Abstract
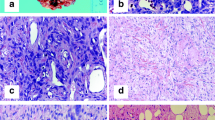
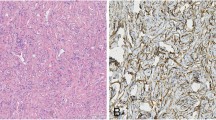
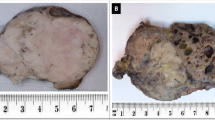
Solitary fibrous tumor is a rare mesenchymal neoplasm that exhibits a broad spectrum of biological behaviors. Few studies relative to clinical-pathologic features and predictive factors have been reported, all involving a mixed population of tumors occurring at different anatomic sites. In this study, we described a cohort of 41 patients with solitary fibrous tumor of the extremities and evaluated the prognostic role of clinical and histological features, presence of C228T and C250T mutations at the TERT promoter region, and NAB2–STAT6 fusion variants. Patients were stratified according to the latest risk stratification model proposed by Demicco. The two patients with metastasis at presentation were in the high-risk group; the one with metastasis after surgery was classified in the intermediate-risk group. TERT promoter mutations were detected in 9 out of 38 DNA available. All patients with metastasis were characterized by a TERT promoter mutation. TERT promoter mutation was associated with mitoses > 4 per high-power field (p = 0.001), necrosis (p = 0.049), and size > 10 cm (p = 0.031). NAB2–STAT6 fusion variants were detected in 27 out of 41 cases without any prognostic value. In conclusion, we confirmed that the patients with solitary fibrous tumor of the limbs have a better prognosis than other solitary fibrous tumors, with a very low percentage of metastatic events. Besides, our data support an association between TERT promoter mutations and histologically malignant features, suggesting a possible molecular role in stratifying patients into intermediate- to high-risk tumor.



Similar content being viewed by others
References
Gengler C, Guillou L (2006) Solitary fibrous tumour and haemangiopericytoma: evolution of a concept. Histopathology 48:63–74
Park MS, Araujo DM (2009) New insights into the hemangiopericytoma/solitary fibrous tumor spectrum of tumors. Curr Opin Oncol 21:327–331
Robinson LA (2006) Solitary fibrous tumor of the pleura. Cancer Control 13:264–269
Boddaert G, Guiraudet P, Grand B, Venissac N, le Pimpec-Barthes F, Mouroux J, Riquet M (2015) Solitary fibrous tumors of the pleura: a poorly defined malignancy profile. Ann Thorac Surg 99:1025–1031
Lahon B, Mercier O, Fadel E, Ghigna MR, Petkova B, Mussot S, Fabre D, le Chevalier T, Dartevelle P (2012) Solitary fibrous tumor of the pleura: outcomes of 157 complete resections in a single center. Ann Thorac Surg 94:394–400
Gholami S, Cassidy MR, Kirane A, Kuk D, Zanchelli B, Antonescu CR, Singer S, Brennan M (2017) Size and location are the most important risk factors for malignant behavior in resected solitary fibrous tumors. Ann Surg Oncol 24:3865–3871
Demicco EG, Wagner MJ, Maki RG, Gupta V, Iofin I, Lazar AJ, Wang WL (2017) Risk assessment in solitary fibrous tumors: validation and refinement of a risk stratification model. Mod Pathol 30:1433–1442
Chmielecki J, Crago AM, Rosenberg M, O’Connor R, Walker SR, Ambrogio L, Auclair D, McKenna A, Heinrich MC, Frank DA, Meyerson M (2013) Whole-exome sequencing identifies a recurrent NAB2-STAT6 fusion in solitary fibrous tumors. Nat Genet 45:131–132
Robinson DR, Wu YM, Kalyana-Sundaram S, Cao X, Lonigro RJ, Sung YS, Chen CL, Zhang L, Wang R, Su F, Iyer MK, Roychowdhury S, Siddiqui J, Pienta KJ, Kunju LP, Talpaz M, Mosquera JM, Singer S, Schuetze SM, Antonescu CR, Chinnaiyan AM (2013) Identification of recurrent NAB2-STAT6 gene fusions in solitary fibrous tumor by integrative sequencing. Nat Genet 45:180–185
Guseva NV, Tanas MR, Stence AA, Sompallae R, Schade JC, Bossler AD, Bellizzi AM, Ma D (2016) The NAB2-STAT6 gene fusion in solitary fibrous tumor can be reliably detected by anchored multiplexed PCR for targeted next-generation sequencing. Cancer Genet 209:303–312
