Abstract
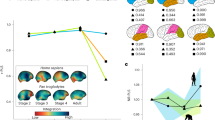
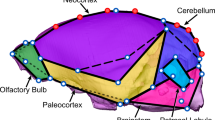
Brain lateralization is a widespread phenomenon although its expression across primates is still controversial due to the reduced number of species analyzed and the disparity of methods used. To gain insight into the diversification of neuroanatomical asymmetries in non-human primates we analyze the endocasts, as a proxy of external brain morphology, of a large sample of New World monkeys and test the effect of brain size, home range and group sizes in the pattern and magnitude of shape asymmetry. Digital endocasts from 26 species were obtained from MicroCT scans and a set of 3D coordinates was digitized on endocast surfaces. Results indicate that Ateles, Brachyteles, Callicebus and Cacajao tend to have a rightward frontal and a leftward occipital lobe asymmetry, whereas Aotus, Callitrichinae and Cebinae have either the opposite pattern or no directional asymmetry. Such differences in the pattern of asymmetry were associated with group and home range sizes. Conversely, its magnitude was significantly associated with brain size, with larger-brained species showing higher inter-hemispheric differences. These findings support the hypothesis that reduction in inter-hemispheric connectivity in larger brains favors the lateralization and increases the structural asymmetries, whereas the patterns of shape asymmetry might be driven by socio-ecological differences among species.





Similar content being viewed by others
Availability of data and material
All data generated and analyzed during this study are included in this published article (Supplementary Tables S1 and S2).
Code availability
Code available upon request.
References
Aristide L, dos Reis SF, Machado AC, Lima I, Lopes RT, Perez SI (2015) Encephalization and diversification of the cranial base in platyrrhine primates. J Hum Evol 81:29–40. https://doi.org/10.1016/j.jhevol.2015.02.003
Aristide L, dos Reis SF, Machado AC, Lima I, Lopes RT, Perez SI (2016) Brain shape convergence in the adaptive radiation of New World monkeys. Proc Natl Acad Sci USA 113(8):2158–2163. https://doi.org/10.1073/pnas.1514473113
Aristide L, Strauss A, Halenar-Price LB, Gilissen E, Cruz FW, Cartelle C, Rosenberger AL, Lopes RT, dos Reis SF, Perez SI (2019) Cranial and endocranial diversity in extant and fossil atelids (Platyrrhini: Atelidae): a geometric morphometric study. Am J Phys Anthropol 169:322–331. https://doi.org/10.1002/ajpa.23837
Atkinson EG, Rogers J, Cheverud JM (2016) Evolutionary and developmental implications of asymmetric brain folding in a large primate pedigree. Evolution 70(3):707–715. https://doi.org/10.1111/evo.12867
Balzeau A, Gilissen E (2010) Endocranial shape asymmetries in Pan paniscus, Pan troglodytes and Gorilla gorilla assessed via skull based landmark analysis. J Hum Evol 59(1):54–69. https://doi.org/10.1016/j.jhevol.2010.03.013
Balzeau A, Grimaud-Hervé D, Gilissen E (2011) Where are inion and endinion? Variations of the exo-and endocranial morphology of the occipital bone during hominin evolution. J Hum Evol 61(4):488–502. https://doi.org/10.1016/j.jhevol.2011.07.002
Bruner E (2014) Human paleoneurology. Springer, Berlin. https://doi.org/10.1007/978-3-319-08500-5
Cameron R, Rogers LJ (1999) Hand preference of the common marmoset (Callithrix jacchus): problem solving and responses in a novel setting. J Comp Psychol 113(2):149. https://doi.org/10.1037/0735-7036.113.2.149
Clutton-Brock TH, Harvey PH (1980) Primates, brains and ecology. J Zool 190(3):309–323. https://doi.org/10.1111/j.1469-7998.1980.tb01430.x
