Abstract
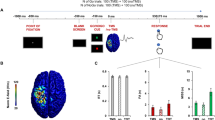
This study investigated the effects of dopaminergic drugs on the EEG mu power during motor imagery, action observation, and execution. This is a double-blind, crossover study with a sample of 15 healthy adults under placebo vs. methylphenidate vs. risperidone conditions during motor imagery, action observation, and execution tasks. The participants had drug dosage adjustment based on body weight/dose (mg/kg). We also analyzed the mu band power by electroencephalography during the study steps. The main result is the interaction between the condition and task factors for the C3 and C4 electrodes, with decreasing EEG mu power in the methylphenidate when compared to risperidone (p ≤ 0.0083). Our results can indicate that the methylphenidate decreases the neurophysiological activity in the central cortical regions during the perceptual experience of tasks with or without body movement.




Similar content being viewed by others
References
Abbruzzese G, Avanzino L, Marchese R (2015) Pelosin E. Innovative Cognitive Tools in the Rehabilitation of Parkinson’s Disease. Parkinsons Dis, Action Observation and Motor Imagery, p 124214
Abou El-Magd RM, Park HK, Kawazoe T, Iwana S, Ono K, Chung SP, Miyano M, Yorita K, Sakai T, Fukui K (2010) The effect of risperidone on D-amino acid oxidase activity as a hypothesis for a novel mechanism of action in the treatment of schizophrenia. J Psy-chopharmacol 24(7):1055–1067
Acton G, Broom C (1989) A dose rising study of the safety and effects on serum prolactin of SK and F 101468, a novel dopamine D2 receptor agonist. Br J Clin Pharmacol 28:435–441
Adhikari BM, Goshorn ES, Lamichhane B, Dhamala M (2013) Temporal-order judgment of audiovisual events involves network activity between parietal and prefrontal cortices. Brain Connect 3(5):536–545
Aprigio D, Tanaka GK, Bittencourt J, Gongora M, Teixeira S, Cagy M, Budde H, Orsini M, Ribeiro P, Velasques B (2020) Dopaminergic drugs alter beta coherence during motor imagery and motor execution in healthy adults. Arq Neuropsiquiatr 78(4):199–205
Balconi M, Cobelli C (2015) rTMS on left prefrontal cortex contributes to memories for positive emotional cues: a comparison between pictures and words. Neuroscience 26(287):93–103
Bastos VH et al (2005) Medidas eletrencefalográficas durante a aprendizagem de tarefa motora sob efeito do bromazepam. Arq Neuropsiquiatr 63.2-B:443–451
Bernier R, Aaronson B, McPartland J (2013) The role of imitation in the observed heterogeneity in EEG mu rhythm in autism and typical development. Brain Cogn 82(1):69–75
Beuter A, Hernández R, Rigal R, Modolo J, Blanchet PJ (2008) Postural sway and effect of levodopa in early Parkinson’s disease. Can J Neurol Sci 35(1):65–68
Braadbaart L, Williams JH, Waiter GD (2013) Do mirror neuron areas mediate mu rhythm suppression during imitation and action observation? Int J Psychophysiol 89(1):99–105
Cannon EN, Yoo KH, Vanderwert RE, Ferrari PF, Woodward AL, Fox NA (2014) Action experience, more than observation, influences mu rhythm desynchronization. PLoS ONE 9(3):e92002
Debarnot U, Clerget E, Olivier E (2011) Role of the primary motor cortex in the early boost in performance following mental imagery training. PLoS ONE 6(10):e26717
Devilbiss DM, Berridge CW (2008) Cognition-enhancing doses of methylphenidate preferentially increase prefrontal cortex neuronal responsiveness. Biol Psychiatry 64(7):626–635
Dickstein R, Deutsch JE (2007) Motor imagery in physical therapist practice. Phys Ther 87(7):942–953
Ehrlichman H, Barrett J (1983) Right hemispheric specialization for mental imagery: a review of the evidence. Brain Cogn 2(1):55–76
