Abstract
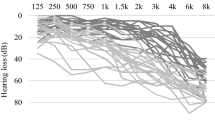
Older adults with hearing loss experience significant difficulties understanding speech in noise, perhaps due in part to limited benefit from supporting executive functions that enable the use of environmental cues signaling changes in listening conditions. Here we examined the degree to which 41 older adults (60.56–86.25 years) exhibited cortical responses to informative listening difficulty cues that communicated the listening difficulty for each trial compared to neutral cues that were uninformative of listening difficulty. Word recognition was significantly higher for informative compared to uninformative cues in a + 10 dB signal-to-noise ratio (SNR) condition, and response latencies were significantly shorter for informative cues in the + 10 dB SNR and the more-challenging + 2 dB SNR conditions. Informative cues were associated with elevated blood oxygenation level-dependent contrast in visual and parietal cortex. A cue–SNR interaction effect was observed in the cingulo-opercular (CO) network, such that activity only differed between SNR conditions when an informative cue was presented. That is, participants used the informative cues to prepare for changes in listening difficulty from one trial to the next. This cue–SNR interaction effect was driven by older adults with more low-frequency hearing loss and was not observed for those with more high-frequency hearing loss, poorer set-shifting task performance, and lower frontal operculum gray matter volume. These results suggest that proactive strategies for engaging CO adaptive control may be important for older adults with high-frequency hearing loss to optimize speech recognition in changing and challenging listening conditions.






Similar content being viewed by others
References
Aarts E, Roelofs A, van Turennout M (2008) Anticipatory activity in anterior cingulate cortex can be independent of conflict and error likelihood. J Neurosci 28(18):4671–4678. https://doi.org/10.1523/JNEUROSCI.4400-07.2008
Alain C, Du Y, Bernstein LJ, Barten T, Banai K (2018) Listening under difficult conditions: an activation likelihood estimation meta-analysis. Hum Brain Mapp 39(7):2695–2709. https://doi.org/10.1002/hbm.24031
Allen PD, Eddins DA (2010) Presbycusis phenotypes form a heterogeneous continuum when ordered by degree and configuration of hearing loss. Hear Res 264(1–2):10–20. https://doi.org/10.1016/j.heares.2010.02.001
Arulpragasam AR, Cooper JA, Nuutinen MR, Treadway MT (2018) Corticoinsular circuits encode subjective value expectation and violation for effortful goal-directed behavior. Proc Natl Acad Sci USA 115(22):E5233–E5242. https://doi.org/10.1073/pnas.1800444115
Avants BB, Tustison N, Song G (2009) Advanced normalization tools (ANTS). Insight J 2:1–35
Belkhiria C, Vergara RC, San Martín S, Leiva A, Marcenaro B, Martinez M, Delgado C, Delano PH (2019) Cingulate cortex atrophy is associated with hearing loss in presbycusis with cochlear amplifier dysfunction. Front Aging Neurosci 11:97. https://doi.org/10.3389/fnagi.2019.00097
Best V, Ozmeral EJ, Shinn-Cunningham BG (2007) Visually-guided attention enhances target identification in a complex auditory scene. J Assoc Res Otolaryngol 8(2):294–304. https://doi.org/10.1007/s10162-007-0073-z
Best V, Marrone N, Mason CR, Kidd G Jr, Shinn-Cunningham BG (2009) Effects of sensorineural hearing loss on visually guided attention in a multitalker environment. J Assoc Res Otolaryngol 10(1):142–149. https://doi.org/10.1007/s10162-008-0146-7
Bidelman GM, Mahmud MS, Yeasin M, Shen D, Arnott SR, Alain C (2019) Age-related hearing loss increases full-brain connectivity while reversing directed signaling within the dorsal–ventral pathway for speech. Brain Struct Funct 224:2661–2676. https://doi.org/10.1007/s00429-019-01922-9
Calhoun VD, Adali T, Pearlson GD, Pekar JJ (2001) A method for making group inferences from functional MRI data using independent component analysis. Hum Brain Mapp 14(3):140–151. https://doi.org/10.1002/hbm.1048
