Q, Ohringer NA et al (2019) Age-related alterations in axonal microstructure in the corpus callosum measured by high-gradient diffusion MRI. Neuroimage 191:325–336. https://doi.org/10.1016/j.neuroimage.2019.02.036
Farrell JA, Landman BA, Jones CK et al (2007) Effects of signal-to-noise ratio on the accuracy and reproducibility of diffusion tensor imaging-derived fractional anisotropy, mean diffusivity, and principal eigenvector measurements at 1.5 T. J Magn Reson Imaging 26(3):756–67. https://doi.org/10.1002/jmri.21053
Fillard P, Descoteaux M, Goh A et al (2011) Quantitative evaluation of 10 tractography algorithms on a realistic diffusion MR phantom. Neuroimage 56(1):220–234. https://doi.org/10.1016/j.neuroimage.2011.01.032
Fortin JP, Parker D, Tunç B et al (2017) Harmonization of multi-site diffusion tensor imaging data. Neuroimage 11(161):149–170. https://doi.org/10.1016/j.neuroimage.2017.08.047
Giorgio A, Santelli L, Tomassini V et al (2010) Age-related changes in grey and white matter structure throughout adulthood. Neuroimage 51(3):943–951. https://doi.org/10.1016/j.neuroimage.2010.03.004
Guevara P, Duclap D, Poupon C et al (2012) Automatic fiber bundle segmentation in massive tractography datasets using a multi-subject bundle atlas. Neuroimage 61(4):1083–1099. https://doi.org/10.1016/j.neuroimage.2012.02.071
Guo Y, Zhang Z, Zhou B et al (2014) Grey-matter volume as a potential feature for the classification of Alzheimer’s disease and mild cognitive impairment: an exploratory study. Neurosci Bull 30(3):477–489. https://doi.org/10.1007/s12264-013-1432-x
Isaac Tseng WY, Hsu YC, Chen CL et al (2021) Microstructural differences in white matter tracts across middle to late adulthood: a diffusion MRI study on 7167 UK Biobank participants. Neurobiol Aging 98:160–172. https://doi.org/10.1016/j.neurobiolaging.2020.10.006
Jefferson AL, Gifford KA, Acosta LM et al (2016) The Vanderbilt memory & aging project: study design and baseline cohort overview. J Alzheimers Dis 52(2):539–559. https://doi.org/10.3233/JAD-150914
Jones DK (2010) Diffusion MRI: theory, methods, and application. Oxford University Press, Oxford, p 767
Jones DK, Basser PJ (2004) “Squashing peanuts and smashing pumpkins”: how noise distorts diffusion-weighted MR data. Magn Reson Med 52(5):979–993. https://doi.org/10.1002/mrm.20283
Jorge L, Martins R, Canario N et al (2021) Investigating the spatial associations between amyloid-beta deposition, grey matter volume, and neuroinflammation in Alzheimer’s disease. J Alzheimers Dis 80(1):113–132. https://doi.org/10.3233/JAD-200840
Kimmel CL, Alhassoon OM, Wollman SC et al (2016) Age-related parieto-occipital and other gray matter changes in borderline personality disorder: a meta-analysis of cortical and subcortical structures. Psychiatry Res Neuroimaging 251:15–25. https://doi.org/10.1016/j.pscychresns.2016.04.005
Landman BA, Farrell JA, Jones CK, Smith SA, Prince JL, Mori S (2007) Effects of diffusion weighting schemes on the reproducibility of DTI-derived fractional anisotropy, mean diffusivity, and principal eigenvector measurements at 1.5T. Neuroimage 36(4):1123–38. https://doi.org/10.1016/j.neuroimage.2007.02.056
Lawrence KE, Nabulsi L, Santhalingam V et al (2021) Age and sex effects on advanced white matter microstructure measures in 15,628 older adults: a UK biobank study. Brain Imaging Behav 15(6):2813–2823. https://doi.org/10.1007/s11682-021-00548-y
Lebel C, Walker L, Leemans A, Phillips L, Beaulieu C (2008) Microstructural maturation of the human brain from childhood to adulthood. Neuroimage 40(3):1044–1055. https://doi.org/10.1016/j.neuroimage.2007.12.053
Lebel C, Gee M, Camicioli R, Wieler M, Martin W, Beaulieu C (2012) Diffusion tensor imaging of white matter tract evolution over the lifespan. Neuroimage 60(1):340–352. https://doi.org/10.1016/j.neuroimage.2011.11.094
Mirzaalian H, Ning L, Savadjiev P et al (2016) Inter-site and inter-scanner diffusion MRI data harmonization. Neuroimage 07(135):311–323. https://doi.org/10.1016/j.neuroimage.2016.04.041
