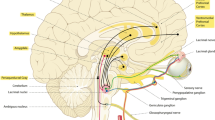
Abstract
The lateral hypothalamic area (LHA) has two major roles: arousal/waking and food intake controls. Here, it is shown that a premammillary part of the LHA is neurochemically and cytoarchitectonically distinct from the tuberal LHA in male rats. This part contains nuclear masses, namely the parasubthalamic nucleus and the calbindin nucleus, involved in pathways that predict its participation in the control of food intake. Analyzing c-Fos expression in experiments related to feeding behavior, this region responded specifically to the ingestion of palatable nutriments.



















Similar content being viewed by others
Abbreviations
- AAA:
-
Anterior amygdalar area
- Ac:
-
Anterior commissure
- ACB:
-
Nucleus accumbens
- Aco:
-
Anterior commissure, olfactory limb
- ADP:
-
Anterodorsal preoptic nucleus
- AHN:
-
Anterior hypothalamic nucleus
- AId:
-
Agranular insular area, dorsal part
- AIp:
-
Agranular insular area, posterior part
- AIv:
-
Agranular insular area, ventral part
- AMB:
-
Nucleus ambiguus
- AMBd:
-
Nucleus ambiguus, dorsal division
- AMY:
-
Amygdala
- AP:
-
Area postrema
- AQ:
-
Cerebral aqueduct proper (sylvius)
- ARH:
-
Arcuate hypothalamic nucleus
- AVP:
-
Anteroventral preoptic nucleus
- B:
-
Barrington’s nucleus
- BLA:
-
Basolateral amygdalar nucleus
- BMA:
-
Basomedial amygdalar nucleus
- BMAa:
-
Basomedial amygdalar nucleus, anterior part
- BMAp:
-
Basomedial amygdalar nucleus, posterior part
- BST:
-
Bed nuclei stria terminalis
- cb:
-
Calbindin
- cbN:
-
Calbindin nucleus
- cc:
-
Corpus callosum
- ccg:
-
Corpus callosum, genu
- CEA:
-
Central amygdalar nucleus
- CEAc:
-
Central amygdalar nucleus, capsular part
- CEAm:
-
Central amygdalar nucleus, medial part
- CGRP:
-
Calcitonin gene-related peptide
- CLA:
-
Claustrum
- CLI:
-
Central linear nucleus raphe
- COAa:
-
Cortical amygdalar nucleus, anterior part
- CP:
-
Caudoputamen
- cpd:
-
Cerebral peduncle
- CS:
-
Superior central nucleus raphe
- CU:
-
Cuneate nucleus
- CUN:
-
Cuneiform nucleus
- dend.:
-
Dendrite
- DMH:
-
Dorsomedial hypothalamic nucleus
- DMX:
-
Dorsal motor nucleus vagus nerve
- DR:
-
Dorsal nucleus raphe
- DTN:
-
Dorsal tegmental nucleus
- EPd:
-
Endopiriform nucleus, dorsal part
- EW:
-
Edinger–Westphal nucleus
- FF:
-
Fields of Forel
- FG:
-
Fluorogold
- fr:
-
Fasciculus retroflexus
- FS:
-
Striatal fundus
- Fx:
-
Columns of the fornix
- GABA:
-
Gamma-aminobutyric acid
- GAD:
-
Glutamic acid decarboxylase
- GP:
-
Globus pallidus
- GR:
-
Gracile nucleus, principal part
- GRN:
-
Gigantocellular reticular nucleus
- GU:
-
Gustatory area
- Hcrt:
-
Hypocretin
- ic:
-
Internal capsule
- IC:
-
Inferior colliculus
- ICc:
-
Inferior colliculus, central nucleus
- IF:
-
Interfascicular nucleus raphe
- IG:
-
Induseum griseum
- III:
-
Oculomotor nucleus
- INC:
-
Interstitial nucleus of cajal
- INS:
-
Insular region
- IO:
-
Inferior olivary complex
- IPN:
-
Interpeduncular nucleus
- KF:
-
Kölliker–Fuse subnucleus (of PB)
- LA:
-
Lateral amygdalar nucleus
- Lat:
-
Lateral part of the LHA
- LC:
-
Locus ceruleus
- LDT:
-
Laterodorsal tegmental nucleus
- LGN:
-
Lateral geniculate nucleus
- LH:
-
Lateral habenula
- LHA:
-
Lateral hypothalamic area
- LHApm:
-
Lateral hypothalamic area, premammillary region
- LHAt:
-
Lateral hypothalamic area, tuberal region
- LM:
-
Lateral mammillary nucleus
- LPO:
-
Lateral preoptic area
- LRNm:
-
Lateral reticular nucleus, magnocellular part
- LSc:
-
Lateral septal nucleus, caudal (caudodorsal) part
- LSr:
