Abstract
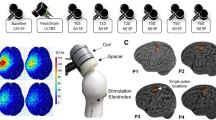
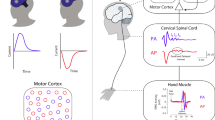
A patterned repetitive transcranial magnetic stimulation protocol, known as continuous theta burst stimulation (cTBS), can suppress corticospinal excitability via mechanisms that appear similar to long-term depression synaptic plasticity. Despite much potential, this technique is currently limited by substantial response variability. The purpose of this study was to investigate whether baseline resting state functional connectivity is a determinant of response to cTBS. Eighteen healthy young adults participated in up to three experimental sessions. Single-pulse transcranial magnetic stimulation was used to quantify change in corticospinal excitability following cTBS. Three minutes of resting electroencephalographic activity was recorded, and functional connectivity was estimated using the debiased weighted phase lag index across different frequency bands. Partial least squares regression identified models of connectivity between a seed region (C3) and the whole scalp that maximally accounted for variance in cTBS responses. There was no group-level effect of a single cTBS train or spaced cTBS trains on corticospinal excitability (p = 0.092). A low beta frequency band model of connectivity accounted for the largest proportion of variance in spaced cTBS response (R2 = 0.50). Based on the low beta frequency model, a-priori regions of interest were identified and predicted 39% of variance in response to spaced cTBS at a subsequent session. Importantly, weaker connectivity between the seed electrode (C3) and a cluster approximating a frontocentral region was associated with greater spaced cTBS response (p = 0.02). It appears M1—frontocentral networks may have an important role in determining the effects of cTBS on corticospinal excitability.







Similar content being viewed by others
References
Andersson CA, Bro R (2000) The N-way toolbox for MATLAB. Chemometr Intell Lab 52(1):1–4. https://doi.org/10.1016/S0169-7439(00)00071-X
Bastos AM, Schoffelen J-M (2016) A tutorial review of functional connectivity analysis methods and their interpretational pitfalls. Front Syst Neurosci. https://doi.org/10.3389/fnsys.2015.00175
Berlim M, Van den Eynde F, Tovar-Perdomo S, Daskalakis Z (2014) Response, remission and drop-out rates following high-frequency repetitive transcranial magnetic stimulation (rTMS) for treating major depression: a systematic review and meta-analysis of randomized, double-blind and sham-controlled trials. Psychol Med 44(2):225–239
Biswal B, Zerrin Yetkin F, Haughton VM, Hyde JS (1995) Functional connectivity in the motor cortex of resting human brain using echo-planar MRI. Magn Reson Med 34(4):537–541
Brown P (2007) Abnormal oscillatory synchronisation in the motor system leads to impaired movement. Curr Opin Neurobiol 17(6):656–664
Cárdenas-Morales L, Volz LJ, Michely J, Rehme AK, Pool EM, Nettekoven C, Eickhoff SB, Fink GR, Grefkes C (2014) Network connectivity and individual responses to brain stimulation in the human motor system. Cereb Cortex 24(7):1697–1707. https://doi.org/10.1093/cercor/bht023
Chaieb L, Antal A, Paulus W (2008) Gender-specific modulation of short-term neuroplasticity in the visual cortex induced by transcranial direct current stimulation. Vis Neurosci 25(1):77–81
Cicchetti DV (1994) Guidelines, criteria, and rules of thumb for evaluating normed and standardized assessment instruments in psychology. Psychol Assess 6(4):284
Civardi C, Cantello R, Asselman P, Rothwell JC (2001) Transcranial magnetic stimulation can be used to test connections to primary motor areas from frontal and medial cortex in humans. Neuroimage 14(6):1444–1453. https://doi.org/10.1006/nimg.2001.0918
Cooke SF, Bliss TVP (2006) Plasticity in the human central nervous system. Brain 129(7):1659–1673. https://doi.org/10.1093/brain/awl082