Molloy CJ, Nugent S, Bokde ALW (2021) Alterations in diffusion measures of white matter integrity associated with healthy aging. J Gerontol A Biol Sci Med Sci 76(6):945–954. https://doi.org/10.1093/gerona/glz289
Nicolas R, Hiba B, Dilharreguy B et al (2020) Changes over time of diffusion MRI in the white matter of aging brain, a good predictor of verbal recall. Front Aging Neurosci 12:218. https://doi.org/10.3389/fnagi.2020.00218
Ning L, Bonet-Carne E, Grussu F et al (2020) Cross-scanner and cross-protocol multi-shell diffusion MRI data harmonization: algorithms and results. Neuroimage 221:117128. https://doi.org/10.1016/j.neuroimage.2020.117128
Pfefferbaum A, Lim KO, Zipursky RB et al (1992) Brain gray and white matter volume loss accelerates with aging in chronic alcoholics: a quantitative MRI study. Alcohol Clin Exp Res 16(6):1078–1089. https://doi.org/10.1111/j.1530-0277.1992.tb00702.x
Ramanoel S, Hoyau E, Kauffmann L et al (2018) Gray matter volume and cognitive performance during normal aging. A Voxel-based morphometry study. Front Aging Neurosci 10:235. https://doi.org/10.3389/fnagi.2018.00235
Rheault F, De Benedictis A, Daducci A et al (2020) Tractostorm: the what, why, and how of tractography dissection reproducibility. Hum Brain Mapp 41(7):1859–1874. https://doi.org/10.1002/hbm.24917
Schilling KG, Janve V, Gao Y, Stepniewska I, Landman BA, Anderson AW (2018) Histological validation of diffusion MRI fiber orientation distributions and dispersion. Neuroimage 01(165):200–221. https://doi.org/10.1016/j.neuroimage.2017.10.046
Schilling KG, Rheault F, Petit L et al (2021a) Tractography dissection variability: what happens when 42 groups dissect 14 white matter bundles on the same dataset? Neuroimage 243:118502. https://doi.org/10.1016/j.neuroimage.2021.118502
Schilling KG, Tax CMW, Rheault F et al (2021b) Fiber tractography bundle segmentation depends on scanner effects, acquisition, diffusion sensitization, and bundle segmentation workflow. bioRxiv. https://doi.org/10.1101/2021.03.17.435872
Schilling KG, Tax CMW, Rheault F et al (2021c) Fiber tractography bundle segmentation depends on scanner effects, vendor effects, acquisition resolution, diffusion sampling scheme, diffusion sensitization, and bundle segmentation workflow. Neuroimage 242:118451. https://doi.org/10.1016/j.neuroimage.2021.118451
Storsve AB, Fjell AM, Yendiki A, Walhovd KB (2016) Longitudinal changes in white matter tract integrity across the adult lifespan and its relation to cortical thinning. PLoS One 11(6):e0156770. https://doi.org/10.1371/journal.pone.0156770
Taki Y, Thyreau B, Kinomura S et al (2011) Correlations among brain gray matter volumes, age, gender, and hemisphere in healthy individuals. PLoS One 6(7):e22734. https://doi.org/10.1371/journal.pone.0022734
Tax CM, Grussu F, Kaden E et al (2019) Cross-scanner and cross-protocol diffusion MRI data harmonisation: a benchmark database and evaluation of algorithms. Neuroimage. https://doi.org/10.1016/j.neuroimage.2019.01.077
Taylor JR, Williams N, Cusack R et al (2017) The Cambridge Centre for Ageing and Neuroscience (Cam-CAN) data repository: structural and functional MRI, MEG, and cognitive data from a cross-sectional adult lifespan sample. Neuroimage 144(Pt B):262–269. https://doi.org/10.1016/j.neuroimage.2015.09.018
Terribilli D, Schaufelberger MS, Duran FL et al (2011) Age-related gray matter volume changes in the brain during non-elderly adulthood. Neurobiol Aging 32(2):354–368. https://doi.org/10.1016/j.neurobiolaging.2009.02.008
Toschi N, Gisbert RA, Passamonti L, Canals S, De Santis S (2020) Multishell diffusion imaging reveals sex-specific trajectories of early white matter degeneration in normal aging. Neurobiol Aging 02(86):191–200. https://doi.org/10.1016/j.neurobiolaging.2019.11.014
