Abstract
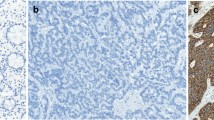
We examined somatostatin receptor type 2A (SSTR2A) expression in primary and metastatic small intestinal neuroendocrine tumors (SI-NETs). We retrieved 156 liver metastases from 26 patients (10 males, 16 females) who had two or more liver lesions resected. A representative formalin-fixed paraffin-embedded section of tumor tissue from each liver metastasis and from the primary tumor, when available, were immunohistochemically stained for SSTR2A. SSTR2A expression was evaluated by the Her2/neu-scoring system and the scoring system proposed by Volante et al. Based on the Her2/neu-scoring system, moderate to strong SSTR2A expression was observed in 121 of 156 (78%) liver metastases. In 15 (58%) subjects, all liver metastases showed moderate to strong SSTR2A expression, whereas in 11 (42%) one or more liver tumors had weak or no expression. Of the 16 stained primaries, 11 (69%) showed heterogeneous SSTR2A expression. The corresponding liver metastases showed only weak to no expression in one, moderate to strong in five, and both weak to no and moderate to strong expression in five of the 11 cases. Using the Volante scoring system, no tumor was scored 0 (0%), two were scored 1 (1%), 38 were scored 2 (24%), and 116 were scored 3 (74%). No statistically significant association was observed between SSTR2A expression and Ki67 index (p = 0.56). Fifteen of 18 (83%) metastatic tumors with a Ki67 index >20% showed moderate to strong SSTR2A. Most liver tumors with weak SSTR2A expression or an IHC score of 2 were detected by OctreoScan. SSTR2A expression in liver metastases of SI-NETs can be variable, even between lesions in the same patient. Expression in metastatic lesions is not always similar to that in the primary tumor. SSTR2A expression is not associated with the Ki67 index.


Similar content being viewed by others
References
Patel YC, Greenwood MT, Panetta R, Demchyshyn L, Niznik H, Srikant CB (1995) The somatostatin receptor family. Life Sci 57:1249–1265
Jais P, Terris B, Ruszniewski P, LeRomancer M, Reyl-Desmars F, Vissuzaine C, Cadiot G, Mignon M, Lewin MJ (1997) Somatostatin receptor subtype gene expression in human endocrine gastroentero-pancreatic tumours. Eur J Clin Investig 27:639–644
Papotti M, Bongiovanni M, Volante M, Allia E, Landolfi S, Helboe L, Schindler M, Cole SL, Bussolati G (2002) Expression of somatostatin receptor types 1-5 in 81 cases of gastrointestinal and pancreatic endocrine tumors. A correlative immunohistochemical and reverse-transcriptase polymerase chain reaction analysis. Virchows Arch 440:461–475
Kubota A, Yamada Y, Kagimoto S, Shimatsu A, Imamura M, Tsuda K, Imura H, Seino S, Seino Y (1994) Identification of somatostatin receptor subtypes and an implication for the efficacy of somatostatin analogue SMS 201-995 in treatment of human endocrine tumors. J Clin Invest 93:1321–1325
Reubi JC, Schaer JC, Waser B, Mengod G (1994) Expression and localization of somatostatin receptor SSTR1, SSTR2, and SSTR3 messenger RNAs in primary human tumors using in situ hybridization. Cancer Res 54:3455–3459
Kimura N, Pilichowska M, Date F, Kimura I, Schindler M (1999) Immunohistochemical expression of somatostatin type 2A receptor in neuroendocrine tumors. Clin Cancer Res 5:3483–3487
Reubi JC, Waser B (2003) Concomitant expression of several peptide receptors in neuroendocrine tumours: molecular basis for in vivo multireceptor tumour targeting. Eur J Nucl Med Mol Imaging 30:781–793