Cavanagh JF, Wiecki TV, Cohen MX, Figueroa CM, Samanta J, Sherman SJ, Frank MJ (2011) Subthalamic nucleus stimulation reverses mediofrontal influence over decision threshold. Nat Neurosci 14:1462–1469. https://doi.org/10.1038/nn.2925
Chauvin JJ, Gillebert CR, Rohenkohl G, Humphreys GW, Nobre AC (2016) Temporal orienting of attention can be preserved in normal aging. Psychol Aging 31(5):442–455. https://doi.org/10.1037/pag0000105
Chiarello C, Vaden KI Jr, Eckert MA (2018) Orthographic influence on spoken word identification: behavioral and fMRI evidence. Neuropsychologia 111:103–111. https://doi.org/10.1016/j.neuropsychologia.2018.01.032
Correa A, Rao A, Nobre AC (2009) Anticipating conflict facilitates controlled stimulus-response selection. J Cogn Neurosci 21(8):1461–1472. https://doi.org/10.1162/jocn.2009.21136
Croxson PL, Walton ME, O’Reilly JX, Behrens TE, Rushworth MF (2009) Effort-based cost-benefit valuation and the human brain. J Neurosci 29(14):4531–4541. https://doi.org/10.1523/JNEUROSCI.4515-08.2009
Davis GJ, Gibson BS (2012) Going rogue in the spatial cuing paradigm: high spatial validity is insufficient to elicit voluntary shifts of attention. J Exp Psychol Hum Percept Perform 38(5):1192–1201. https://doi.org/10.1037/a0027595
Deng Y, Reinhart RM, Choi I, Shinn-Cunningham BG (2019) Causal links between parietal alpha activity and spatial auditory attention. Elife. https://doi.org/10.7554/eLife.51184
Dosenbach NU, Fair DA, Cohen AL, Schlaggar BL, Petersen SE (2008) A dual-networks architecture of top–down control. Trends Cogn Sci 12(3):99–105. https://doi.org/10.1016/j.tics.2008.01.001
Dubno JR, Lee FS, Matthews LJ, Ahlstrom JB, Horwitz AR, Mills JH (2008) Longitudinal changes in speech recognition in older persons. J Acoust Soc Am 123(1):462–475. https://doi.org/10.1121/1.2817362
Dubno JR, Eckert MA, Lee FS, Matthews LJ, Schmiedt RA (2013) Classifying human audiometric phenotypes of age-related hearing loss from animal models. J Assoc Res Otolaryngol 14(5):687–701. https://doi.org/10.1007/s10162-013-0396-x
Eckert MA, Cute SL, Vaden KI Jr, Kuchinsky SE, Dubno JR (2012) Auditory cortex signs of age-related hearing loss. J Assoc Res Otolaryngol 13(5):703–713. https://doi.org/10.1007/s10162-012-0332-5
Eckert MA, Teubner-Rhodes S, Vaden KI Jr (2016) Is listening in noise worth it? The neurobiology of speech recognition in challenging listening conditions. Ear Hear 37(Suppl 1):101S-110S. https://doi.org/10.1097/AUD.0000000000000300
Eckert MA, Vaden KI Jr, Dubno JR (2019) Age-related hearing loss associations with changes in brain morphology. Trends Hear 23:2331216519857267. https://doi.org/10.1177/2331216519857267
Eckert MA, Harris KC, Lang H, Lewis MA, Schmiedt RA, Schulte BA, Steel KP, Vaden KI Jr, Dubno JR (2020) Translational and interdisciplinary insights into presbyacusis: a multidimensional disease. Hear Res. https://doi.org/10.1016/j.heares.2020.108109
Erb J, Obleser J (2013) Upregulation of cognitive control networks in older adults’ speech comprehension. Front Syst Neurosci 7:116. https://doi.org/10.3389/fnsys.2013.00116
Erhardt EB, Rachakonda S, Bedrick EJ, Allen EA, Adali T, Calhoun VD (2011) Comparison of multi-subject ICA methods for analysis of fMRI data. Hum Brain Mapp 32(12):2075–2095. https://doi.org/10.1002/hbm.21170
Eysenck MW, Calvo MG (1992) Anxiety and performance: the processing efficiency theory. Cogn Emot 6(6):409–434. https://doi.org/10.1037/1528-3542.7.2.336
Folstein MF, Robins LN, Helzer JE (1983) The mini-mental state examination. Arch Gen Psychiatry 40(7):812. https://doi.org/10.1001/archpsyc.1983.01790060110016
Gaser C, Dahnke R (2016) CAT-A Computational Anatomty Toolbox for the analysis of structural MRI data. Paper presented at the Human Brain Mapping. Geneva, Switzerland