-
Lateral septal nucleus, rostral (rostroventral) part
- LSv:
-
Lateral septal nucleus, ventral part
- MA:
-
Magnocellular nucleus
- MARN:
-
Magnocellular reticular nucleus
- MCH:
-
Melanin-concentrating hormone
- MDRN:
-
Medullary reticular nucleus
- ME:
-
Median eminence
- MEA:
-
Medial amygdalar nucleus
- MH:
-
Medial habenula
- MM:
-
Medial mammillary nucleus, body
- MPN:
-
Medial preoptic nucleus
- MPO:
-
Medial preoptic area
- MRNm:
-
Midbrain reticular nucleus, magnocellular part
- MS:
-
Medial septal nucleus
- mtt:
-
Mammillothalamic tract
- MV:
-
Medial vestibular nucleus
- NADPH:
-
Nicotinamide adenine dinucleotide phosphate
- ND:
-
Nucleus of Darkschewitsch
- NDB:
-
Diagonal band nucleus
- NG:
-
Nucleus gemini
- NLOT:
-
Nucleus of the lateral olfactory tract
- NPC:
-
Nucleus of the posterior commissure
- NR:
-
Nucleus of Roller
- NTB:
-
Nucleus of the trapezoid body
- NTS:
-
Nucleus of the solitary tract
- och:
-
Optic chiasm
- opt:
-
Optic tract
- OT:
-
Olfactory tubercle
- PAG:
-
Periaqueductal gray
- PARN:
-
Parvicellular reticular nucleus
- PB:
-
Parabrachial nucleus
- PBl:
-
Parabrachial nucleus, lateral division
- PBm:
-
Parabrachial nucleus, medial division
- PCG:
-
Pontine central gray
- PF:
-
Perifornical region of the LHA
- PG:
-
Pontine gray
- PGRNl:
-
Paragigantocellular reticular nucleus, lateral part
- PH:
-
Posterior hypothalamic nucleus
- PHA-L:
-
Phaseolus vulgaris leucoagglutinin
- PIR:
-
Piriform area
- pm:
-
Principal mammillary tract
- PMd:
-
Dorsal premammillary nucleus
- PMv:
-
Ventral premammillary nucleus
- POR:
-
Periolivary region
- PPN:
-
Pedunculopontine nucleus
- PRC:
-
Precommissural nucleus, periaqueductal gray
- PRNc:
-
Pontine reticular nucleus, caudal part
- PRNr:
-
Pontine reticular nucleus, rostral part
- PS:
-
Parastrial nucleus
- PSTN:
-
Parasubthalamic nucleus
- PSV:
-
Principal sensory nucleus of the trigeminal
- pTK1:
-
Preprotachykinin 1
- PV:
-
Parvalbumin
- PV1:
-
Parvalbumin nucleus
- PVH:
-
Paraventricular hypothalamic nucleus
- PVHdp:
-
Paraventricular hypothalamic nucleus, dorsal parvicellular part
- PVHmpv:
-
Paraventricular hypothalamic nucleus, medial parvicellular part, ventral zone
- PVHpml:
-
Paraventricular hypothalamic nucleus, posterior magnocellular part, lateral zone
- PVp:
-
Periventricular hypothalamic nucleus, posterior part
- PVT:
-
Paraventricular thalamic nucleus
- RE:
-
Nucleus reuniens
- RL:
-
Rostral linear nucleus raphe
- RM:
-
Nucleus raphe magnus
- RN:
-
Red nucleus
- SC:
-
Superior colliculus
- SCH:
-
Suprachiasmatic nucleus
- SCi:
-
Superior colliculus, intermediate layer
- SF:
-
Septofimbrial nucleus
- SH:
-
Septohippocampal nucleus
- SI:
-
Substantia innominata
- SLD:
-
Sublaterodorsal nucleus
- sm:
-
Stria medullaris
- SNc:
-
Substantia nigra, compact part
- SNr:
-
Substantia nigra, reticular part
- SON:
-
Supraoptic nucleus
- SP:
-
Substance P
- SPVC:
-
Spinal nucleus of the trigeminal, caudal part
- SPVI:
-
Spinal nucleus of the trigeminal, interpolar part
- STN:
-
Subthalamic nucleus
- SUM:
-
Supramammillary nucleus
- TH:
-
Tyrosine hydroxylase
- TMd:
-
Tuberomammillary nucleus, dorsal part
- TMv:
-
Tuberomammillary nucleus, ventral part
- TRN:
-
Tegmental reticular nucleus, pontine gray
- TTd2:
-
Tenia tecta, dorsal part, layers 2
- TU:
-
Tuberal nucleus
- TUte:
-
Tuberal nucleus, terete subnucleus
- V3:
-
Third ventricle
- V3m:
-
Third ventricle, mammillary recess
- V4:
-
Fourth ventricle proper
- VIS:
-
Visual area
- vlt:
-
Ventrolateral hypothalamic tract
- Vma:
-
Motor nucleus of the trigeminal, magnocellular part
- VMH:
-
Ventromedial hypothalamic nucleus
- VPMpc:
-
Ventral posteromedial nucleus thalamus, parvicellular part
- VTA:
-
Ventral tegmental area
- VTN:
-
Ventral tegmental nucleus
- XII:
-
Hypoglossal nucleus
- ZI:
-
Zona incerta
- ZIda:
-
Zona incerta, dopaminergic group
References
Anand BK, Brobeck JR (1951) Hypothalamic control of food intake in rats and cats. Yale J Biol Med 24(2):123–140
Aponte Y, Atasoy D, Sternson SM (2011) AGRP neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nat Neurosci 14(3):351–355. doi:10.1038/nn.2739
Bester H, Besson JM, Bernard JF (1997) Organization of efferent projections from the parabrachial area to the hypothalamus: a Phaseolus vulgaris-leucoagglutinin study in the rat. J Comp Neurol 383(3):245–281
Bilella A, Alvarez-Bolado G, Celio MR (2014) Coaxiality of Foxb1- and parvalbumin-expressing neurons in the lateral hypothalamic PV1-nucleus. Neurosci Lett 566:111–114. doi:10.1016/j.neulet.2014.02.028
Bourgeais L, Gauriau C, Bernard JF (2001) Projections from the nociceptive area of the central nucleus of the amygdala to the forebrain: a PHA-L study in the rat. Eur J Neurosci 14(2):229–255
Carter ME, Soden ME, Zweifel LS, Palmiter RD (2013) Genetic identification of a neural circuit that suppresses appetite. Nature 503(7474):111–114. doi:10.1038/nature12596
Carter ME, Han S, Palmiter RD (2015) Parabrachial calcitonin gene-related Peptide neurons mediate conditioned taste aversion. J Neurosci 35(11):4582–4586. doi:10.1523/jneurosci.3729-14.2015
Celio MR (1990) Calbindin D-28 k and parvalbumin in the rat nervous system. Neuroscience 35(2):375–475
Cole S, Hobin MP, Petrovich GD (2015) Appetitive associative learning recruits a distinct network with cortical, striatal, and hypothalamic regions. Neuroscience 286:187–202. doi:10.1016/j.neuroscience.2014.11.026
Comoli E, Ribeiro-Barbosa ER, Negrao N, Goto M, Canteras NS (2005) Functional mapping of the prosencephalic systems involved in organizing predatory behavior in rats. Neuroscience 130(4):1055–1067. doi:10.1016/j.neuroscience.2004.10.020
Cui H, Sohn JW, Gautron L, Funahashi H, Williams KW, Elmquist JK, Lutter M (2012) Neuroanatomy of melanocortin-4 receptor pathway in the lateral hypothalamic area. J Comp Neurol 520(18):4168–4183. doi:10.1002/cne.23145
Cvetkovic V, Poncet F, Fellmann D, Griffond B, Risold PY (2003) Diencephalic neurons producing melanin-concentrating hormone are influenced by local and multiple extra-hypothalamic tachykininergic projections through the neurokinin 3 receptor. Neuroscience 119(4):1113–1145
Cvetkovic V, Brischoux F, Jacquemard C, Fellmann D, Griffond B, Risold PY (2004) Characterization of subpopulations of neurons producing melanin-concentrating hormone in the rat ventral diencephalon. J Neurochem 91(4):911–919. doi:10.1111/j.1471-4159.2004.02776.x
Dobolyi A, Irwin S, Makara G, Usdin TB, Palkovits M (2005) Calcitonin gene-related peptide-containing pathways in the rat forebrain. J Comp Neurol 489(1):92–119. doi:10.1002/cne.20618
Esclapez M, Tillakaratne NJ, Tobin AJ, Houser CR (1993) Comparative localization of mRNAs encoding two forms of glutamic acid decarboxylase with nonradioactive in situ hybridization methods. J Comp Neurol 331(3):339–362. doi:10.1002/cne.903310305
Goto M, Swanson LW (2004) Axonal projections from the parasubthalamic nucleus. J Comp Neurol 469(4):581–607. doi:10.1002/cne.11036
Grove EA (1988) Efferent connections of the substantia innominata in the rat. J Comp Neurol 277(3):347–364. doi:10.1002/cne.902770303