Cramer RD (1993) Partial least squares (PLS): its strengths and limitations. Perspect Drug Discov 1(2):269–278. https://doi.org/10.1007/BF02174528
Delorme A, Makeig S (2004) EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods 134(1):9–21. https://doi.org/10.1016/j.jneumeth.2003.10.009
Di Lazzaro V, Profice P, Ranieri F, Capone F, Dileone M, Oliviero A, Pilato F (2012) I-wave origin and modulation. Brain Stimul 5(4):512–525. https://doi.org/10.1016/j.brs.2011.07.008
Freitas C, Perez J, Knobel M, Tormos JM, Oberman L, Eldaief M, Bashir S, Vernet M, Pena-Gomez C, Pascual-Leone A (2011) Changes in cortical plasticity across the lifespan. Front Aging Neurosci 3:5. https://doi.org/10.3389/fnagi.2011.00005
Gentner R, Wankerl K, Reinsberger C, Zeller D, Classen J (2008) Depression of human corticospinal excitability induced by magnetic theta-burst stimulation: evidence of rapid polarity-reversing metaplasticity. Cereb Cortex 18(9):2046–2053. https://doi.org/10.1093/cercor/bhm239
Goldsworthy MR, Pitcher JB, Ridding MC (2012) The application of spaced theta burst protocols induces long-lasting neuroplastic changes in the human motor cortex. Eur J Neurosci 35(1):125–134. https://doi.org/10.1111/j.1460-9568.2011.07924.x
Goldsworthy MR, Pitcher JB, Ridding MC (2013) Neuroplastic modulation of inhibitory motor cortical networks by spaced theta burst stimulation protocols. Brain Stimul 6(3):340–345. https://doi.org/10.1016/j.brs.2012.06.005
Goldsworthy MR, Muller-Dahlhaus F, Ridding MC, Ziemann U (2014a) Resistant against de-depression: LTD-like plasticity in the human motor cortex induced by spaced cTBS. Cereb Cortex. https://doi.org/10.1093/cercor/bht353
Goldsworthy MR, Müller-Dahlhaus F, Ridding MC, Ziemann U (2014b) Inter-subject variability of LTD-like plasticity in human motor cortex: a matter of preceding motor activation. Brain Stimul 7(6):864–870. https://doi.org/10.1016/j.brs.2014.08.004
Goldsworthy MR, Hordacre B, Ridding MC (2016) Minimum number of trials required for within- and between-session reliability of TMS measures of corticospinal excitability. Neuroscience 320:205–209. https://doi.org/10.1016/j.neuroscience.2016.02.012
Goodwill AM, Lum JAG, Hendy AM, Muthalib M, Johnson L, Albein-Urios N, Teo W-P (2017) Using non-invasive transcranial stimulation to improve motor and cognitive function in Parkinson’s disease: a systematic review and meta-analysis. Sci Rep 7(1):14840. https://doi.org/10.1038/s41598-017-13260-z
Greicius MD, Menon V, Supekar K, Dougherty RF (2008) Resting-state functional connectivity reflects structural connectivity in the default mode network. Cereb Cortex 19(1):72–78. https://doi.org/10.1093/cercor/bhn059
Hamada M, Murase N, Hasan A, Balaratnam M, Rothwell JC (2013) The role of interneuron networks in driving human motor cortical plasticity. Cereb Cortex 23(7):1593–1605. https://doi.org/10.1093/cercor/bhs147
Heidegger T, Hansen-Goos O, Batlaeva O, Annak O, Ziemann U, Lötsch J (2017) A data-driven approach to responder subgroup identification after paired continuous theta burst stimulation. Front Hum Neurosci. https://doi.org/10.3389/fnhum.2017.00382
Hinder MR, Goss EL, Fujiyama H, Canty AJ, Garry MI, Rodger J, Summers JJ (2014) Inter- and intra-individual variability following intermittent theta burst stimulation: Implications for rehabilitation and recovery. Brain Stimul 7(3):365–371. https://doi.org/10.1016/j.brs.2014.01.004
Hordacre B, Ridding MC, Goldsworthy MR (2015) Response variability to non-invasive brain stimulation protocols. Clin Neurophysiol 126(12):2249–2250. https://doi.org/10.1016/j.clinph.2015.04.052
Hordacre B, Goldsworthy MR, Vallence A-M, Darvishi S, Moezzi B, Hamada M, Rothwell JC, Ridding MC (2017a) Variability in neural excitability and plasticity induction in the human cortex: a brain stimulation study. Brain Stimul 10(3):588–595. https://doi.org/10.1016/j.brs.2016.12.001
