Abstract
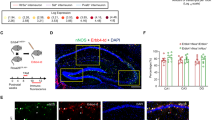
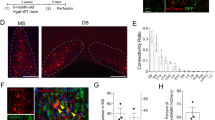
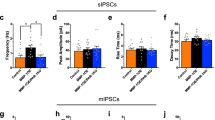
The basal forebrain (BF) is an important regulator of hippocampal and cortical activity. In Alzheimer’s disease (AD), there is a significant loss and dysfunction of cholinergic neurons within the BF, and also a hypertrophy of fibers containing the neuropeptide galanin. Understanding how galanin interacts with BF circuitry is critical in determining what role galanin overexpression plays in the progression of AD. Here, we examined the location and function of galanin in the medial septum/diagonal band (MS/DBB) region of the BF. We show that galanin fibers are located throughout the MS/DBB and intermingled with both cholinergic and GABAergic neurons. Whole-cell patch clamp recordings from MS/DBB neurons in acute slices reveal that galanin decreases tetrodotoxin-sensitive spontaneous GABA release and dampens muscarinic receptor-mediated increases in GABA release in the MS/DBB. These effects are not blocked by pre-exposure to β-amyloid peptide (Aβ1–42). Optogenetic activation of cholinergic neurons in the MS/DBB increases GABA release back onto cholinergic neurons, forming a functional circuit within the MS/DBB. Galanin disrupts this cholinergic-GABAergic circuit by blocking the cholinergic-induced increase in GABA release. These data suggest that galanin works in the BF to reduce inhibitory input onto cholinergic neurons and to prevent cholinergic-induced increase in inhibitory tone. This disinhibition of cholinergic neurons could serve as a compensatory mechanism to counteract the loss of cholinergic signaling that occurs during the progression of AD.








Similar content being viewed by others
References
Alreja M, Wu M, Liu W, Atkins JB, Leranth C, Shanabrough M (2000) Muscarinic tone sustains impulse flow in the septohippocampal GABA but not cholinergic pathway: implications for learning and memory. J Neurosci Off J Soc Neurosci 20(21):8103–8110
Anselmi L, Stella SL Jr, Brecha NC, Sternini C (2009) Galanin inhibition of voltage-dependent Ca(2 +) influx in rat cultured myenteric neurons is mediated by galanin receptor 1. J Neurosci Res 87(5):1107–1114. doi:10.1002/jnr.21923
Barreda-Gomez G, Lombardero L, Giralt MT, Manuel I, Rodriguez-Puertas R (2015) Effects of galanin subchronic treatment on memory and muscarinic receptors. Neuroscience 293:23–34. doi:10.1016/j.neuroscience.2015.02.039
Bartus RT, Dean RL, Beer B, Lippa AS (1982) The cholinergic hypothesis of geriatric memory dysfunction. Science 217(4558):408–417. doi:10.1126/science.7046051
Beach TG, Kuo YM, Spiegel K, Emmerling MR, Sue LI, Kokjohn K, Roher AE (2000) The cholinergic deficit coincides with Abeta deposition at the earliest histopathologic stages of Alzheimer disease. J Neuropathol Exp Neurol 59(4):308–313
Benzing WC, Kordower JH, Mufson EJ (1993) Galanin immunoreactivity within the primate basal forebrain: evolutionary change between monkeys and apes. J Comp Neurol 336(1):31–39. doi:10.1002/cne.903360103
Bowser R, Kordower JH, Mufson EJ (1997) A confocal microscopic analysis of galaninergic hyperinnervation of cholinergic basal forebrain neurons in Alzheimer’s disease. Brain Pathol 7(2):723–730
