Abstract
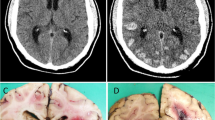
Cerebral amyloid angiopathy (CAA) is the second most common cause of non-traumatic intracerebral haemorrhage (ICH) accounting for 12–15% of lobar haemorrhages in the elderly. Definitive diagnosis of CAA requires histological evaluation. We aimed to evaluate the spectrum of cerebrovascular changes in CAA-related ICH. Between 2011 and 2015, biopsy-confirmed cases of CAA were retrieved and clinical, radiological and pathological features were reviewed. The spectrum of vascular alterations was evaluated and amyloid deposition was graded in accordance with the Greenberg and Vonsattel scale. Seven cases of sporadic CAA [5 males and 2 females] were diagnosed, none of whom were suspected to have CAA pre-operatively. Six presented with large intracerebral haematoma (ICH) requiring neurosurgical intervention (age range: 56–70 years) and one had episodic headache and multiple microhaemorrhages requiring a diagnostic brain biopsy (45 years). In the presence of large ICH, vascular amyloid deposits were of moderate to severe grade (grade 4 in 4, grades 2 and 3 in 1 case each) with predominant involvement of medium (200–500 μm) to large (> 500 μm) leptomeningeal vessels. Fibrinoid necrosis was noted in four. Two were hypertensive and on antiplatelet agents. β-Amyloid plaques were detected in two, one of whom had symptomatic dementia. MRI performed in 3 of 6 cases with ICH did not reveal any microhaemorrhages. Amyloid deposits in small (50–200 μm) to medium (200–500 μm) calibre intracortical vessels produced parenchymal microhemorrhages. Histopathological examination of ICH is essential for diagnosing CAA. The vascular calibre rather than grade of amyloid deposits dictates size of the bleed. Presence of co-morbidities such as antiplatelet agents may predispose to haemorrhage.




Similar content being viewed by others
References
Pezzini A, Padovani A (2008) Cerebral amyloid angiopathy-related hemorrhages. Neurol Sci 29:260–263. https://doi.org/10.1007/s10072-008-0957-7
Knudsen KA, Rosand J, Karluk D, Greenberg SM (2001) Clinical diagnosis of cerebral amyloid angiopathy: validation of the Boston criteria. Neurology 56:537–539
Greenberg SM, Vonsattel JP (1997) Diagnosis of cerebral amyloid angiopathy. Sensitivity and specificity of cortical biopsy. Stroke 28:1418–1422
Vonsattel JP, Myers RH, Hedley-Whyte ET, Ropper AH, Bird ED, Richardson EPJ Jr (1991) Cerebral amyloid angiopathy without and with cerebral hemorrhages: a comparative histological study. Ann Neurol 30:637–649
Oppenheim G (1909) Über “drusige Nekrosen” in der Grosshirnrinde. Neurol Centralbl 28:410–413
Pantelakis S (1954) A particular type of senile angiopathy of the central nervous system: congophilic angiopathy cerebral amyloid angiopathy without and with cerebral hemorrhages: a comparative histological study, topography and frequency. Monatsschr Psychiatr Neurol 128:219–256
Okazaki H, Reagan TJ, Campbell RJ (1979) Clinicopathologic studies of primary cerebral amyloid angiopathy. Mayo Clin Proc 54:22–31
Biffi A, Greenberg SM (2011) Cerebral amyloid angiopathy: a systematic review. J Clin Neurol 7:1–9. https://doi.org/10.3988/jcn.2011.7.1.1
Mehndiratta P, Manjila S, Ostergard T, Eisele S, Cohen ML, Sila C, Selman WR (2012) Cerebral amyloid angiopathy–associated intracerebral hemorrhage: pathology and management. Neurosurg Focus 32:E7. https://doi.org/10.3171/2012.1.FOCUS11370
Iadecola C (2003) Cerebrovascular effects of amyloid-beta peptides: mechanisms and implications for Alzheimer’s dementia. Cell Mol Neurobiol 23:681–689
Zipfel GJ, Han H, Ford AL, Lee JM (2009) Cerebral amyloid angiopathy: progressive disruption of the neurovascular unit. Stroke 40:S16–S19. https://doi.org/10.1161/STROKEAHA.108.533174
Jung SS, Zhang W, Van Nostrand WE (2003) Pathogenic A beta induces the expression and activation of matrix metalloproteinase-2 in human cerebrovascular smooth muscle cells. J Neurochem 85:1208–1215