Barthelmess S, Geddert H, Boltze C et al (2014) Solitary fibrous tumors/hemangiopericytomas with different variants of the NAB2-STAT6 gene fusion are characterized by specific histomorphology and distinct clinicopathological features. Am J Pathol 184:1209–1218
Tai HC, Chuang IC, Chen TC, Li CF, Huang SC, Kao YC, Lin PC, Tsai JW, Lan J, Yu SC, Yen SL, Jung SM, Liao KC, Fang FM, Huang HY (2015) NAB2-STAT6 fusion types account for clinicopathological variations in solitary fibrous tumors. Mod Pathol 28:1324–1335
Koelsche C, Renner M, Hartmann W, Brandt R, Lehner B, Waldburger N, Alldinger I, Schmitt T, Egerer G, Penzel R, Wardelmann E, Schirmacher P, von Deimling A, Mechtersheimer G (2014) TERT promoter hotspot mutations are recurrent in myxoid liposarcomas but rare in other soft tissue sarcoma entities. J Exp Clin Cancer Res 33:33
Killela PJ, Reitman ZJ, Jiao Y, Bettegowda C, Agrawal N, Diaz LA, Friedman AH, Friedman H, Gallia GL, Giovanella BC, Grollman AP, He TC, He Y, Hruban RH, Jallo GI, Mandahl N, Meeker AK, Mertens F, Netto GJ, Rasheed BA, Riggins GJ, Rosenquist TA, Schiffman M, Shih IM, Theodorescu D, Torbenson MS, Velculescu VE, Wang TL, Wentzensen N, Wood LD, Zhang M, McLendon RE, Bigner DD, Kinzler KW, Vogelstein B, Papadopoulos N, Yan H (2013) TERT promoter mutations occur frequently in gliomas and a subset of tumors derived from cells with low rates of self-renewal. Proc Natl Acad Sci U S A 110:6021–6026
Lin Y, Seger N, Tsagkozis P, Hesla AC, Ghaderi M, Chen Y, Ehnman M, Warsito D, Wejde J, Larsson O, Haglund F (2018) Telomerase promoter mutations and copy number alterations in solitary fibrous tumours. J Clin Pathol 71:832–839
Vogels R, Macagno N, Griewank K et al (2019) Prognostic significance of NAB2-STAT6 fusion variants and TERT promotor mutations in solitary fibrous tumors/hemangiopericytomas of the CNS: not (yet) clear. Acta Neuropathol 137:679–682
Demicco EG, Park MS, Araujo DM, Fox PS, Bassett RL, Pollock RE, Lazar AJ, Wang WL (2012) Solitary fibrous tumor: a clinicopathological study of 110 cases and proposed risk assessment model. Mod Pathol 25:1298–1306
Righi A, Gambarotti M, Benini S, Gamberi G, Cocchi S, Picci P, Bertoni F (2015) MDM2 and CDK4 expression in periosteal osteosarcoma. Hum Pathol 46:549–553
Lee SE, Chang SH, Kim WY, Lim SD, Kim WS, Hwang TS, Han HS (2016) Frequent somatic TERT promoter mutations and CTNNB1 mutations in hepatocellular carcinoma. Oncotarget 7:69267–69275
Stacchiotti S, Negri T, Palassini E, Conca E, Gronchi A, Morosi C, Messina A, Pastorino U, Pierotti MA, Casali PG, Pilotti S (2010) Sunitinib malate and figitumumab in solitary fibrous tumor: patterns and molecular bases of tumor response. Mol Cancer Ther 9:1286–1297
Fletcher CD (2014) The evolving classification of soft tissue tumours: an update based on the new 2013 WHO classification. Histopathology 64:2–11
O’Neill AC, Tirumani SH, Do WS, Keraliya AR, Hornick JL, Shinagare AB, Ramaiya NH (2017) Metastatic patterns of solitary fibrous tumors: a single-institution experience. Am J Roentgenol 208:2–9
Enzinger FM, Smith BH (1976) Hemangiopericytoma. An analysis of 106 cases. Hum Pathol 7:61–82
Cardillo G, Carbone L, Carleo F, Masala N, Graziano P, Bray A, Martelli M (2009) Solitary fibrous tumors of the pleura: an analysis of 110 patients treated in a single institution. Ann Thorac Surg 88:1632–1637
Musyoki FN, Nahal A, Powell TI (2012) Solitary fibrous tumor: an update on the spectrum of extrapleural manifestations. Skelet Radiol 41:5–13
Demicco EG, Wani K, Ingram D, Wagner M, Maki RG, Rizzo A, Meeker A, Lazar AJ, Wang WL (2018) TERT promoter mutations in solitary fibrous tumour. Histopathol 73:843–851
Bahrami A, Lee S, Schaefer IM, Boland JM, Patton KT, Pounds S, Fletcher CD (2016) TERT promoter mutations and prognosis in solitary fibrous tumor. Mod Pathol 29:1511–1522