Chan-Palay V (1988) Galanin hyperinnervates surviving neurons of the human basal nucleus of Meynert in dementias of Alzheimer’s and Parkinson’s disease: a hypothesis for the role of galanin in accentuating cholinergic dysfunction in dementia. J Comp Neurol 273(4):543–557. doi:10.1002/cne.902730409
Cheng Y, Yu LC (2015) Galanin up-regulates the expression of M1 muscarinic acetylcholine receptor via the ERK signaling pathway in primary cultured prefrontal cortical neurons. Neurosci Lett 590:161–165. doi:10.1016/j.neulet.2015.02.011
Chin JH, Ma L, MacTavish D, Jhamandas JH (2007) Amyloid beta protein modulates glutamate-mediated neurotransmission in the rat basal forebrain: involvement of presynaptic neuronal nicotinic acetylcholine and metabotropic glutamate receptors. J Neurosci Off J Soc Neurosci 27(35):9262–9269. doi:10.1523/JNEUROSCI.1843-07.2007
Colgin LL, Denninger T, Fyhn M, Hafting T, Bonnevie T, Jensen O, Moser MB, Moser EI (2009) Frequency of gamma oscillations routes flow of information in the hippocampus. Nature 462(7271):353–357. doi:10.1038/nature08573
Colom LV (2006) Septal networks: relevance to theta rhythm, epilepsy and Alzheimer’s disease. J Neurochem 96(3):609–623. doi:10.1111/j.1471-4159.2005.03630.x
Cortes R, Ceccatelli S, Schalling M, Hokfelt T (1990) Differential effects of intracerebroventricular colchicine administration on the expression of mRNAs for neuropeptides and neurotransmitter enzymes, with special emphasis on galanin: an in situ hybridization study. Synapse 6(4):369–391. doi:10.1002/syn.890060410
Counts SE, He B, Che S, Ginsberg SD, Mufson EJ (2008) Galanin hyperinnervation upregulates choline acetyltransferase expression in cholinergic basal forebrain neurons in Alzheimer’s disease. Neuro-degenerative diseases 5(3–4):228–231. doi:10.1159/000113710
Counts SE, He B, Che S, Ginsberg SD, Mufson EJ (2009) Galanin fiber hyperinnervation preserves neuroprotective gene expression in cholinergic basal forebrain neurons in Alzheimer’s disease. J Alzheimer’s Dis JAD 18(4):885–896. doi:10.3233/JAD-2009-1196
Crawley JN (2008) Galanin impairs cognitive abilities in rodents: relevance to Alzheimer’s disease. Cell Molecul Life Sci CMLS 65(12):1836–1841. doi:10.1007/s00018-008-8158-3
Dannenberg H, Pabst M, Braganza O, Schoch S, Niediek J, Bayraktar M, Mormann F, Beck H (2015) Synergy of direct and indirect cholinergic septo-hippocampal pathways coordinates firing in hippocampal networks. J Neurosci Off J Soc Neurosci 35(22):8394–8410. doi:10.1523/JNEUROSCI.4460-14.2015
Davies P, Maloney AJ (1976) Selective loss of central cholinergic neurons in Alzheimer’s disease. Lancet 2(8000):1403
Ding X, MacTavish D, Kar S, Jhamandas JH (2006) Galanin attenuates beta-amyloid (Abeta) toxicity in rat cholinergic basal forebrain neurons. Neurobiol Dis 21(2):413–420. doi:10.1016/j.nbd.2005.08.016
Duque A, Tepper JM, Detari L, Ascoli GA, Zaborszky L (2007) Morphological characterization of electrophysiologically and immunohistochemically identified basal forebrain cholinergic and neuropeptide Y-containing neurons. Brain Struct Funct 212(1):55–73. doi:10.1007/s00429-007-0143-3
Dutar P, Lamour Y, Nicoll RA (1989) Galanin blocks the slow cholinergic EPSP in CA1 pyramidal neurons from ventral hippocampus. Eur J Pharmacol 164(2):355–360