Gatehouse S, Akeroyd MA (2008) The effects of cueing temporal and spatial attention on word recognition in a complex listening task in hearing-impaired listeners. Trends Amplif 12(2):145–161. https://doi.org/10.1177/1084713808317395
Grady C (2012) The cognitive neuroscience of ageing. Nat Rev Neurosci 13(7):491–505. https://doi.org/10.1038/nrn3256
Hammerer D, Li SC, Muller V, Lindenberger U (2010) An electrophysiological study of response conflict processing across the lifespan: assessing the roles of conflict monitoring, cue utilization, response anticipation, and response suppression. Neuropsychologia 48(11):3305–3316. https://doi.org/10.1016/j.neuropsychologia.2010.07.014
Hancock PA, Meshkati N, Robertson MM (1985) Physiological reflections of mental workload. Aviat Space Environ Med 56(11):1110–1114
Haupt M, Ruiz-Rizzo AL, Sorg C, Finke K (2019) Phasic alerting effects on visual processing speed are associated with intrinsic functional connectivity in the cingulo-opercular network. Neuroimage 196:216–226. https://doi.org/10.1016/j.neuroimage.2019.04.019
Heideman SG, Rohenkohl G, Chauvin JJ, Palmer CE, van Ede F, Nobre AC (2018) Anticipatory neural dynamics of spatial-temporal orienting of attention in younger and older adults. Neuroimage 178:46–56. https://doi.org/10.1016/j.neuroimage.2018.05.002
Hill KT, Miller LM (2010) Auditory attentional control and selection during cocktail party listening. Cereb Cortex 20(3):583–590. https://doi.org/10.1093/cercor/bhp124
Holmes E, Kitterick PT, Summerfield AQ (2018) Cueing listeners to attend to a target talker progressively improves word report as the duration of the cue-target interval lengthens to 2000 ms. Atten Percept Psychophys 80(6):1520–1538. https://doi.org/10.3758/s13414-018-1531-x
Humes LE, Young LA (2016) Sensory-cognitive interactions in older adults. Ear Hear 37(Suppl 1):52S-61S. https://doi.org/10.1097/AUD.0000000000000303
Jayakody DM, Friedland PL, Nel E, Martins RN, Atlas MD, Sohrabi HR (2017) Impact of cochlear implantation on cognitive functions of older adults: pilot test results. Otol Neurotol 38(8):e289–e295
Jiang J, Beck J, Heller K, Egner T (2015) An insula-frontostriatal network mediates flexible cognitive control by adaptively predicting changing control demands. Nat Commun 6:8165. https://doi.org/10.1038/ncomms9165
Kiesel A, Steinhauser M, Wendt M, Falkenstein M, Jost K, Philipp AM, Koch I (2010) Control and interference in task switching–a review. Psychol Bull 136(5):849–874. https://doi.org/10.1037/a0019842
Kitterick PT, Bailey PJ, Summerfield AQ (2010) Benefits of knowing who, where, and when in multi-talker listening. J Acoust Soc Am 127(4):2498–2508. https://doi.org/10.1121/1.3327507
Knutson B, Greer SM (2008) Anticipatory affect: neural correlates and consequences for choice. Philos Trans R Soc Lond B Biol Sci 363(1511):3771–3786. https://doi.org/10.1098/rstb.2008.0155
Kongs SK, Thompson LL, Iverson GL, Heaton RK (2000) Wisconsin card sorting test, 64 Card Version. Psychological Assessment Resources, Lutz
Kuchinsky SE, Vaden KI Jr, Keren NI, Harris KC, Ahlstrom JB, Dubno JR, Eckert MA (2012) Word intelligibility and age predict visual cortex activity during word listening. Cereb Cortex 22(6):1360–1371. https://doi.org/10.1093/cercor/bhr211
Kuchinsky SE, Vaden KI Jr, Ahlstrom JB, Cute SL, Humes LE, Dubno JR, Eckert MA (2016) Task-related vigilance during word recognition in noise for older adults with hearing loss. Exp Aging Res 42(1):50–66. https://doi.org/10.1080/0361073X.2016.1108712
Lewellen MJ, Goldinger SD, Pisoni DB, Greene BG (1993) Lexical familiarity and processing efficiency: individual differences in naming, lexical decision, and semantic categorization. J Exp Psychol Gen 122(3):316–330. https://doi.org/10.1037//0096-3445.122.3.316
Lin FR, Ferrucci L, Metter EJ, An Y, Zonderman AB, Resnick SM (2011) Hearing loss and cognition in the Baltimore longitudinal study of aging. Neuropsychology 25(6):763. https://doi.org/10.1037/a0024238