Dryden IL, Mardia KV (1998) Statistical shape analysis. Willey, Chichester
Dumoncel J, Subsol G, Durrleman S, Bertrand A, de Jager E, Oettlé AC, Lockhat Z, Suleman FE, Beaudet A (2021) Are endocasts reliable proxies for brains? A 3D quantitative comparison of the extant human brain and endocast. J Anat 238:480–488. https://doi.org/10.1111/joa.13318
Dunbar RIM (1998) The social brain hypothesis. Evol Anthropol 6(5):178–190. https://doi.org/10.1002/(SICI)1520-6505(1998)6:5%3c178::AID-EVAN5%3e3.0.CO;2-8
Early CM, Iwaniuk AM, Ridgely RC, Lawrence MW (2020) Endocast structures are reliable proxies for the sizes of corresponding regions of the brain in extant birds. J Anat 237:1162–1176. https://doi.org/10.1111/joa.13285
Falk D (1980) Language, handedness, and primate brains: did the australopithecines sign? Am Anthropol 82(1):72–78. https://doi.org/10.1525/aa.1980.82.1.02a00040
Fournier M, Combès B, Roberts N, Braga J, Prima S (2011) Mapping the distance between the brain and the inner surface of the skull and their global asymmetries. Med Imaging Image Process 7962:79620Y. https://doi.org/10.1117/12.876795
Freckleton RP, Harvey PH, Pagel M (2002) Phylogenetic analysis and comparative data: a test and review of evidence. Am Nat 160:712–726. https://doi.org/10.1086/343873
Ghirlanda S, Vallortigara G (2004) The evolution of brain lateralization: a game theoretical analysis of population structure. Proc R Soc Lond B 271:853–857. https://doi.org/10.1098/rspb.2003.2669
Giljov A, Karenina K, Malashichev Y (2018) Facing each other: mammal mothers and infants prefer the position favouring right hemisphere processing. Biol Lett 14(1):20170707. https://doi.org/10.1098/rsbl.2017.0707
Gómez-Robles A, Hopkins WD, Sherwood CC (2013) Increased morphological asymmetry, evolvability and plasticity in human brain evolution. Proc Biol Sci 280:20130575. https://doi.org/10.1098/rspb.2013.0575
Gomez- Robles A, Hopkins WD, Schapiro SJ, Sherwood CC (2016) The heritability of chimpanzee and human brain asymmetry. Proc Biol Sci 283:20161319. https://doi.org/10.1098/rspb.2016.1319
Gorrie CA, Waite PM, Rogers LJ (2008) Correlations between hand preference and cortical thickness in the secondary somatosensory (SII) cortex of the common marmoset, Callithrix Jacchus. Behav Neurosci 122(6):1343. https://doi.org/10.1037/a0013279
Gunz P, Mitteroecker P, Bookstein FL (2005) Semilandmarks in three dimensions. In: Slice DE (ed) Modern morphometrics in physical anthropology. Developments in primatology: progress and prospects. Springer, Boston, pp 73–98
Hartwig W, Rosenberger AL, Norconk MA, Owl MY (2011) Relative brain size, gut size, and evolution in New World monkeys. Anat Rec 294(12):2207–2221. https://doi.org/10.1002/ar.21515
Heilbroner PL, Holloway RL (1988) Anatomical brain asymmetries in New World and Old World monkeys: stages of temporal lobe development in primate evolution. Am J Phys Anthropol 76(1):39–48. https://doi.org/10.1002/ajpa.1330760105
Holloway RL, de la Cost Lareymondie MC (1982) Brain endocast asymmetry in pongids and hominids: some preliminary findings on the paleontology of cerebral dominance. Am J Phys Anthropol 58:101–110. https://doi.org/10.1002/ajpa.1330580111
Hopkins WD, Marino L (2000) Asymmetries in cerebral width in nonhuman primate brains as revealed by magnetic resonance imaging (MRI). Neuropsychologia 38(4):493–499. https://doi.org/10.1016/S0028-3932(99)00090-1
Hopkins WD, Pilcher DL, MacGregor L (2000) Sylvian fissure asymmetries in nonhuman primates revisited: a comparative MRI study. Brain Behav Evol 56(6):293–299. https://doi.org/10.1159/000047213