Dutar P, Bassant MH, Senut MC, Lamour Y (1995) The septohippocampal pathway: structure and function of a central cholinergic system. Physiol Rev 75(2):393–427
Elliott-Hunt CR, Holmes FE, Hartley DM, Perez S, Mufson EJ, Wynick D (2011) Endogenous galanin protects mouse hippocampal neurons against amyloid toxicity in vitro via activation of galanin receptor-2. J Alzheimer’s Dis JAD 25(3):455–462. doi:10.3233/JAD-2011-110011
Elvander E, Ogren SO (2005) Medial septal galanin and acetylcholine: influence on hippocampal acetylcholine and spatial learning. Neuropeptides 39(3):245–248. doi:10.1016/j.npep.2004.12.018
Elvander E, Schott PA, Sandin J, Bjelke B, Kehr J, Yoshitake T, Ogren SO (2004) Intraseptal muscarinic ligands and galanin: influence on hippocampal acetylcholine and cognition. Neuroscience 126(3):541–557. doi:10.1016/j.neuroscience.2004.03.058
Endoh T, Sato D, Wada Y, Shibukawa Y, Ishihara K, Hashimoto S, Yoshinari M, Matsuzaka K, Tazaki M, Inoue T (2008) Galanin inhibits calcium channels via Galpha(i)-protein mediated by GalR1 in rat nucleus tractus solitarius. Brain Res 1229:37–46. doi:10.1016/j.brainres.2008.06.036
Fisone G, Wu CF, Consolo S, Nordstrom O, Brynne N, Bartfai T, Melander T, Hokfelt T (1987) Galanin inhibits acetylcholine release in the ventral hippocampus of the rat: histochemical, autoradiographic, in vivo, and in vitro studies. Proc Natl Acad Sci USA 84(20):7339–7343
Fisone G, Bartfai T, Nilsson S, Hokfelt T (1991) Galanin inhibits the potassium-evoked release of acetylcholine and the muscarinic receptor-mediated stimulation of phosphoinositide turnover in slices of monkey hippocampus. Brain Res 568(1–2):279–284
Griffith WH, Matthews RT (1986) Electrophysiology of AChE-positive neurons in basal forebrain slices. Neurosci Lett 71(2):169–174
Griffith WH, Dubois DW, Fincher A, Peebles KA, Bizon JL, Murchison D (2014) Characterization of age-related changes in synaptic transmission onto F344 rat basal forebrain cholinergic neurons using a reduced synaptic preparation. J Neurophysiol 111(2):273–286. doi:10.1152/jn.00129.2013
Gritti I, Mainville L, Mancia M, Jones BE (1997) GABAergic and other noncholinergic basal forebrain neurons, together with cholinergic neurons, project to the mesocortex and isocortex in the rat. J Comp Neurol 383(2):163–177
Gu Z, Yakel JL (2011) Timing-dependent septal cholinergic induction of dynamic hippocampal synaptic plasticity. Neuron 71(1):155–165. doi:10.1016/j.neuron.2011.04.026
Habert-Ortoli E, Amiranoff B, Loquet I, Laburthe M, Mayaux JF (1994) Molecular cloning of a functional human galanin receptor. Proc Natl Acad Sci USA 91(21):9780–9783
Hasselmo ME (2006) The role of acetylcholine in learning and memory. Curr Opin Neurobiol 16(6):710–715. doi:10.1016/j.conb.2006.09.002
Hasselmo ME, Sarter M (2011) Modes and models of forebrain cholinergic neuromodulation of cognition. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 36(1):52–73. doi:10.1038/npp.2010.104
Hasselmo ME, Bodelon C, Wyble BP (2002) A proposed function for hippocampal theta rhythm: separate phases of encoding and retrieval enhance reversal of prior learning. Neural Comput 14(4):793–817. doi:10.1162/089976602317318965
He B, Counts SE, Perez SE, Hohmann JG, Koprich JB, Lipton JW, Steiner RA, Crawley JN, Mufson EJ (2005) Ectopic galanin expression and normal galanin receptor 2 and galanin receptor 3 mRNA levels in the forebrain of galanin transgenic mice. Neuroscience 133(2):371–380. doi:10.1016/j.neuroscience.2005.01.068