Lin F, Ferrucci L, An Y, Goh J, Doshi J, Metter E, Davatzikos C, Kraut MA, Resnick SM (2014) Association of hearing impairment with brain volume changes in older adults. Neuroimage 90:84–92. https://doi.org/10.1016/j.neuroimage.2013.12.059
Loughrey DG, Kelly ME, Kelley GA, Brennan S, Lawlor BA (2018) Association of age-related hearing loss with cognitive function, cognitive impairment, and dementia: a systematic review and meta-analysis. JAMA Otolaryngol Head Neck Surg 144(2):115–126. https://doi.org/10.1001/jamaoto.2017.2513
Luks TL, Simpson GV, Feiwell RJ, Miller WL (2002) Evidence for anterior cingulate cortex involvement in monitoring preparatory attentional set. Neuroimage 17(2):792–802
McGuire JT, Botvinick MM (2010) Prefrontal cortex, cognitive control, and the registration of decision costs. Proc Natl Acad Sci USA 107(17):7922–7926. https://doi.org/10.1073/pnas.0910662107
Nair S, Nenert RE, Allendorfer JB, Goodman AM, Vannest J, Mirman D, Szaflarski JP (2019) Sex, age, and handedness modulate the neural correlates of active learning. Front Neurosci 13:961. https://doi.org/10.3389/fnins.2019.00961
Oberfeld D, Kloeckner-Nowotny F (2016) Individual differences in selective attention predict speech identification at a cocktail party. Elife 5:e16747. https://doi.org/10.7554/eLife.16747
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9(1):97–113. https://doi.org/10.1016/0028-3932(71)90067-4
Pauszek JR, Gibson BS (2018) The Least Costs Hypothesis: a rational analysis approach to the voluntary symbolic control of attention. J Exp Psychol Hum Percept Perform 44(8):1199–1215. https://doi.org/10.1037/xhp0000527
Paxton JL, Barch DM, Racine CA, Braver TS (2008) Cognitive control, goal maintenance, and prefrontal function in healthy aging. Cereb Cortex 18(5):1010–1028. https://doi.org/10.1093/cercor/bhm135
Peelle JE (2014) Methodological challenges and solutions in auditory functional magnetic resonance imaging. Front Neurosci 8:253. https://doi.org/10.3389/fnins.2014.00253
Peelle JE (2018) Listening effort: How the cognitive consequences of acoustic challenge are reflected in brain and behavior. Ear Hear 39(2):204–214. https://doi.org/10.1097/AUD.0000000000000494
Perrachione TK, Ghosh SS (2013) Optimized design and analysis of sparse-sampling FMRI experiments. Front Neurosci 7:55. https://doi.org/10.3389/fnins.2013.00055
Pichora-Fuller MK, Kramer SE, Eckert MA, Edwards B, Hornsby BW, Humes LE, Lemke U, Lunner T, Matthen M, Mackersie CL, Naylor G, Phillips NA, Richter M, Rudner M, Sommers MS, Tremblay KL, Wingfield A (2016) Hearing impairment and cognitive energy: the framework for understanding effortful listening (FUEL). Ear Hear 37(Suppl 1):5S-27S. https://doi.org/10.1097/AUD.0000000000000312
Prevost C, Pessiglione M, Metereau E, Clery-Melin ML, Dreher JC (2010) Separate valuation subsystems for delay and effort decision costs. J Neurosci 30(42):14080–14090. https://doi.org/10.1523/JNEUROSCI.2752-10.2010
Price CN, Alain C, Bidelman GM (2019) Auditory-frontal channeling in α and β bands is altered by age-related hearing loss and relates to speech perception in noise. Neurosci 15(423):18–28. https://doi.org/10.1016/j.neuroscience.2019.10.044
Rahnev D, Nee DE, Riddle J, Larson AS, D’Esposito M (2016) Causal evidence for frontal cortex organization for perceptual decision making. Proc Natl Acad Sci USA 113:6059–6064. https://doi.org/10.1073/pnas.1522551113
Ridderinkhof KR, Span MM, van der Molen MW (2002) Perseverative behavior and adaptive control in older adults: performance monitoring, rule induction, and set shifting. Brain Cogn 49(3):382–401. https://doi.org/10.1006/brcg.2001.1506
Rigters SC, Bos D, Metselaar M, Roshchupkin GV, Baatenburg de Jong RJ, Ikram MA, Vernooij MW, Goedegebure A (2017) Hearing impairment is associated with smaller brain volume in aging. Front Aging Neurosci 9:2. https://doi.org/10.3389/fnagi.2017.00002