Hopkins WD, Misiura M, Pope SM, Latash EM (2015) Behavioral and brain asymmetries in primates: a preliminary evaluation of two evolutionary hypotheses. Ann N Y Acad Sci 1359(1):65–83. https://doi.org/10.1111/nyas.12936
Hou L, Xiang L, Crow TJ, Leroy F, Rivière D, Mangin JF, Roberts N (2019) Measurement of Sylvian Fissure asymmetry and occipital bending in humans and Pan troglodytes. Neuroimage 184:855–870. https://doi.org/10.1016/j.neuroimage.2018.08.045
Karolis VR, Corbetta M, De Schotten MT (2019) The architecture of functional lateralisation and its relationship to callosal connectivity in the human brain. Nat Commun 10(1):1–9. https://doi.org/10.1038/s41467-019-09344-1
Klingenberg CP, Barluenga M, Meyer A (2002) Shape analysis of symmetric structures: quantifying variation among individuals and asymmetry. Evolution 56:1909–1920
LeMay M (1976) Morphological cerebral asymmetries of modern man, fossil man, and nonhuman primate. Ann N Y Acad Sci 280:349–366. https://doi.org/10.1111/j.1749-6632.1976.tb25499.x
Liu ST, Phillips KA (2009) Sylvian fissure asymmetry in capuchin monkeys (Cebus apella). Laterality 14(3):217–227. https://doi.org/10.1080/13576500802344404
Mesulam MM (1985) Attention, confusional states, and neglect. In: Mesulam MM (ed) Principles of behavioral neurology. Davis, Philadelphia, pp 125–168
Neubauer S, Gunz P, Hublin JJ (2010) Endocranial shape changes during growth in chimpanzees and humans: a morphometric analysis of unique and shared aspects. J Hum Evol 59:555–566
Neubauer S, Gunz P, Scott NA, Hublin JJ, Mitteroecker P (2020) Evolution of brain lateralization: a shared hominid pattern of endocranial asymmetry is much more variable in humans than in great apes. Sci Adv 6(7):eaax9935. https://doi.org/10.1126/sciadv.aax9935
Ocklenburg S, Güntürkün O (2012) Hemispheric asymmetries: the comparative view. Front Psychol 3:5. https://doi.org/10.3389/fpsyg.2012.00005
Olivares R, Montiel J, Aboitiz F (2001) Species differences and similarities in the fine structure of the mammalian corpus callosum. Brain Behav Evol 57:98–105. https://doi.org/10.1159/000047229
Phillips KA, Sherwood CC (2005) Primary motor cortex asymmetry is correlated with handedness in capuchin monkeys (cebus apella). Behav Neurosci 119(6):1701–1704. https://doi.org/10.1037/0735-7044.119.6.1701
Phillips KA, Sherwood CC (2007) Cerebral petalias and their relationship to handedness in capuchin monkeys (Cebus apella). Neuropsychologia 45(10):2398–2401. https://doi.org/10.1016/j.neuropsychologia.2007.02.021
Phillips KA, Stimpson CD, Smaers JB, Raghanti MA, Jacobs B, Popratiloff A, Hof P, Sherwood CC (2015) The corpus callosum in primates: processing speed of axons and the evolution of hemispheric asymmetry. Proc R Soc B 282(1818):20151535. https://doi.org/10.1098/rspb.2015.1535
Pilcher DL, Hammock EA, Hopkins WD (2001) Cerebral volumetric asymmetries in non-human primates: a magnetic resonance imaging study. Laterality 6(2):165–179. https://doi.org/10.1080/713754406
Powell LE, Isler K, Barton RA (2017) Re-evaluating the link between brain size and behavioural ecology in primates. Proc R Soc B 284(1865):20171765. https://doi.org/10.1098/rspb.2017.1765
Rilling JK, Insel TR (1999) Differential expansion of neural projection systems in primate brain evolution. NeuroReport 10:1453–1459. https://doi.org/10.1097/00001756-199905140-00012
Ringo J, Doty R, Demeter S, Simard P (1994) Timing is of essence: a conjecture that hemispheric specialization arises from inter-hemispheric conduction delay. Cereb Cortex 4:331–343. https://doi.org/10.1093/cercor/4.4.331