Hordacre B, Moezzi B, Goldsworthy MR, Rogasch NC, Graetz LJ, Ridding MC (2017b) Resting state functional connectivity measures correlate with the response to anodal transcranial direct current stimulation. Eur J Neurosci 45(6):837–845. https://doi.org/10.1111/ejn.13508
Hordacre B, Moezzi B, Ridding MC (2018a) Neuroplasticity and network connectivity of the motor cortex following stroke: a transcranial direct current stimulation study. Hum Brain Mapp 39(8):3326–3339. https://doi.org/10.1002/hbm.24079
Hordacre B, Moezzi B, Ridding MC (2018b) Towards targeted brain stimulation in stroke: connectivity as a biomarker of response. J Exp Neurosci. https://doi.org/10.1177/1179069518809060
Huang YZ, Edwards MJ, Rounis E, Bhatia KP, Rothwell JC (2005) Theta burst stimulation of the human motor cortex. Neuron 45(2):201–206
Huang Y-Z, Chen R-S, Rothwell JC, Wen H-Y (2007) The after-effect of human theta burst stimulation is NMDA receptor dependent. Clin Neurophysiol 118(5):1028–1032. https://doi.org/10.1016/j.clinph.2007.01.021
Izuma K, Akula S, Murayama K, Wu D-A, Iacoboni M, Adolphs R (2015) A causal role for posterior medial frontal cortex in choice-induced preference change. J Neurosci 35(8):3598–3606
Jacobs J, Kahana MJ (2010) Direct brain recordings fuel advances in cognitive electrophysiology. Trends Cogn Sci 14(4):162–171. https://doi.org/10.1016/j.tics.2010.01.005
López-Alonso V, Cheeran B, Río-Rodríguez D, Fernández-del-Olmo M (2014) Inter-individual variability in response to non-invasive brain stimulation paradigms. Brain Stimul 7(3):372–380. https://doi.org/10.1016/j.brs.2014.02.004
Marquez J, van Vliet P, McElduff P, Lagopoulos J, Parsons M (2015) Transcranial direct current stimulation (tDCS): does it have merit in stroke rehabilitation? A systematic review. Int J Stroke 10(3):306–316. https://doi.org/10.1111/ijs.12169
Menzies L, Achard S, Chamberlain SR, Fineberg N, Chen CH, del Campo N, Sahakian BJ, Robbins TW, Bullmore E (2007) Neurocognitive endophenotypes of obsessive-compulsive disorder. Brain 130(Pt 12):3223–3236. https://doi.org/10.1093/brain/awm205
Nettekoven C, Volz LJ, Leimbach M, Pool E-M, Rehme AK, Eickhoff SB, Fink GR, Grefkes C (2015) Inter-individual variability in cortical excitability and motor network connectivity following multiple blocks of rTMS. Neuroimage 118:209–218. https://doi.org/10.1016/j.neuroimage.2015.06.004
Neuper C, Pfurtscheller G (1996) Post-movement synchronization of beta rhythms in the EEG over the cortical foot area in man. Neurosci Lett 216(1):17–20
Ogiue-Ikeda M, Kawato S, Ueno S (2003) The effect of repetitive transcranial magnetic stimulation on long-term potentiation in rat hippocampus depends on stimulus intensity. Brain Res 993(1–2):222–226
Oostenveld R, Fries P, Maris E, Schoffelen J-M (2011) FieldTrip: open source software for advanced analysis of MEG, EEG, and invasive electrophysiological data. Comput Intell Neurosci. https://doi.org/10.1155/2011/156869
Opie GM, Vosnakis E, Ridding MC, Ziemann U, Semmler JG (2017) Priming theta burst stimulation enhances motor cortex plasticity in young but not old adults. Brain Stimul 10(2):298–304. https://doi.org/10.1016/j.brs.2017.01.003
Pfurtscheller G, Stancak A Jr, Edlinger G (1997) On the existence of different types of central beta rhythms below 30 Hz. Electroencephalogr Clin Neurophysiol 102(4):316–325
Pfurtscheller G, Zalaudek K, Neuper C (1998) Event-related beta synchronization after wrist, finger and thumb movement. Electroencephalogr Clin Neurophysiol 109(2):154–160
Raichle ME, MacLeod AM, Snyder AZ, Powers WJ, Gusnard DA, Shulman GL (2001) A default mode of brain function. Proc Natl Acad Sci 98(2):676–682
Ridding MC, Ziemann U (2010) Determinants of the induction of cortical plasticity by non-invasive brain stimulation in healthy subjects. J Physiol (lond) 588(13):2291–2304. https://doi.org/10.1113/jphysiol.2010.190314
