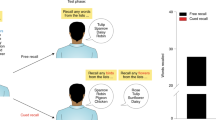
Abstract
Dominant theories of episodic memory propose that a key mechanism of memory consolidation is replay—a process, whereby neural patterns of activation during learning are reinstated during offline post-learning periods. Here, we tested whether key signatures of replay defined by studies in rodents, such as recapitulation of specific memory traces, as well as sequences, are apparent in humans during post-encoding memory reactivation. Thirty participants underwent functional imaging that consisted of interleaved encoding and rest periods. During an offline period of wakeful rest, we biased reactivation towards some memories by presenting sound cues that had previously been associated with particular stimulus sequences. Results showed that targeted hippocampal reactivation was biased towards cued memory sequences and that reactivation signatures preserved the temporal order of particular sequences. Importantly, the biased reactivation was related to differences in subsequent memory, suggesting that preferential reactivation may be a mechanism by which specific memory traces can be strengthened for enhanced subsequent memory retrieval.






Similar content being viewed by others
References
Bendor D, Wilson MA (2012) Biasing the content of hippocampal replay during sleep. Nat Neurosci 15:1439–1444. https://doi.org/10.1038/nn.3203
Carr MF, Jadhav SP, Frank LM (2011) Hippocampal replay in the awake state: a potential substrate for memory consolidation and retrieval. Nat Neurosci 14:147–153. https://doi.org/10.1038/nn.2732
Davidson TJ, Kloosterman F, Wilson MA (2009) Hippocampal replay of extended experience. Neuron 63:497–507. https://doi.org/10.1016/j.neuron.2009.07.027
Deuker L, Olligs J, Fell J et al (2013) Memory consolidation by replay of stimulus-specific neural activity. J Neurosci 33:19373–19383. https://doi.org/10.1523/JNEUROSCI.0414-13.2013
Diba K, Buzsáki G (2007) Forward and reverse hippocampal place-cell sequences during ripples. Nat Neurosci 10:1241–1242. https://doi.org/10.1038/nn1961
Diekelmann S, Büchel C, Born J, Rasch B (2011) Labile or stable: opposing consequences for memory when reactivated during waking and sleep. Nat Neurosci 14:381–388. https://doi.org/10.1038/nn.2744
Foster DJ, Wilson MA (2006) Reverse replay of behavioural sequences in hippocampal place cells during the awake state. Nature 440:680–683. https://doi.org/10.1038/nature04587
Gruber MJ, Ritchey M, Wang S et al (2016) Post-learning hippocampal dynamics promote preferential retention of rewarding events. Neuron 89:1110–1120. https://doi.org/10.1016/j.neuron.2016.01.017
Jadhav SP, Kemere C, German PW, Frank LM (2012) Awake hippocampal sharp-wave ripples support spatial memory. Science 336:1454–1458. https://doi.org/10.1126/science.1217230
James EL, Bonsall MB, Hoppitt L et al (2015) Computer game play reduces intrusive memories of experimental trauma via reconsolidation-update mechanisms. Psychol Sci 26:1201–1215. https://doi.org/10.1177/0956797615583071
Ji D, Wilson MA (2007) Coordinated memory replay in the visual cortex and hippocampus during sleep. Nat Neurosci 10:100–107. https://doi.org/10.1038/nn1825
Kalm K, Davis MH, Norris D (2013) Individual sequence representations in the medial temporal lobe. J Cogn Neurosci 25:1111–1121. https://doi.org/10.1162/jocn
Karlsson MP, Frank LM (2009) Awake replay of remote experiences in the hippocampus. Nat Neurosci 12:913–918. https://doi.org/10.1038/nn.2344
Kriegeskorte N, Mur M, Bandettini P (2008) Representational similarity analysis—connecting the branches of systems neuroscience. Front Syst Neurosci 2:1–28. https://doi.org/10.3389/neuro.06.004.2008
Kumaran D, Hassabis D, McClelland JL (2016) What learning systems do intelligent agents need? Complementary learning systems theory updated. Trends Cogn Sci 20:512–534. https://doi.org/10.1016/j.tics.2016.05.004
Lee AK, Wilson MA (2002) Memory of sequential experience in the hippocampus during slow wave sleep. Neuron 36:1183–1194. https://doi.org/10.1016/S0896-6273(02)01096-6
Marr D (1971) Simple memory: a theory for archicortex. Philos Trans R Soc London Ser B Biol Sci 262:23–81
McClelland JL, McNaughton BL, O’Reilly RC (1995) Why there are complementary learning systems in the hippocampus and neocortex: insights from the successes and failures of connectionist models of learning and memory. Psychol Rev 102:419–457. https://doi.org/10.1037/0033-295X.102.3.419