Howard AD, Tan C, Shiao LL, Palyha OC, McKee KK, Weinberg DH, Feighner SD, Cascieri MA, Smith RG, Van Der Ploeg LH, Sullivan KA (1997) Molecular cloning and characterization of a new receptor for galanin. FEBS Lett 405(3):285–290
Huerta PT, Lisman JE (1993) Heightened synaptic plasticity of hippocampal CA1 neurons during a cholinergically induced rhythmic state. Nature 364(6439):723–725. doi:10.1038/364723a0
Janickova H, Rudajev V, Zimcik P, Jakubik J, Tanila H, El-Fakahany EE, Dolezal V (2013) Uncoupling of M1 muscarinic receptor/G-protein interaction by amyloid beta(1–42). Neuropharmacology 67:272–283. doi:10.1016/j.neuropharm.2012.11.014
Jasek MC, Griffith WH (1998) Pharmacological characterization of ionotropic excitatory amino acid receptors in young and aged rat basal forebrain. Neuroscience 82(4):1179–1194
Jelic V, Johansson SE, Almkvist O, Shigeta M, Julin P, Nordberg A, Winblad B, Wahlund LO (2000) Quantitative electroencephalography in mild cognitive impairment: longitudinal changes and possible prediction of Alzheimer’s disease. Neurobiol Aging 21(4):533–540
Jhamandas JH, Harris KH, MacTavish D, Jassar BS (2002) Novel excitatory actions of galanin on rat cholinergic basal forebrain neurons: implications for its role in Alzheimer’s disease. J Neurophysiol 87(2):696–704
Keimpema E, Zheng K, Barde SS, Berghuis P, Dobszay MB, Schnell R, Mulder J, Luiten PG, Xu ZD, Runesson J, Langel U, Lu B, Hokfelt T, Harkany T (2014) GABAergic terminals are a source of galanin to modulate cholinergic neuron development in the neonatal forebrain. Cereb Cortex 24(12):3277–3288. doi:10.1093/cercor/bht192
Kelly JF, Furukawa K, Barger SW, Rengen MR, Mark RJ, Blanc EM, Roth GS, Mattson MP (1996) Amyloid beta-peptide disrupts carbachol-induced muscarinic cholinergic signal transduction in cortical neurons. Proc Natl Acad Sci USA 93(13):6753–6758
Kinney GA, Emmerson PJ, Miller RJ (1998) Galanin receptor-mediated inhibition of glutamate release in the arcuate nucleus of the hypothalamus. The Journal of neuroscience: the official journal of the Society for Neuroscience 18(10):3489–3500
Lang R, Gundlach AL, Holmes FE, Hobson SA, Wynick D, Hokfelt T, Kofler B (2015) Physiology, signaling, and pharmacology of galanin peptides and receptors: three decades of emerging diversity. Pharmacol Rev 67(1):118–175. doi:10.1124/pr.112.006536
Leao RN, Colom LV, Borgius L, Kiehn O, Fisahn A (2012) Medial septal dysfunction by Abeta-induced KCNQ channel-block in glutamatergic neurons. Neurobiol Aging 33(9):2046–2061. doi:10.1016/j.neurobiolaging.2011.07.013
Li L, Yu L, Kong Q (2013) Exogenous galanin attenuates spatial memory impairment and decreases hippocampal beta-amyloid levels in rat model of Alzheimer’s disease. Int J Neurosci 123(11):759–765. doi:10.3109/00207454.2013.800976
Lim YY, Maruff P, Schindler R, Ott BR, Salloway S, Yoo DC, Noto RB, Santos CY, Snyder PJ (2015) Disruption of cholinergic neurotransmission exacerbates Abeta-related cognitive impairment in preclinical Alzheimer’s disease. Neurobiol Aging 36(10):2709–2715. doi:10.1016/j.neurobiolaging.2015.07.009
Lisman J, Buzsaki G (2008) A neural coding scheme formed by the combined function of gamma and theta oscillations. Schizophr Bull 34(5):974–980. doi:10.1093/schbul/sbn060