Tournier JD, Smith R, Raffelt D et al (2019) MRtrix3: a fast, flexible and open software framework for medical image processing and visualisation. Neuroimage 202:116137. https://doi.org/10.1016/j.neuroimage.2019.116137
Wang J, Knol MJ, Tiulpin A et al (2019) Gray matter age prediction as a biomarker for risk of dementia. Proc Natl Acad Sci USA 116(42):21213–21218. https://doi.org/10.1073/pnas.1902376116
Warrington S, Bryant KL, Khrapitchev AA et al (2020) XTRACT—standardised protocols for automated tractography in the human and macaque brain. Neuroimage 217:116923. https://doi.org/10.1016/j.neuroimage.2020.116923
Wasserthal J, Neher P, Maier-Hein KH (2018) TractSeg—fast and accurate white matter tract segmentation. Neuroimage 183:239–253. https://doi.org/10.1016/j.neuroimage.2018.07.070
Wheeler-Kingshott CA, Cercignani M (2009) About, “axial” and “radial” diffusivities. Magn Reson Med 61(5):1255–1260. https://doi.org/10.1002/mrm.21965
Williams OA, An Y, Beason-Held L et al (2019) Vascular burden and APOE epsilon4 are associated with white matter microstructural decline in cognitively normal older adults. Neuroimage 188:572–583. https://doi.org/10.1016/j.neuroimage.2018.12.009
Winter M, Tallantyre EC, Brice TAW, Robertson NP, Jones DK, Chamberland M (2021) Tract-specific MRI measures explain learning and recall differences in multiple sclerosis. Brain Commun. https://doi.org/10.1093/braincomms/fcab065
Yap QJ, Teh I, Fusar-Poli P, Sum MY, Kuswanto C, Sim K (2013) Tracking cerebral white matter changes across the lifespan: insights from diffusion tensor imaging studies. J Neural Transm (Vienna) 120(9):1369–1395. https://doi.org/10.1007/s00702-013-0971-7
Yeh F-C (2020) Shape analysis of the human association pathways. bioRxiv. https://doi.org/10.1101/2020.04.19.049544
Yeh FC, Wedeen VJ, Tseng WY (2010) Generalized q-sampling imaging. IEEE Trans Med Imaging 29(9):1626–1635. https://doi.org/10.1109/TMI.2010.2045126
Yeh FC, Verstynen TD, Wang Y, Fernandez-Miranda JC, Tseng WY (2013) Deterministic diffusion fiber tracking improved by quantitative anisotropy. PLoS One 8(11):e80713. https://doi.org/10.1371/journal.pone.0080713
Yeh FC, Panesar S, Fernandes D et al (2018) Population-averaged atlas of the macroscale human structural connectome and its network topology. Neuroimage 178:57–68. https://doi.org/10.1016/j.neuroimage.2018.05.027
Yeh FC, Panesar S, Barrios J et al (2019) Automatic removal of false connections in diffusion MRI tractography using topology-informed pruning (TIP). Neurotherapeutics 16(1):52–58. https://doi.org/10.1007/s13311-018-0663-y
Yendiki A, Panneck P, Srinivasan P et al (2011) Automated probabilistic reconstruction of white-matter pathways in health and disease using an atlas of the underlying anatomy. Front Neuroinform 5:23. https://doi.org/10.3389/fninf.2011.00023
Zuo N, Hu T, Liu H, Sui J, Liu Y, Jiang T (2021) Gray matter-based age prediction characterizes different regional patterns. Neurosci Bull 37(1):94–98. https://doi.org/10.1007/s12264-020-00558-8
Funding
This work was supported by the National Science Foundation Career Award #1452485, the National Institutes of Health under award numbers R01EB017230, K01AG073584, and in part by ViSE/VICTR VR3029 and the National Center for Research Resources, Grant UL1 RR024975-01.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Data collection was performed by the Baltimore Longitudinal Study of Aging at the National Institutes of Aging, and the Vanderbilt Memory & Aging Project (VMAP). All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
All human data sets from Vanderbilt University were acquired after informed consent under supervision of the appropriate Institutional Review Board. All additional data sets are freely available and unrestricted for non-commercial research purposes. This study accessed only de-identified patient information.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Appendix
Appendix
The bundles resulting from each segmentation pipeline are given as a list below, with acronyms used in the text.