Ronnberg J, Lunner T, Zekveld A, Sorqvist P, Danielsson H, Lyxell B, Dahlstrom O, Signoret C, Stenfelt S, Pichora-Fuller MK, Rudner M (2013) The Ease of Language Understanding (ELU) model: theoretical, empirical, and clinical advances. Front Syst Neurosci 7:31. https://doi.org/10.3389/fnsys.2013.00031
Rudner M, Seeto M, Keidser G, Johnson B, Ronnberg J (2019) Poorer speech reception threshold in noise Is associated with lower brain volume in auditory and cognitive processing regions. J Speech Lang Hear Res 62(4S):1117–1130. https://doi.org/10.1044/2018_JSLHR-H-ASCC7-18-0142
Ruge H, Jamadar S, Zimmermann U, Karayanidis F (2013) The many faces of preparatory control in task switching: reviewing a decade of fMRI research. Hum Brain Mapp 34(1):12–35. https://doi.org/10.1002/hbm.21420
Sadaghiani S, D’Esposito M (2015) Functional characterization of the cingulo-opercular network in the maintenance of tonic alertness. Cereb Cortex 25(9):2763–2773. https://doi.org/10.1093/cercor/bhu072
Saliasi E, Geerligs L, Lorist MM, Maurits NM (2014) Neural correlates associated with successful working memory performance in older adults as revealed by spatial ICA. PLoS ONE 9(6):e99250. https://doi.org/10.1371/journal.pone.0099250
Schulte T, Muller-Oehring EM, Chanraud S, Rosenbloom MJ, Pfefferbaum A, Sullivan EV (2011) Age-related reorganization of functional networks for successful conflict resolution: a combined functional and structural MRI study. Neurobiol Aging 32(11):2075–2090. https://doi.org/10.1016/j.neurobiolaging.2009.12.002
Shende SA, Nguyen LT, Lydon EA, Husain FT, Mudar RA (2021) Cognitive flexibility and inhibition in individuals with age-related hearing loss. Geriatrics 6(1):22. https://doi.org/10.3389/fnagi.2017.00002
Shomstein S, Yantis S (2006) Parietal cortex mediates voluntary control of spatial and nonspatial auditory attention. J Neurosci 26(2):435–439. https://doi.org/10.1523/JNEUROSCI.4408-05.2006
Teubner-Rhodes S, Vaden KI Jr, Dubno JR, Eckert MA (2017) Cognitive persistence: development and validation of a novel measure from the Wisconsin Card Sorting Test. Neuropsychologia 102:95–108. https://doi.org/10.1016/j.neuropsychologia.2017.05.027
Tillman TW, Carhart R (1966) An expanded test for speech discrimination utilizing CNC monosyllabic words Northwestern University Auditory Test No. 6. SAM-TR-66-55. Tech Rep SAM TR. https://doi.org/10.21236/ad0639638
Tremel JJ, Ortiz DM, Fiez JA (2018) Manipulating memory efficacy affects the behavioral and neural profiles of deterministic learning and decision-making. Neuropsychologia 114:214–230. https://doi.org/10.1016/j.neuropsychologia.2018.04.023
Trenerry MR, Crosson B, DeBoe J, Leber WR (1989) Stroop neuropsychological screening test. Psychological Assessment Resources, Odessa
Vaden KI Jr, Kuchinsky SE, Cute SL, Ahlstrom JB, Dubno JR, Eckert MA (2013) The cingulo-opercular network provides word-recognition benefit. J Neurosci 33(48):18979–18986. https://doi.org/10.1523/JNEUROSCI.1417-13.2013
Vaden KI Jr, Kuchinsky SE, Ahlstrom JB, Dubno JR, Eckert MA (2015) Cortical activity predicts which older adults recognize speech in noise and when. J Neurosci 35(9):3929–3937. https://doi.org/10.1523/JNEUROSCI.2908-14.2015
Vaden KI Jr, Kuchinsky SE, Ahlstrom JB, Teubner-Rhodes SE, Dubno JR, Eckert MA (2016) Cingulo-opercular function during word recognition in noise for older adults with hearing loss. Exp Aging Res 42(1):67–82. https://doi.org/10.1080/0361073X.2016.1108784
Vaden KI Jr, Teubner-Rhodes S, Ahlstrom JB, Dubno JR, Eckert MA (2017) Cingulo-opercular activity affects incidental memory encoding for speech in noise. Neuroimage 157:381–387. https://doi.org/10.1016/j.neuroimage.2017.06.028
Vaden KI Jr, Eckert MA, Dubno JR, Harris KC (2020) Cingulo-opercular adaptive control for younger and older adults during a challenging gap detection task. J Neurosci Res 98(4):680–691. https://doi.org/10.1002/jnr.24506