Farias TL, Marinho V, Carvalho V, Rocha K, da Silva P, Silva F, Teles AS, Gupta D, Ribeiro P, Velasques B, Cagy M, Bastos VH, Silva-Junior F, Teixeira S (2019) Methylphenidate modifies activity in the prefrontal and parietal cortex accelerating the time judgment. Neurol Sci Off J Ital Neurol Soc Ital Soc Clin Neurophysiol 40(4):829–837
Fayers PM, Machin D (1995) Sample size: how many patients are necessary? Br J Cancer 72:1–9
Fayyazi A, Salari E, Khajeh A, Gajarpour A (2014) A comparison of risperidone and buspirone for treatment of behavior disorders in children with phenylketonuria. Iran J Child Neurol 8(4):33–38 (Fall)
Gualberto CJ (2002) The effects of imagery perspective as a function of skill level on alpha activity. Int J Psychophysiol 43(3):261–271
Hayashi MJ, Kantele M, Walsh V, Carlson S, Kanai R (2014) Dissociable neuroanatomical correlates of subsecond and suprasecond time perception. J Cogn Neurosci 26(8):1685–1693
Hobson HM, Bishop DVM (2016) Mu suppression: a good measure of the human mirror neuron system? Cortex 82:290–310
Hobson HM, Bishop DV (2017) The interpretation of mu suppression as an index of mirror neuron activity: past, present and future. R Soc Open Sci 4(3):160662
Jacobi-Polishook T, Shorer Z, Melzer I (2009) The effect of methylphenidate on postural stability under single and dual task conditions in children with attention deficit hyperactivity disorder: a double blind randomized control trial. J Neurol Sci 280(1–2):15–21
Jeannerod M (2001) Neural simulation of action: a unifying mechanism for motor cognition. Neuroimage 14(1 Pt 2):S103–S109
Kratz O, Diruf MS, Studer P, Gierow W, Buchmann J, Moll GH, Heinrich H (2009) Effects of methylphenidate on motor system excitability in a response inhibition task. Behav Brain Funct 27(5):12
Linssen AM, Vuurman EF, Sambeth A, Riedel WJ (2012) Methylphenidate produces selective enhancement of declarative memory consolidation in healthy volunteers. Psychopharmacology 221(4):611–619
Llanos C, Rodriguez M, Rodriguez-Sabate C, Morales I, Sabate M (2013) Mu-rhythm changes during the planning of motor and motor imagery actions. Neuropsychologia 51(6):1019–1026
Lorey B, Naumann T, Pilgramm S, Petermann C, Bischoff M, Zentgraf K, Stark R, Vaitl D, Munzert J (2013) How equivalent are the action execution, imagery, and observation of intransitive movements? Revisiting the concept of somatotopy during action simulation. Brain Cogn 81(1):139–150
Lotze M, Cohen LG (2006) Volition and imagery in neurorehabilitation. Cogn Behav Neurol 19(3):135–140
Lotze M, Halsband U (2006) Motor imagery. J Physiol Paris 99(4–6):386–395
Malouin F, Richards CL (2010) Mental practice for relearning locomotor skills. Phys Ther 90(2):240–251
Marinho V, Oliveira T, Rocha K, Ribeiro J, Magalhães F, Bento T, Pinto GR, Velasques B, Ribeiro P, Di Giorgio L, Orsini M, Gupta DS, Bittencourt J, Bastos VH, Teixeira S (2018) The dopaminergic system dynamic in the time perception: a review of the evidence. Int J Neurosci 128(3):262–282
Mehta MA, Owen AM, Sahakian BJ, Mavaddat N, Pickard JD, Robbins TW (2000) Methylphenidate enhances working memory by modulating discrete frontal and parietal lobe regions in the human brain. J Neurosci 20(6):RC65
Mizuhara H (2012) Cortical dynamics of human scalp EEG origins in a visually guided motor execution. Neuroimage 62(3):1884–1895
Monte-Silva K, Kuo MF, Thirugnanasambandam N, Liebetanz D, Paulus W, Nitsche MA (2009) Dose-dependent inverted U-shaped effect of dopamine (D2-Like) receptor activation on focal and nonfocal plasticity in humans. J Neurosci 29:6124–6131
Mulder T (2007) Motor imagery and action observation: cognitive tools for rehabilitation. J Neural Transm (Vienna) 114(10):1265–1278