TractSeg: Arcuate fascicle left (AF_L); Arcuate fascicle right (AF_R); Anterior Thalamic Radiation left (ATR_L); Thalamic Radiation right; (ATR_R); Commissure Anterior (CA); Rostrum (CC_1; Genu (CC_2); Rostral body (Premotor) (CC_3); Anterior midbody (Primary Motor) (CC_4); Posterior midbody (Primary Somatosensory) (CC_5); Isthmus (CC_6); Splenium (CC_7); Corpus Callosum—all (CC); Cingulum left (CG_L); Cingulum right (CG_R); Corticospinal tract left (CST_L); Corticospinal tract right (CST_R); Fronto-pontine tract left (FPT_L); Fronto-pontine tract right (FPT_R); Fornix left (FX_L); Fornix right (FX_R); Inferior cerebellar peduncle left (ICP_L); Inferior cerebellar peduncle right (ICP_R); Inferior occipito-frontal fascicle left (IFO_L); Inferior occipito-frontal fascicle right (IFO_R); Inferior longitudinal fascicle left (ILF_L); Inferior longitudinal fascicle right (ILF_R); Middle cerebellar peduncle (MCP); Middle longitudinal fascicle left (MLF_L); Middle longitudinal fascicle right (MLF_R); Optic radiation left (OR_L); Optic radiation right (OR_R); Parieto-occipital pontine left (POPT_L); Parieto-occipital pontine right (POPT_R); Superior cerebellar peduncle left (SCP_L); Superior cerebellar peduncle right (SCP_R); Superior longitudinal fascicle III left SLF_III_L); Superior longitudinal fascicle III right (SLF_III_R); Superior longitudinal fascicle II left (SLF_II_L); Superior longitudinal fascicle II right (SLF_II_R); Superior longitudinal fascicle I left (SLF_I_L); Superior longitudinal fascicle I right (SLF_I_R); Striato-fronto-orbital left (ST_FO_L); Striato-fronto-orbital right (ST_FO_R); Striato-occipital left (ST_OCC_L); Striato-occipital right (ST_OCC_R); Striato-parietal left (ST_PAR_L); Striato-parietal right (ST_PAR_R); Striato-postcentral left (ST_POSTC_L); Striato-postcentral right (ST_POSTC_R); Striato-precentral left (ST_PREC_L); Striato-precentral right (ST_PREC_R); Striato-prefrontal left (ST_PREF_L); Striato-prefrontal right (ST_PREF_R); Striato-premotor left (ST_PREM_L); Striato-premotor right (ST_PREM_R); Thalamo-occipital left (T_OCC_L); Thalamo-occipital right (T_OCC_R); Thalamo-parietal left (T_PAR_L); Thalamo-parietal right (T_PAR_R); Thalamo-postcentral left (T_POSTC_L); Thalamo-postcentral right (T_POSTC_R); Thalamo-precentral left (T_PREC_L); Thalamo-precentral right (T_PREC_R); Thalamo-prefrontal left (T_PREF_L); Thalamo-prefrontal right (T_PREF_R); Thalamo-premotor left (T_PREM_L); Thalamo-premotor right (T_PREM_R); Uncinate fascicle left (UF_L); Uncinate fascicle right (UF_R).
ATR: Arcuate_Fasciculus_L (AF_L); Arcuate Fasciculus R (AF_R); Cortico Spinal Tract L (CST_L); Cortico Spinal Tract R (CST_R); Cortico Striatal Pathway L (CS_L); Cortico Striatal Pathway R (CS_R); Corticobulbar Tract L (CBT_L); Corticobulbar Tract R (CBT_R); Corticopontine Tract L (CPT_L); Corticopontine Tract R (CPT_R); Corticothalamic Pathway L (CTP_L); Corticothalamic Pathway R (CTP_R); Inferior Cerebellar Peduncle L (ICP_L); Inferior Cerebellar Peduncle R (ICP_R); Inferior Fronto Occipital Fasciculus L (IFOF_L); Inferior Fronto Occipital Fasciculus R (IFOF_R); Inferior Longitudinal Fasciculus L (ILF_L); Inferior Longitudinal Fasciculus R (ILF_R); Optic Radiation L (OR_L); Optic Radiation R (OR_R); Middle Longitudinal Fasciculus L (MdLF_L); Middle Longitudinal Fasciculus R (MdLF_R); Uncinate Fasciculus L (UF_L); Uncinate Fasciculus R (UF_R).
Rights and permissions
About this article
Cite this article
Schilling, K.G., Archer, D., Yeh, FC. et al. Aging and white matter microstructure and macrostructure: a longitudinal multi-site diffusion MRI study of 1218 participants. Brain Struct Funct 227, 2111–2125 (2022). https://doi.org/10.1007/s00429-022-02503-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-022-02503-z