Van Maanen L, Brown SD, Eichele T, Wagenmakers EJ, Ho T, Serences J, Forstmann BU (2011) Neural correlates of trial-to-trial fluctuations in response caution. J Neurosci 31:17488–17495. https://doi.org/10.1523/JNEURsOSCI.2924-11.2011
Varghese LA, Ozmeral EJ, Best V, Shinn-Cunningham BG (2012) How visual cues for when to listen aid selective auditory attention. J Assoc Res Otolaryngol 13(3):359–368. https://doi.org/10.1007/s10162-012-0314-7
Wilke M (2012) An alternative approach towards assessing and accounting for individual motion in fMRI timeseries. Neuroimage 59(3):2062–2072. https://doi.org/10.1016/j.neuroimage.2011.10.043
Wong PC, Ettlinger M, Sheppard JP, Gunasekera GM, Dhar S (2010) Neuroanatomical characteristics and speech perception in noise in older adults. Ear Hear 31(4):471–479. https://doi.org/10.1097/AUD.0b013e3181d709c2
Wostmann M, Herrmann B, Maess B, Obleser J (2016) Spatiotemporal dynamics of auditory attention synchronize with speech. Proc Natl Acad Sci USA 113(14):3873–3878. https://doi.org/10.1073/pnas.1523357113
Xue G, Aron AR, Poldrack RA (2008) Common neural substrates for inhibition of spoken and manual responses. Cereb Cortex 18(8):1923–1932. https://doi.org/10.1093/cercor/bhm220
Ystad M, Eichele T, Lundervold AJ, Lundervold A (2010) Subcortical functional connectivity and verbal episodic memory in healthy elderly–a resting state fMRI study. Neuroimage 52(1):379–388. https://doi.org/10.1016/j.neuroimage.2010.03.062
Funding
This work was supported (in part) by the National Institutes of Health/National Institute on Deafness and Other Communication Disorders (grant number P50 DC 000422) and the National Center for Advancing Translational Sciences of the National Institutes of Health under grant number UL1 TR001450. This investigation was conducted in a facility constructed with support from Research Facilities Improvement Program (grant number C06 RR 014516) from the National Institutes of Health/National Center for Research Resources.
Author information
Authors and Affiliations
Contributions
MAE: Conceptualization, methodology, software, formal analysis, investigation, resources, data curation, writing—original draft, writing—review and editing, visualization, supervision, project administration, funding acquisition. ST-R: Conceptualization, methodology, investigation, writing—review and editing. KIV, Jr.: Conceptualization, methodology, formal analysis, investigation, writing—review and editing. JBA: Methodology, writing—review and editing. CMM: Formal analysis, writing—review and editing. JRD: Methodology, writing—review and editing, funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no financial or personal conflicts of interest to report.
Availability of data and material
The de-identified raw data used in this study are available upon request and completion of institutional data use agreements, as required by the Medical University of South Carolina. The de-identified pre-processed data used for data analysis, stimulus materials used in this study, including the E-Prime script used to deliver the stimuli, is available through Mendeley Data: http://dx.doi.org/10.17632/xkfnvnjmsn.1.
Code availability
R Markdown analysis code will be available through Mendeley Data: http://dx.doi.org/10.17632/xkfnvnjmsn.1.
Ethics approval
The Medical University of South Carolina Institutional Review Board approved study, and experiments were conducted in accordance with the Declaration of Helsinki.
Consent to participate
Written informed consent was obtained from each participant.
Consent for publication
The data shared as part of this individual differences study are de-identified.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Eckert, M.A., Teubner-Rhodes, S., Vaden, K.I. et al. Unique patterns of hearing loss and cognition in older adults’ neural responses to cues for speech recognition difficulty. Brain Struct Funct 227, 203–218 (2022). https://doi.org/10.1007/s00429-021-02398-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02398-2