Lee JM, Yin KJ, Hsin I, Chen S, Fryer JD, Holtzman DM, Hsu CY, Xu J (2003) Matrix metalloproteinase-9 and spontaneous hemorrhage in an animal model of cerebral amyloid angiopathy. Ann Neurol 54:379–382
Meretoja A, Strbian D, Putaala J, Curtze S, Haapaniemi E, Mustanoja S, Sairanen T, Satopää J, Silvennoinen H, Niemelä M, Kaste M, Tatlisumak T (2012) SMASH-U: a proposal for etiologic classification of intracerebral hemorrhage. Stroke 43:2592–2597. https://doi.org/10.1161/STROKEAHA.112.661603
Yeh SJ, Tang SC, Tsai LK, Jeng JS (2014) Pathogenetical subtypes of recurrent intracerebral hemorrhage: designations by SMASH-U classification system. Stroke 45:2636–2642. https://doi.org/10.1161/STROKEAHA.114.005598
Hirohata M, Yoshita M, Ishida C, Ikeda SI, Tamaoka A, Kuzuhara S, Shoji M, Ando Y, Tokuda T, Yamada M (2010) Clinical features of non-hypertensive lobar intracerebral hemorrhage related to cerebral amyloid angiopathy. Eur J Neurol 17:823–829. https://doi.org/10.1111/j.1468-1331.2009.02940.x
Mandybur TI (1986) Cerebral amyloid angiopathy: the vascular pathology and complications. J Neuropathol Exp Neurol 45:79–90
Li XQ, Su DF, Chen HS, Fang Q (2015) Clinical neuropathological analysis of 10 cases of cerebral amyloid angiopathy-related cerebral lobar hemorrhage. J Korean Neurosurg Soc 58:30–35. https://doi.org/10.3340/jkns.2015.58.1.30
Panicker JN, Nagaraja D, Chickabasaviah YT (2010) Cerebral amyloid angiopathy: a clinicopathological study of three cases. Ann Indian Acad Neurol 13:216–220. https://doi.org/10.4103/09722327.70879
Gurol ME, Dierksen G, Betensky R, Gidicsin C, Halpin A, Becker A, Carmasin J, Ayres A, Schwab K, Viswanathan A, Salat D, Rosand J, Johnson KA, Greenberg SM (2012) Predicting sites of new hemorrhage with amyloid imaging in cerebral amyloid angiopathy. Neurology 79:320–326. https://doi.org/10.1212/WNL.0b013e31826043a9
Wagle WA, Smith TW, Weiner M (1984) Intracerebral hemorrhage caused by cerebral amyloid angiopathy: radiographic-pathologic correlation. Am J Neuroradiol 5:171–176
Doubal FN, Mac Lullich AM, Ferguson KJ, Dennis MS, Wardlaw JM (2010) Enlarged perivascular spaces on MRI are a feature of cerebral small vessel disease. Stroke 41:450–454. https://doi.org/10.1161/STROKEAHA.109.564914
Charidimou A, Jaunmuktane Z, Baron J, Burnell M, Varlet P, Peeters A, Xuereb J, Jäger R, Brandner S, Werring DJ (2014) White matter perivascular spaces an MRI marker in pathology-proven cerebral amyloid angiopathy? Neurology 82:57–62. https://doi.org/10.1212/01.wnl.0000438225.02729.04
Linn J, Halpin A, Demaerel P, Ruhland J, Giese AD, Dichgans M, van Buchem MA, Bruckmann H, Greenberg SM (2010) Prevalence of superficial siderosis in patients with cerebral amyloid angiopathy. Neurology 74:1346–1350. https://doi.org/10.1212/WNL.0b013e3181dad605
Walker DA, Broderick DF, Kotsenas AL, Rubino FA (2004) Routine use of gradient-echo MRI to screen for cerebral amyloid angiopathy in elderly patients. Am J Roentgenol 182:1547–1550. https://doi.org/10.2214/ajr.182.6.1821547
Atlas SW, Mark AS, Grossman RI, Gomori JM (1988) Intracranial hemorrhage: gradient-echo MR imaging at 1.5 T-comparison with spin-echo imaging and clinical applications. Radiology 168:803–807
Chao CP, Kotsenas AL, Broderick DF (2006) Cerebral amyloid angiopathy: CT and MR imaging findings. RadioGraphics 26:1517–1531. https://doi.org/10.1148/rg.265055090
Dierksen GA, Skehan ME, Khan MA, Jeng J, Nandigam RN, Becker JA, Kumar A, Neal KL, Betensky RA, Frosch MP, Rosand J, Johnson KA, Viswanathan A, Salat DH, Greenberg SM (2010) Spatial relation between microbleeds and amyloid deposits in amyloid angiopathy. Ann Neurol 68:545–548. https://doi.org/10.1002/ana.22099
van Etten ES, Auriel E, Haley KE, Ayres AM, Vashkevich A, Schwab KM, Rosand J, Viswanathan A, Greenberg SM, Gurol ME (2014) Incidence of symptomatic hemorrhage in patients with lobar microbleeds. Stroke 45:2280–2285. https://doi.org/10.1161/STROKEAHA.114.005151