Nadel L, Moscovitch M (1997) Memory consolidation and the hippocampal complex. Curr Opin Neurobiol 7:217–227. https://doi.org/10.1016/S0959-4388(97)80010-4
Oudiette D, Paller KA (2013) Upgrading the sleeping brain with targeted memory reactivation. Trends Cogn Sci 17:142–149. https://doi.org/10.1016/j.tics.2013.01.006
Oudiette D, Antony JW, Creery JD, Paller KA (2013) The role of memory reactivation during wakefulness and sleep in determining which memories endure. J Neurosci 33:6672–6678. https://doi.org/10.1523/JNEUROSCI.5497-12.2013
Patenaude B, Smith SM, Kennedy DN, Jenkinson M (2011) A Bayesian model of shape and appearance for subcortical brain segmentation. Neuroimage 56:907–922. https://doi.org/10.1016/j.neuroimage.2011.02.046
Rothschild G, Eban E, Frank LM (2017) A cortical-hippocampal-cortical loop of information processing during memory consolidation. Nat Neurosci 20:251–259. https://doi.org/10.1038/nn.4457
Rudoy JD, Voss JL, Westerberg CE, Paller KA (2009) Strengthening individual memories by reactivating them during sleep. Science 326:1079. https://doi.org/10.1126/science.1179013
Schlichting ML, Preston AR (2014) Memory reactivation during rest supports upcoming learning of related content. Proc Natl Acad Sci USA 111:15845–15850. https://doi.org/10.1073/pnas.1404396111
Smith SM, Jenkinson M, Woolrich MW et al (2004) Advances in functional and structural MR image analysis and implementation as FSL. Neuroimage 23(Suppl 1):S208–S219. https://doi.org/10.1016/j.neuroimage.2004.07.051
Squire LR, Alvarez P (1995) Retrograde amnesia and memory consolidation: a neurobiological perspective. Curr Opin Neurobiol 5:169–177
Squire LR, Genzel L, Wixted JT, Morris RG (2015) Memory consolidation. Cold Spring Harb Perspect Biol 7:1–21
Staresina BP, Alink A, Kriegeskorte N, Henson RN (2013) Awake reactivation predicts memory in humans. Proc Natl Acad Sci USA 110:21159–21164. https://doi.org/10.1073/pnas.1311989110
Tambini A, Davachi L (2013) Persistence of hippocampal multivoxel patterns into postencoding rest is related to memory. Proc Natl Acad Sci USA 110:19591–19596. https://doi.org/10.1073/pnas.1308499110
Tambini A, Ketz N, Davachi L (2010) Enhanced brain correlations during rest are related to memory for recent experiences. Neuron 65:280–290. https://doi.org/10.1016/j.neuron.2010.01.001
Teyler TJ, DiScenna P (1985) The role of hippocampus in memory: a hypothesis. Neurosci Biobehav Rev 9:377–389
Tompary A, Duncan K, Davachi L (2015) Consolidation of associative and item memory is related to post-encoding functional connectivity between the ventral tegmental area and different medial temporal lobe subregions during an unrelated task. J Neurosci 35:7326–7331. https://doi.org/10.1523/JNEUROSCI.4816-14.2015
Troiani V, Dougherty CC, Michael AM, Olson IR (2016) Characterization of face-selective patches in orbitofrontal cortex. Front Hum Neurosci 10:1–14. https://doi.org/10.3389/fnhum.2016.00279
Vilberg K, Davachi L (2013) Perirhinal-hippocampal connectivity during reactivation is a marker for object-based memory consolidation. Neuron 79:1232–1242. https://doi.org/10.1016/j.neuron.2013.07.013
Wilson MA, McNaughton BL (1994) Reactivation of hippocampal ensemble memories during sleep. Science 265:676–679
Wimmer GE, Shohamy D (2012) Preference by association: how memory mechanisms in the hippocampus bias decisions. Science 338:270–273
Acknowledgements
We would like to thank Drs. Feroze Mohamed and Chris Conklin for assistance with pulse sequence development, Drs. Charan Ranganath, Anthony Wagner, Jason Chein, and Vinod Venkatraman for helpful advice and commentary on early versions of this manuscript.
Funding
This work was supported by a National Institute of Mental Health Grant to I.R.O. [R01 MH091113] and a Temple University Dissertation Completion Grant to K.H.A. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institute of Mental Health or the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Alm, K.H., Ngo, C.T. & Olson, I.R. Hippocampal signatures of awake targeted memory reactivation. Brain Struct Funct 224, 713–726 (2019). https://doi.org/10.1007/s00429-018-1790-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-018-1790-2