Roopun AK, Kramer MA, Carracedo LM, Kaiser M, Davies CH, Traub RD, Kopell NJ, Whittington MA (2008) Temporal interactions between cortical rhythms. Front Neurosci 2(2):145–154. https://doi.org/10.3389/neuro.01.034.2008
Rossi S, Hallett M, Rossini PM, Pascual-Leone A (2011) Screening questionnaire before TMS: an update. Clin Neurophysiol 122(8):1686. https://doi.org/10.1016/j.clinph.2010.12.037
Rushworth M, Hadland K, Paus T, Sipila P (2002) Role of the human medial frontal cortex in task switching: a combined fMRI and TMS study. J Neurophysiol 87(5):2577–2592
Sale MV, Ridding MC, Nordstrom MA (2008) Cortisol inhibits neuroplasticity induction in human motor cortex. J Neurosci 28(33):8285–8293
Schölvinck ML, Maier A, Ye FQ, Duyn JH, Leopold DA (2010) Neural basis of global resting-state fMRI activity. Proc Natl Acad Sci 107(22):10238–10243. https://doi.org/10.1073/pnas.0913110107
Stefan K, Kunesch E, Benecke R, Cohen LG, Classen J (2002) Mechanisms of enhancement of human motor cortex excitability induced by interventional paired associative stimulation. J Physiol (lond) 543(2):699–708
Varela F, Lachaux JP, Rodriguez E, Martinerie J (2001) The brainweb: phase synchronization and large-scale integration. Nat Rev Neurosci 2(4):229–239. https://doi.org/10.1038/35067550
Vinck M, Oostenveld R, Van Wingerden M, Battaglia F, Pennartz CMA (2011) An improved index of phase-synchronization for electrophysiological data in the presence of volume-conduction, noise and sample-size bias. Neuroimage 55(4):1548–1565. https://doi.org/10.1016/j.neuroimage.2011.01.055
Volz LJ, Hamada M, Rothwell JC, Grefkes C (2015) What makes the muscle twitch: motor system connectivity and TMS-induced activity. Cereb Cortex 25(9):2346–2353. https://doi.org/10.1093/cercor/bhu032
von Stein A, Sarnthein J (2000) Different frequencies for different scales of cortical integration: from local gamma to long range alpha/theta synchronization. Int J Psychophysiol 38(3):301–313
Ward NS (2006) Compensatory mechanisms in the aging motor system. Ageing Res Rev 5(3):239–254. https://doi.org/10.1016/j.arr.2006.04.003
Westerhuis JA, de Jong S, Smilde AK (2001) Direct orthogonal signal correction. Chemometr Intell Lab 56(1):13–25. https://doi.org/10.1016/S0169-7439(01)00102-2
Wu J, Srinivasan R, Kaur A, Cramer SC (2014) Resting-state cortical connectivity predicts motor skill acquisition. Neuroimage 91:84–90. https://doi.org/10.1016/j.neuroimage.2014.01.026
Wu J, Quinlan EB, Dodakian L, McKenzie A, Kathuria N, Zhou RJ, Augsburger R, See J, Le VH, Srinivasan R, Cramer SC (2015) Connectivity measures are robust biomarkers of cortical function and plasticity after stroke. Brain 138(8):2359–2369
Funding
BH funded by and National Health and Medical Research Council (NHMRC) fellowship (1125054). MRG is supported by an NHMRC-ARC Dementia Research Development fellowship (1102272). This study was supported by an Australian Physiotherapy Association—Physiotherapy Research Foundation Grant (S15-009).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have potential conflicts of interest to be disclosed.
Research involving human participants
This study involved human participants. All study participants provided written informed consent in accordance with the World Medical Association Declaration of Helsinki. Ethical approval to conduct the study was provided by the University of Adelaide Human Research Ethics Committee.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hordacre, B., Goldsworthy, M.R., Graetz, L. et al. Motor network connectivity predicts neuroplastic response following theta burst stimulation in healthy adults. Brain Struct Funct 226, 1893–1907 (2021). https://doi.org/10.1007/s00429-021-02299-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02299-4