Newcorn JH, Nagy P, Childress AC, Frick G, Yan B, Pliszka S (2017) Randomized, double-blind, placebo-controlled acute comparator trials of lisdexamfetamine and extended-release methylphenidate in adolescents with attention-deficit/hyperactivity disorder. CNS Drugs 31(11):999–1014
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh inventory. Neuropsychologia 9(1):97–113
Paes F, Machado S, Arias-Carrión O, Domingues CA, Teixeira S, Velasques B, Cunha M, Minc D, Basile LF, Budde H, Cagy M, Piedade R, Kerick S, Menéndez-González M, Skaper SD, Norwood BA, Ribeiro P, Nardi AE (2011) Effects of Methylphenidate on performance of a practical pistol shooting task: a quantitative electroencephalography (qEEG) study. Int Arch Med 4(1):6
Reiser M, Büsch D, Munzert J (2011) Strength gains by motor imagery with different ratios of physical to mental practice. Front Psychol 19(2):194
Ritter P, Moosmann M, Villringer A (2009) Rolandic alpha and beta EEG rhythms’ strengths are inversely related to fMRI-BOLD signal in primary somatosensory and motor cortex. Hum Brain Mapp 30(4):1168–1187
Rüther NN, Brown EC, Klepp A, Bellebaum C (2014) Observed manipulation of novel tools leads to mu rhythm suppression over sensory-motor cortices. Behav Brain Res 261:328–335
Serrien DJ, Spapé MM (2009) Effects of task complexity and sensory conflict on goal-directed movement. Neurosci Lett 464(1):10–13
Stecklow MV, Infantosi AF, Cagy M (2010) EEG changes during sequences of visual and kinesthetic motor imagery. Arq Neuropsiquiatr 68(4):556–561
Stray LL, Ellertsen B, Stray T (2010) Motor function and methylphenidate effect in children with attention deficit hyperactivity disorder. Acta Paediatr 99(8):1199–1204
Szameitat AJ, McNamara A, Shen S, Sterr A (2012) Neural activation and functional connectivity during motor imagery of bimanual everyday actions. PLoS ONE 7(6):e38506
Ulloa ER, Pineda JA (2007) Recognition of point-light biological motion: mu rhythms and mirror neuron activity. Behav Brain Res 183(2):188–194
Vanderwert RE, Fox NA, Ferrari PF (2013) The mirror mechanism and mu rhythm in social development. Neurosci Lett 540:15–20
Yarossi M, Manuweera T, Adamovich SV, Tunik E (2017) The effects of mirror feedback during target directed movements on ipsilateral corticospinal excitability. Front Hum Neurosci 11(11):242
Yi W, Qiu S, Qi H, Zhang L, Wan B, Ming D (2013) EEG feature comparison and classification of simple and compound limb motor imagery. J Neuroeng Rehabil 12(10):106
Yi W, Qiu S, Wang K, Qi H, Zhang L, Zhou P, He F, Ming D (2014) Evaluation of EEG oscillatory patterns and cognitive process during simple and compound limb motor imagery. PLoS ONE 9(12):e114853
Yin S, Liu Y, Ding M (2016) Amplitude of sensorimotor mu rhythm is correlated with BOLD from multiple brain regions: a simultaneous EEG-fMRI study. Front Hum Neurosci 10:364
Acknowledgements
We thank Zil-English and Carla Ayres for the manuscript review.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: DA, JB, VM, MG, PR, and BV. Performed the experiments: DA, JB, MG, and BV. Analyzed the data: DA, VM, and BV. Contributed reagents/materials/analysis tools: VHB, VM, ST, MC, HB, PR, LFB, and BV. Wrote the paper: DA, VM, PR, and BV. Headed the analysis: DA, VM, PR, and BV.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval
All procedures performed in this study involving human participants were approved by the institutional and/or national research committee's ethical standards and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Aprigio, D., Bittencourt, J., Gongora, M. et al. Methylphenidate decreases the EEG mu power in the right primary motor cortex in healthy adults during motor imagery and execution. Brain Struct Funct 226, 1185–1193 (2021). https://doi.org/10.1007/s00429-021-02233-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-021-02233-8