Manns ID, Mainville L, Jones BE (2001) Evidence for glutamate, in addition to acetylcholine and GABA, neurotransmitter synthesis in basal forebrain neurons projecting to the entorhinal cortex. Neuroscience 107(2):249–263
McKenna JT, Yang C, Franciosi S, Winston S, Abarr KK, Rigby MS, Yanagawa Y, McCarley RW, Brown RE (2013) Distribution and intrinsic membrane properties of basal forebrain GABAergic and parvalbumin neurons in the mouse. J Comp Neurol 521(6):1225–1250. doi:10.1002/cne.23290
McLean CA, Cherny RA, Fraser FW, Fuller SJ, Smith MJ, Beyreuther K, Bush AI, Masters CL (1999) Soluble pool of Abeta amyloid as a determinant of severity of neurodegeneration in Alzheimer’s disease. Ann Neurol 46(6):860–866
Melander T, Staines WA, Hokfelt T, Rokaeus A, Eckenstein F, Salvaterra PM, Wainer BH (1985) Galanin-like immunoreactivity in cholinergic neurons of the septum-basal forebrain complex projecting to the hippocampus of the rat. Brain Res 360(1–2):130–138
Mennicken F, Hoffert C, Pelletier M, Ahmad S, O’Donnell D (2002) Restricted distribution of galanin receptor 3 (GalR3) mRNA in the adult rat central nervous system. J Chem Neuroanat 24(4):257–268
Mesulam MM, Mufson EJ, Levey AI, Wainer BH (1983) Cholinergic innervation of cortex by the basal forebrain: cytochemistry and cortical connections of the septal area, diagonal band nuclei, nucleus basalis (substantia innominata), and hypothalamus in the rhesus monkey. J Comp Neurol 214(2):170–197. doi:10.1002/cne.902140206
Miller MA, Kolb PE, Raskind MA (1997) GALR1 galanin receptor mRNA is co-expressed by galanin neurons but not cholinergic neurons in the rat basal forebrain. Brain Res Mol Brain Res 52(1):121–129
Miller MA, Kolb PE, Planas B, Raskind MA (1998) Few cholinergic neurons in the rat basal forebrain coexpress galanin messenger RNA. J Comp Neurol 391(2):248–258
Mufson EJ, Cochran E, Benzing W, Kordower JH (1993) Galaninergic innervation of the cholinergic vertical limb of the diagonal band (Ch2) and bed nucleus of the stria terminalis in aging. Alzheimer’s disease and Down’s syndrome. Dementia 4(5):237–250
Mufson EJ, Deecher DC, Basile M, Izenwasse S, Mash DC (2000) Galanin receptor plasticity within the nucleus basalis in early and late Alzheimer’s disease: an in vitro autoradiographic analysis. Neuropharmacology 39(8):1404–1412
Mufson EJ, Counts SE, Perez SE, Ginsberg SD (2008) Cholinergic system during the progression of Alzheimer’s disease: therapeutic implications. Expert Rev Neurother 8(11):1703–1718. doi:10.1586/14737175.8.11.1703
Nava-Mesa MO, Jimenez-Diaz L, Yajeya J, Navarro-Lopez JD (2014) GABAergic neurotransmission and new strategies of neuromodulation to compensate synaptic dysfunction in early stages of Alzheimer’s disease. Front Cell Neurosci 8:167. doi:10.3389/fncel.2014.00167
O’Donnell D, Ahmad S, Wahlestedt C, Walker P (1999) Expression of the novel galanin receptor subtype GALR2 in the adult rat CNS: distinct distribution from GALR1. J Comp Neurol 409(3):469–481
Ovsepian SV, Anwyl R, Rowan MJ (2004) Endogenous acetylcholine lowers the threshold for long-term potentiation induction in the CA1 area through muscarinic receptor activation: in vivo study. Euro J Neurosci 20(5):1267–1275. doi:10.1111/j.1460-9568.2004.03582.x