Kagimoto S, Yamada Y, Kubota A, Someya Y, Ihara Y, Yasuda K, Kozasa T, Imura H, Seino S, Seino Y (1994) Human somatostatin receptor, SSTR2, is coupled to adenylyl cyclase in the presence of Gi alpha 1 protein. Biochem Biophys Res Commun 202:1188–1195
Modlin IM, Lye KD, Kidd M (2003) A 5-decade analysis of 13,715 carcinoid tumors. Cancer 97:934–959
Yao JC, Hassan M, Phan A, Dagohoy C, Leary C, Mares JE, Abdalla EK, Fleming JB, Vauthey JN, Rashid A, Evan DB (2008) One hundred years after “carcinoid”: epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol 26:3063–3072
Caplin ME, Pavel M, Ruszniewski P (2014) Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med 371:1556–1557
Wynick D, Anderson JV, Williams SJ, Bloom SR (1989) Resistance of metastatic pancreatic endocrine tumours after long-term treatment with the somatostatin analogue octreotide (SMS 201-995). Clin Endocrinol 30:385–388
Reubi JC, Kvols LK, Waser B, Nagorney DM, Heitz PU, Charboneau JW, Reading CC, Moertel C (1990) Detection of somatostatin receptors in surgical and percutaneous needle biopsy samples of carcinoids and islet cell carcinomas. Cancer Res 50:5969–5977
Kvols LK, Buck M, Moertel CG, Schutt AJ, Rubin J, O'Connell MJ, Hahn RG (1987) Treatment of metastatic islet cell carcinoma with a somatostatin analogue (SMS 201-995). Ann Intern Med 107:162–168
Kvols LK, Moertel CG, O'Connell MJ, Schutt AJ, Rubin J, Hahn RG (1986) Treatment of the malignant carcinoid syndrome. Evaluation of a long-acting somatostatin analogue. N Engl J Med 315:663–666
Gardner-Roehnelt NM (2012) Update on the management of neuroendocrine tumors: focus on somatostatin antitumor effects. Clin J Oncol Nurs 16:56–64
Laznicek M, Laznickova A, Maecke HR (2012) Receptor affinity and preclinical biodistribution of radiolabeled somatostatin analogs. Anticancer Res 32:761–766
Reubi JC, Schar JC, Waser B, Wenger S, Heppeler A, Schmitt JS, Macke HR (2000) Affinity profiles for human somatostatin receptor subtypes SST1-SST5 of somatostatin radiotracers selected for scintigraphic and radiotherapeutic use. Eur J Nucl Med 27:273–282
Janson ET, Gobl A, Kalkner KM, Oberg K (1996) A comparison between the efficacy of somatostatin receptor scintigraphy and that of in situ hybridization for somatostatin receptor subtype 2 messenger RNA to predict therapeutic outcome in carcinoid patients. Cancer Res 56:2561–2565
Bakker WH, Krenning EP, Reubi JC, Breeman WA, Setyono-Han B, de Jong M, Kooij PP, Bruns C, van Hagen PM, Marbach P et al (1991) In vivo application of [111In-DTPA-D-Phe1]-octreotide for detection of somatostatin receptor-positive tumors in rats. Life Sci 49:1593–1601
Krenning EP, Kwekkeboom DJ, Bakker WH, Breeman WA, Kooij PP, Oei HY, van Hagen M, Postema PT, de Jong M, Reubi JC et al (1993) Somatostatin receptor scintigraphy with [111In-DTPA-D-Phe1]- and [123I-Tyr3]-octreotide: the Rotterdam experience with more than 1000 patients. Eur J Nucl Med 20:716–731
Maecke HR, Reubi JC (2011) Somatostatin receptors as targets for nuclear medicine imaging and radionuclide treatment. J Nucl Med 52:841–844
Ambrosini V, Fani M, Fanti S, Forrer F, Maecke HR (2011) Radiopeptide imaging and therapy in Europe. J Nucl Med 52(Suppl 2):42S–55S
Treglia G, Castaldi P, Rindi G, Giordano A, Rufini V (2012) Diagnostic performance of gallium-68 somatostatin receptor PET and PET/CT in patients with thoracic and gastroenteropancreatic neuroendocrine tumours: a meta-analysis. Endocrine 42:80–87
Sharma N, Naraev BG, Engelman EG, Zimmerman MB, Bushnell DL, Jr., O'Dorisio TM, O'Dorisio MS, Menda Y, Muller-Brand J, Howe JR et al. (2016) Peptide receptor radionuclide therapy outcomes in a North American cohort with metastatic well-differentiated neuroendocrine tumors. Pancreas.