Hahn JD (2010) Comparison of melanin-concentrating hormone and hypocretin/orexin peptide expression patterns in a current parceling scheme of the lateral hypothalamic zone. Neurosci Lett 468(1):12–17. doi:10.1016/j.neulet.2009.10.047
Huang T, Yan J, Kang Y (2003) Role of the central amygdaloid nucleus in shaping the discharge of gustatory neurons in the rat parabrachial nucleus. Brain Res Bull 61(4):443–452
Jennings JH, Rizzi G, Stamatakis AM, Ung RL, Stuber GD (2013) The inhibitory circuit architecture of the lateral hypothalamus orchestrates feeding. Science (New York, NY) 341(6153):1517–1521. doi:10.1126/science.1241812
Krause JE, Chirgwin JM, Carter MS, Xu ZS, Hershey AD (1987) Three rat preprotachykinin mRNAs encode the neuropeptides substance P and neurokinin A. Proc Natl Acad Sci USA 84(3):881–885
Laque A, Zhang Y, Gettys S, Nguyen TA, Bui K, Morrison CD, Munzberg H (2013) Leptin receptor neurons in the mouse hypothalamus are colocalized with the neuropeptide galanin and mediate anorexigenic leptin action. Am J Physiol Endocrinol Metab 304(9):E999–E1011. doi:10.1152/ajpendo.00643.2012
Larsen PJ (1992) Distribution of substance P-immunoreactive elements in the preoptic area and the hypothalamus of the rat. J Comp Neurol 316(3):287–313. doi:10.1002/cne.903160304
Leinninger GM et al (2011) Leptin action via neurotensin neurons controls orexin, the mesolimbic dopamine system and energy balance. Cell Metab 14(3):313–323. doi:10.1016/j.cmet.2011.06.016
Mathur BN, Caprioli RM, Deutch AY (2009) Proteomic analysis illuminates a novel structural definition of the claustrum and insula. Cereb Cortex (New York, NY 1991) 19(10):2372–2379. doi:10.1093/cercor/bhn253
Merchenthaler I, Lopez FJ, Negro-Vilar A (1993) Anatomy and physiology of central galanin-containing pathways. Prog Neurobiol 40(6):711–769
Meszar Z, Girard F, Saper CB, Celio MR (2012) The lateral hypothalamic parvalbumin-immunoreactive (PV1) nucleus in rodents. J Comp Neurol 520(4):798–815. doi:10.1002/cne.22789
Nambu T, Sakurai T, Mizukami K, Hosoya Y, Yanagisawa M, Goto K (1999) Distribution of orexin neurons in the adult rat brain. Brain Res 827(1–2):243–260. doi:10.1016/s0006-8993(99)01336-0
Norgren R (1995) Gustatory system. In: Paxinos G (ed) The rat nervous system. Academic Press, San Diego, pp 751–771
Paxinos G, Watson CH (1986) The rat brain in stereotaxic coordinates, 2nd edn. Academic Press, Sydney
Paxinos G, Watson C (2005) The rat brain in stereotaxic coordinates, vol 5. Elsevier, New York
Petrovich GD, Risold PY, Swanson LW (1996) Organization of projections from the basomedial nucleus of the amygdala: a PHAL study in the rat. J Comp Neurol 374(3):387–420. doi:10.1002/(SICI)1096-9861(19961021)374:3<387:AID-CNE6>3.0.CO;2-Y
Petrovich GD, Setlow B, Holland PC, Gallagher M (2002) Amygdalo-hypothalamic circuit allows learned cues to override satiety and promote eating. J Neurosci 22(19):8748–8753
Price JL, Slotnick BM, Revial MF (1991) Olfactory projections to the hypothalamus. J Comp Neurol 306(3):447–461. doi:10.1002/cne.903060309
Puelles L, Martinez-de-la-Torre M, Bardet S, JLR R (2012) Hypothalamus. In: The mouse nervous system. Elsevier, pp 221–312
Qu D et al (1996) A role for melanin-concentrating hormone in the central regulation of feeding behaviour. Nature 380(6571):243–247. doi:10.1038/380243a0
Risold PY, Swanson LW (1995) Cajal’s nucleus of the stria medullaris: characterization by in situ hybridization and immunohistochemistry for enkephalin. J Chem Neuroanat 9(4):235–240