Ovsepian SV, Dolly JO, Zaborszky L (2012) Intrinsic voltage dynamics govern the diversity of spontaneous firing profiles in basal forebrain noncholinergic neurons. J Neurophysiol 108(2):406–418. doi:10.1152/jn.00642.2011
Palazzi E, Felinska S, Zambelli M, Fisone G, Bartfai T, Consolo S (1991) Galanin reduces carbachol stimulation of phosphoinositide turnover in rat ventral hippocampus by lowering Ca2+ influx through voltage-sensitive Ca2+ channels. J Neurochem 56(3):739–747
Papas S, Bourque CW (1997) Galanin inhibits continuous and phasic firing in rat hypothalamic magnocellular neurosecretory cells. J Neurosci Off J Soc Neurosci 17(16):6048–6056
Perez SE, Wynick D, Steiner RA, Mufson EJ (2001) Distribution of galaninergic immunoreactivity in the brain of the mouse. J Comp Neurol 434(2):158–185
Preda S, Govoni S, Lanni C, Racchi M, Mura E, Grilli M, Marchi M (2008) Acute beta-amyloid administration disrupts the cholinergic control of dopamine release in the nucleus accumbens. Neuropsychopharmacol Off Publ Am Coll Neuropsychopharmacol 33(5):1062–1070. doi:10.1038/sj.npp.1301485
Rossi J, Balthasar N, Olson D, Scott M, Berglund E, Lee CE, Choi MJ, Lauzon D, Lowell BB, Elmquist JK (2011) Melanocortin-4 receptors expressed by cholinergic neurons regulate energy balance and glucose homeostasis. Cell Metab 13(2):195–204. doi:10.1016/j.cmet.2011.01.010
Santos-Torres J, Fuente A, Criado JM, Riolobos AS, Heredia M, Yajeya J (2007) Glutamatergic synaptic depression by synthetic amyloid beta-peptide in the medial septum. J Neurosci Res 85(3):634–648. doi:10.1002/jnr.21150
Schliebs R, Arendt T (2011) The cholinergic system in aging and neuronal degeneration. Behav Brain Res 221(2):555–563. doi:10.1016/j.bbr.2010.11.058
Smith KE, Walker MW, Artymyshyn R, Bard J, Borowsky B, Tamm JA, Yao WJ, Vaysse PJ, Branchek TA, Gerald C, Jones KA (1998) Cloned human and rat galanin GALR3 receptors. Pharmacology and activation of G-protein inwardly rectifying K+ channels. J Biol Chem 273(36):23321–23326
Sotty F, Danik M, Manseau F, Laplante F, Quirion R, Williams S (2003) Distinct electrophysiological properties of glutamatergic, cholinergic and GABAergic rat septohippocampal neurons: novel implications for hippocampal rhythmicity. J Physiol 551(Pt 3):927–943. doi:10.1113/jphysiol.2003.046847
Steiner RA, Hohmann JG, Holmes A, Wrenn CC, Cadd G, Jureus A, Clifton DK, Luo M, Gutshall M, Ma SY, Mufson EJ, Crawley JN (2001) Galanin transgenic mice display cognitive and neurochemical deficits characteristic of Alzheimer’s disease. Proc Natl Acad Sci USA 98(7):4184–4189. doi:10.1073/pnas.061445598
Tatemoto K, Rokaeus A, Jornvall H, McDonald TJ, Mutt V (1983) Galanin: a novel biologically active peptide from porcine intestine. FEBS Lett 164(1):124–128
Toneff T, Funkelstein L, Mosier C, Abagyan A, Ziegler M, Hook V (2013) Beta-amyloid peptides undergo regulated co-secretion with neuropeptide and catecholamine neurotransmitters. Peptides 46:126–135. doi:10.1016/j.peptides.2013.04.020
Tsai HC, Zhang F, Adamantidis A, Stuber GD, Bonci A, de Lecea L, Deisseroth K (2009) Phasic firing in dopaminergic neurons is sufficient for behavioral conditioning. Science 324(5930):1080–1084. doi:10.1126/science.1168878