Greenberg SM, Eng JA, Ning M, Smith EE, Rosand J (2004) Hemorrhage burden predicts recurrent intracerebral hemorrhage after lobar hemorrhage. Stroke 35:1415–1420. https://doi.org/10.1161/01.STR.0000126807.69758.0e
Charidimou A, Kakar P, Fox Z, Werring DJ (2013) Cerebral microbleeds and the risk of intracerebral haemorrhage after thrombolysis for acute ischaemic stroke: systematic review and meta-analysis. J Neurol Neurosurg Psychiatry 84:277–280. https://doi.org/10.1136/jnnp-2012-303379
Charidimou A, Shakeshaft C, Werring DJ (2012) Cerebral microbleeds on magnetic resonance imaging and anticoagulant-associated intracerebral hemorrhage risk. Front Neurol 3:133. https://doi.org/10.3389/fneur.2012.00133
Vernooij MW, Haag MD, van der Lugt A, Hofman A, Krestin GP, Stricker BH, Breteler MM (2009) Use of antithrombotic drugs and the presence of cerebral microbleeds: the Rotterdam scan study. Arch Neurol 66:714–720. https://doi.org/10.1001/archneurol.2009.42
Vinters HV (1987) Cerebral amyloid angiopathy. A critical review. Stroke 18:311–324
Gregoire SM, Charidimou A, Gadapa N, Dolan E, Antoun N, Peeters A, Vandermeeren Y, Laloux P, Baron JC, Jäger HR, Werring DJ (2011) Acute ischaemic brain lesions in intracerebral haemorrhage: multicentre cross-sectional magnetic resonance imaging study. Brain 134:2376–2386. https://doi.org/10.1093/brain/awr172
Arima H, Tzourio C, Anderson C, Woodward M, Bousser MG, MacMahon S, Neal B, Chalmers J (2010) Effects of perindopril-based lowering of blood pressure on intracerebral hemorrhage related to amyloid angiopathy: the PROGRESS trial. Stroke 41:394–396. https://doi.org/10.1161/STROKEAHA.109.563932
Passero S, Burgalassi L, D’Andrea P, Battistini N (1995) Recurrence of bleeding in patients with primary intracerebral hemorrhage. Stroke 26:1189–1192
Tang YJ, Wang S, Zhu MW, Sun YL, Zhao JZ (2013) Severe pathological manifestation of cerebral amyloid angiopathy correlates with poor outcome from cerebral amyloid angiopathy related intracranial hemorrhage. Chin Med J 126:603–608
Acknowledgements
We would like to acknowledge Dr. Natarajan M, Neurosugeon, K G Hospital, Coimbatore, Tamil Nadu, India for referring a case for histopathological diagnosis.
Author information
Authors and Affiliations
Contributions
Author 1, Rajalakshmi Poyuran, -analysed the cases; performed data collection, analysis and interpretation; drafted the manuscript and reviewed the literature.
Author 2, Anita Mahadevan, conceived and designed the study, performed data interpretation and analysis and critically reviewed and finalized the manuscript.
Authors 3 and 7, Arimappamagan Arivazhagan and K V L Narasinga Rao, are neurosurgeons who operated and managed the patients and performed clinical data acquisition, analysis and review of the manuscript with intellectual inputs.
Authors 4 and 8, Nandeesh BN and Yasha T Chickabasaviah, performed diagnosis of cases, data analysis and interpretation; reviewed the manuscript and provided inputs.
Author 5, Madhu Nagappa, is a neurologist who managed the patients, provided and interpreted clinical data, performed clinicopathological analysis and reviewed the manuscript with intellectual inputs.
Author 6, Jitender Saini, is a neuroradiologist who carried out neuroimaging studies, reviewed imaging findings and analysed and interpreted the neuroimaging data and reviewed the manuscript providing critical inputs.
All the authors gave final approval for publication and agree to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Corresponding author takes full responsibility for the work as a whole, including the study design, access to data and the decision to submit and publish the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Poyuran, R., Mahadevan, A., Arimappamagan, A. et al. Cerebrovascular pathology in cerebral amyloid angiopathy presenting as intracerebral haemorrhage. Virchows Arch 474, 235–245 (2019). https://doi.org/10.1007/s00428-018-2505-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-018-2505-z