Gold JS, Antonescu CR, Hajdu C, Ferrone CR, Hussain M, Lewis JJ, Brennan MF, Coit DG (2002) Clinicopathologic correlates of solitary fibrous tumors. Cancer 94:1057–1068
Insabato L, Siano M, Somma A, Gentile R, Santangelo M, Pettinato G (2009) Extrapleural solitary fibrous tumor: a clinicopathologic study of 19 cases. Int J Surg Pathol 17:250–254
Akaike K, Kurisaki-Arakawa A, Hara K, Suehara Y, Takagi T, Mitani K, Kaneko K, Yao T, Saito T (2015) Distinct clinicopathological features of NAB2-STAT6 fusion gene variants in solitary fibrous tumor with emphasis on the acquisition of highly malignant potential. Hum Pathol 46:347–356
Vogels RJ, Vlenterie M, Versleijen-Jonkers YM et al (2014) Solitary fibrous tumor - clinicopathologic, immunohistochemical and molecular analysis of 28 cases. Diagn Pathol 9:224
Mohajeri A, Tayebwa J, Collin A, Nilsson J, Magnusson L, von Steyern FV, Brosjö O, Domanski HA, Larsson O, Sciot R, Debiec-Rychter M, Hornick JL, Mandahl N, Nord KH, Mertens F (2013) Comprehensive genetic analysis identifies a pathognomonic NAB2/STAT6 fusion gene, nonrandom secondary genomic imbalances, and a characteristic gene expression profile in solitary fibrous tumor. Genes Chromosomes Cancer 52:873–886
Bishop AJ, Zagars GK, Demicco EG, Wang WL, Feig BW, Guadagnolo BA (2018) Soft tissue solitary fibrous tumor: combined surgery and radiation therapy results in excellent local control. Am J Clin Oncol 41(1):81–85
DeVito N, Henderson E, Han G, Reed D, Bui MM, Lavey R, Robinson L, Zager JS, Gonzalez RJ, Sondak VK, Letson GD, Conley A (2015) Clinical characteristics and outcomes for solitary fibrous tumor (SFT): a single center experience. PLoS One 10(10):e0140362
Pasquali S, Gronchi A, Strauss D et al (2016) Resectable extra-pleural and extra-meningeal solitary fibrous tumours: a multi-centre prognostic study. Eur J Surg Oncol 42(7):1064–1070
Wilky BA, Montgomery EA, Guzzetta AA et al (2013) Extrathoracic location and “borderline” histology are associated with recurrence of solitary fibrous tumors after surgical resection. Ann Surg Oncol 20(13):4080–4089
Salas S, Resseguier N, Blay JY, le Cesne A, Italiano A, Chevreau C, Rosset P, Isambert N, Soulie P, Cupissol D, Delcambre C, Bay JO, Dubray-Longeras P, Krengli M, de Bari B, Villa S, Kaanders JHAM, Torrente S, Pasquier D, Thariat JO, Myroslav L, Sole CV, Dincbas HF, Habboush JY, Zilli T, Dragan T, Khanfir K, Ugurluer G, Cena T, Duffaud F, Penel N, Bertucci F, Ranchere-Vince D, Terrier P, Bonvalot S, Macagno N, Lemoine C, Lae M, Coindre JM, Bouvier C (2017) Prediction of local and metastatic recurrence in solitary fibrous tumor: construction of a risk calculator in a multicenter cohort from the French sarcoma group (FSG) database. Ann Oncol 28(8):1979–1987
Funding
This study was supported by 5% citizen income tax contribution to the Rizzoli Orthopaedic Institute and Italian Health Ministry.
Author information
Authors and Affiliations
Contributions
GB and AS analyzed the patient data and gave a major contribution to writing the manuscript. AR performed the histological examination and wrote and edited the manuscript. EP and LP performed genetic analysis. MSB, LS, and DD designed the study and edited and reviewed the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
All procedures were performed in accordance with the ethical standards of the Helsinki declaration. The study was approved by the ethical institutional committee on September 3, 2014 (study code: 0030167/IOR).
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 16 kb)
Rights and permissions
About this article
Cite this article
Bianchi, G., Sambri, A., Pedrini, E. et al. Histological and molecular features of solitary fibrous tumor of the extremities: clinical correlation. Virchows Arch 476, 445–454 (2020). https://doi.org/10.1007/s00428-019-02650-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-019-02650-5