Rogers LJ (2014) Asymmetry of brain and behavior in animals: its development, function, and human relevance. Genesis 52(6):555–571. https://doi.org/10.1002/dvg.22741
Rohlf FJ, Slice D (1990) Extensions of the Procrustes method for the optimal superimposition of landmarks. Syst Biol 39(1):40–59. https://doi.org/10.2307/2992207
R Core Team (2020) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/. Accessed 20 Sept 2020
Schlager S (2012) Sliding semi-landmarks on symmetric structures in three dimensions. Am J Phys Anthropol 147:261. https://doi.org/10.1002/ajpa.21502
Schlager S (2017) Morpho and Rvcg-shape analysis in R. In: Zheng G, Li S, Szekely G (eds) Statistical shape and deformation analysis. Academic Press, New York, pp 217–256
Stephan KE, Marshall JC, Friston KJ, Rowe JB, Ritzl A, Zilles K, Fink GR (2003) Lateralized cognitive processes and lateralized task control in the human brain. Science 301(5631):384–386. https://doi.org/10.1126/science.1086025
Vallortigara G (2006) The evolutionary psychology of left and right: costs and benefits of lateralization. Dev Psychobiol 48(6):418–427. https://doi.org/10.1002/dev.20166
Vallortigara G, Rogers LJ (2005) Survival with an asymmetrical brain: advantages and disadvantages of cerebral lateralization. Behav Brain Sci 28:575–633. https://doi.org/10.1017/S0140525X05000105
Watanabe A, Gignac PM, Balanoff AM, Green TL, Kley NJ, Norell MA (2019) Are endocasts good proxies for brain size and shape in archosaurs throughout ontogeny? J Anat 234:291–305. https://doi.org/10.1111/joa.12918
Xiang L, Crow TJ, Hopkins WD, Roberts N (2020) Comparison of surface area and cortical thickness asymmetry in the human and chimpanzee brain. Cereb Cortex. https://doi.org/10.1093/cercor/bhaa202
Acknowledgements
We thank two anonymous reviewers for the comments that helped to improve the clarity of this article.
Funding
This work is supported by Universidad Nacional de La Plata Grant # 911 to S.I.P. and P.N.G. (4113), FAPESP (2017/17357-0) to S.F.d.R.
Author information
Authors and Affiliations
Contributions
PNG: study conception and design, analysis and interpretation of data, drafting of manuscript, critical revision. MVA: study conception and design, analysis and interpretation of data, critical revision. LA: acquisition of data, analysis and interpretation of data, critical revision. RTL: acquisition of data, critical revision. SFR: acquisition of data, critical revision. SIP: study conception and design, acquisition of data, analysis and interpretation of data, drafting of manuscript, critical revision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
429_2021_2371_MOESM1_ESM.pdf
Supplementary Fig. S1. Distribution of specimens along the first two principal components (PC1 and PC2) obtained from the asymmetric shape component (PDF 43 kb)
429_2021_2371_MOESM2_ESM.xlsx
Supplementary Table S1. List of specimens included in this study. The information about the genus, species, specimen ID, endocranial volume and sex is provided (XLSX 11 kb)
429_2021_2371_MOESM3_ESM.csv
Supplementary Table S2. Morphometric and socio-ecological variables used for the comparative phylogenetic analyses (CSV 4 kb)
Rights and permissions
About this article
Cite this article
Gonzalez, P.N., Vallejo-Azar, M., Aristide, L. et al. Endocranial asymmetry in New World monkeys: a comparative phylogenetic analysis of morphometric data. Brain Struct Funct 227, 469–477 (2022). https://doi.org/10.1007/s00429-021-02371-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02371-z