Unal CT, Golowasch JP, Zaborszky L (2012) Adult mouse basal forebrain harbors two distinct cholinergic populations defined by their electrophysiology. Front Behav Neurosci 6:21. doi:10.3389/fnbeh.2012.00021
van Deursen JA, Vuurman EF, Verhey FR, van Kranen-Mastenbroek VH, Riedel WJ (2008) Increased EEG gamma band activity in Alzheimer’s disease and mild cognitive impairment. J Neural Trans 115(9):1301–1311. doi:10.1007/s00702-008-0083-y
Varga V, Hangya B, Kranitz K, Ludanyi A, Zemankovics R, Katona I, Shigemoto R, Freund TF, Borhegyi Z (2008) The presence of pacemaker HCN channels identifies theta rhythmic GABAergic neurons in the medial septum. J Phys 586(Pt 16):3893–3915. doi:10.1113/jphysiol.2008.155242
Wang S, He C, Hashemi T, Bayne M (1997) Cloning and expressional characterization of a novel galanin receptor. Identification of different pharmacophores within galanin for the three galanin receptor subtypes. J Biol Chem 272(51):31949–31952
Wang J, Dickson DW, Trojanowski JQ, Lee VM (1999) The levels of soluble versus insoluble brain Abeta distinguish Alzheimer’s disease from normal and pathologic aging. Exp Neurol 158(2):328–337. doi:10.1006/exnr.1999.7085
Whitehouse PJ, Price DL, Struble RG, Clark AW, Coyle JT, Delon MR (1982) Alzheimer’s disease and senile dementia: loss of neurons in the basal forebrain. Science 215(4537):1237–1239
Witten IB, Lin SC, Brodsky M, Prakash R, Diester I, Anikeeva P, Gradinaru V, Ramakrishnan C, Deisseroth K (2010) Cholinergic interneurons control local circuit activity and cocaine conditioning. Science 330(6011):1677–1681. doi:10.1126/science.1193771
Wu M, Shanabrough M, Leranth C, Alreja M (2000) Cholinergic excitation of septohippocampal GABA but not cholinergic neurons: implications for learning and memory. J Neurosci Off J Soc Neurosci 20(10):3900–3908
Xu M, Chung S, Zhang S, Zhong P, Ma C, Chang WC, Weissbourd B, Sakai N, Luo L, Nishino S, Dan Y (2015) Basal forebrain circuit for sleep-wake control. Nat Neurosci 18(11):1641–1647. doi:10.1038/nn.4143
Yang C, McKenna JT, Zant JC, Winston S, Basheer R, Brown RE (2014) Cholinergic neurons excite cortically projecting basal forebrain GABAergic neurons. J Neurosci Off J Soc Neurosci 34(8):2832–2844. doi:10.1523/JNEUROSCI.3235-13.2014
Yoshitake T, Yoshitake S, Savage S, Elvander-Tottie E, Ogren SO, Kehr J (2011) Galanin differentially regulates acetylcholine release in ventral and dorsal hippocampus: a microdialysis study in awake rat. Neuroscience 197:172–180. doi:10.1016/j.neuroscience.2011.09.035
Zaborszky L, Duque A (2000) Local synaptic connections of basal forebrain neurons. Behav Brain Res 115(2):143–158
Zaborszky L, Pang K, Somogyi J, Nadasdy Z, Kallo I (1999) The basal forebrain corticopetal system revisited. Ann N Y Acad Sci 877:339–367
Acknowledgments
We thank Patricia Lamb for plasmid preparation, Dr. Bernd Gloss for virus packaging, and Charles J. Tucker for assistance with confocal microscopy. We also thank Dr. Guohong Cui for critical reading of this manuscript. This research was supported by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Damborsky, J.C., Smith, K.G., Jensen, P. et al. Local cholinergic-GABAergic circuitry within the basal forebrain is modulated by galanin. Brain Struct Funct 222, 1385–1400 (2017). https://doi.org/10.1007/s00429-016-1283-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-016-1283-0