Risold PY, Swanson LW (1996) Structural evidence for functional domains in the rat hippocampus. Science (New York, NY) 272(5267):1484–1486
Risold PY, Swanson LW (1997) Chemoarchitecture of the rat lateral septal nucleus. Brain Res Rev 24(2–3):91–113
Risold PY, Fellmann D, Rivier J, Vale W, Bugnon C (1992) Immunoreactivities for antisera to three putative neuropeptides of the rat melanin-concentrating hormone precursor are coexpressed in neurons of the rat lateral dorsal hypothalamus. Neurosci Lett 136(2):145–149
Sakurai T et al (1998) Orexins and orexin receptors: a family of hypothalamic neuropeptides and G protein-coupled receptors that regulate feeding behavior. Cell 92(4):573–585. doi:10.1016/s0092-8674(00)80949-6
Saper CB, Chou TC, Elmquist JK (2002) The need to feed: homeostatic and hedonic control of eating. Neuron 36(2):199–211
Shin JW, Geerling JC, Stein MK, Miller RL, Loewy AD (2011) FoxP2 brainstem neurons project to sodium appetite regulatory sites. J Chem Neuroanat 42(1):1–23. doi:10.1016/j.jchemneu.2011.05.003
Shirasu M, Takahashi T, Yamamoto T, Itoh K, Sato S, Nakamura H (2011) Direct projections from the central amygdaloid nucleus to the mesencephalic trigeminal nucleus in rats. Brain Res 1400:19–30. doi:10.1016/j.brainres.2011.05.026
Smith KS, Berridge KC (2005) The ventral pallidum and hedonic reward: neurochemical maps of sucrose “liking” and food intake. J Neurosci 25(38):8637–8649. doi:10.1523/jneurosci.1902-05.2005
Swanson LW (1998) Brain Maps: structure of the rat brain, 2nd edn. Elsevier, Amsterdam
Swanson LW (2004) Brain Maps: Structure of the rat brain, vol 3. Elsevier, New York
Swanson LW, Sanchez-Watts G, Watts AG (2005) Comparison of melanin-concentrating hormone and hypocretin/orexin mRNA expression patterns in a new parceling scheme of the lateral hypothalamic zone. Neurosci Lett 387(2):80–84. doi:10.1016/j.neulet.2005.06.066
Valdes JL, Sanchez C, Riveros ME, Blandina P, Contreras M, Farias P, Torrealba F (2010) The histaminergic tuberomammillary nucleus is critical for motivated arousal. Eur J Neurosci 31(11):2073–2085. doi:10.1111/j.1460-9568.2010.07241.x
Wang PY, Zhang FC (1995) Outline and atlas of learning rat brain slices. Westnorth University Press, China
Yamamoto T, Matsuo R, Kiyomitsu Y, Kitamura R (1989) Response properties of lateral hypothalamic neurons during ingestive behavior with special reference to licking of various taste solutions. Brain Res 481(2):286–297
Yasui Y, Breder CD, Saper CB, Cechetto DF (1991) Autonomic responses and efferent pathways from the insular cortex in the rat. J Comp Neurol 303(3):355–374. doi:10.1002/cne.903030303
Acknowledgments
This work was supported by funding from the French Ministère de la Recherche et de la Technologie (EA 3922) and from the Région de Franche-Comté. The authors thank C. Houdayer, G. Franchi-Bernard, F. Poncet and A. Mariot for technical assistances. We thank G. Sanchez-Watts, D. M. Simmons, R. H. Thompson, G. D. Petrovich, E. A. Markakis, A. B. Kelly and L. W. Swanson for helping to prepare the in situ hybridization material and A. Tobin for the GAD65 and GAD67 probes.
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chometton, S., Pedron, S., Peterschmitt, Y. et al. A premammillary lateral hypothalamic nuclear complex responds to hedonic but not aversive tastes in the male rat. Brain Struct Funct 221, 2183–2208 (2016). https://doi.org/10.1007/s00429-015-1038-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00429-015-1038-3