Yalchin M, Oliveira A, Theocharidou E, Pencharz D, Navalkissoor S, Quigley AM, Walker M, Caplin M, Toumpanakis C (2016) The impact of radiological response to peptide receptor radionuclide therapy on overall survival in patients with metastatic midgut neuroendocrine tumors. Clin Nucl Med.
Shi C, Gonzalez RS, Zhao Z, Koyama T, Cornish TC, Hande KR, Walker R, Sandler M, Berlin J, Liu EH (2015) Liver metastases of small intestine neuroendocrine tumors: Ki-67 heterogeneity and World Health Organization grade discordance with primary tumors. Am J Clin Pathol 143:398–404
Kaemmerer D, Peter L, Lupp A, Schulz S, Sanger J, Baum RP, Prasad V, Hommann M (2013) Comparing of IRS and Her2 as immunohistochemical scoring schemes in gastroenteropancreatic neuroendocrine tumors. Int J Clin Exp Pathol 5:187–194
Daniel K, Maria A, Amelie L, Isabell L, Stefan S, Luisa P, Merten H, Vikas P, Gerd B, Paul BR (2014) Somatostatin receptor immunohistochemistry in neuroendocrine tumors: comparison between manual and automated evaluation. Int J Clin Exp Pathol 7:4971–4980
Volante M, Brizzi MP, Faggiano A, La Rosa S, Rapa I, Ferrero A, Mansueto G, Righi L, Garancini S, Capella C et al (2007) Somatostatin receptor type 2A immunohistochemistry in neuroendocrine tumors: a proposal of scoring system correlated with somatostatin receptor scintigraphy. Mod Pathol 20:1172–1182
Team R R: A language and environment for statistical computing. (3.3):Vienna, Austria.
Kloppel G (2011) Classification and pathology of gastroenteropancreatic neuroendocrine neoplasms. Endocr Relat Cancer 18(Suppl 1):S1–16
Heidari P, Wehrenberg-Klee E, Habibollahi P, Yokell D, Kulke M, Mahmood U (2013) Free somatostatin receptor fraction predicts the antiproliferative effect of octreotide in a neuroendocrine tumor model: implications for dose optimization. Cancer Res 73:6865–6873
Nicolas G, Giovacchini G, Muller-Brand J, Forrer F (2011) Targeted radiotherapy with radiolabeled somatostatin analogs. Endocrinol Metab Clin N Am 40:187–204 ix-x
Oksuz MO, Winter L, Pfannenberg C, Reischl G, Mussig K, Bares R, Dittmann H (2014) Peptide receptor radionuclide therapy of neuroendocrine tumors with (90)Y-DOTATOC: is treatment response predictable by pre-therapeutic uptake of (68)Ga-DOTATOC? Diagn Interv Imaging 95:289–300
Kwekkeboom DJ, de Herder WW, van Eijck CH, Kam BL, van Essen M, Teunissen JJ, Krenning EP (2010) Peptide receptor radionuclide therapy in patients with gastroenteropancreatic neuroendocrine tumors. Semin Nucl Med 40:78–88
Frilling A, Clift AK (2015) Therapeutic strategies for neuroendocrine liver metastases. Cancer 121:1172–1186
Bodei L, Mueller-Brand J, Baum RP, Pavel ME, Horsch D, O'Dorisio MS, O'Dorisio TM, Howe JR, Cremonesi M, Kwekkeboom DJ et al (2013) The joint IAEA, EANM, and SNMMI practical guidance on peptide receptor radionuclide therapy (PRRNT) in neuroendocrine tumours. Eur J Nucl Med Mol Imaging 40:800–816
Korner M, Waser B, Reubi JC (2015) Does somatostatin or gastric inhibitory peptide receptor expression correlate with tumor grade and stage in gut neuroendocrine tumors? Neuroendocrinology 101:45–57
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was reviewed and approved by the Vanderbilt Institutional Review Board. Consent was not required for this study.
Funding
This work was partially supported by NIH/NIDDK DK058404-11 (ZZ and TK), NIH/NCI P50CA095103 (CS), and NCI CA 096625 (EL).
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Charoenpitakchai, M., Liu, E., Zhao, Z. et al. In liver metastases from small intestinal neuroendocrine tumors, SSTR2A expression is heterogeneous. Virchows Arch 470, 545–552 (2017). https://doi.org/10.1007/s00428-017-2093-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00428-017-2093-3