Abstract
Main conclusion
CsNAC086 was found to promote the expression of CsFLS, thus promoting the accumulation of flavonols in Camellia sinensis.
Abstract
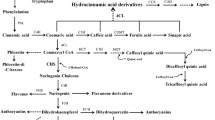
Flavonols, the main flavonoids in tea plants, play an important role in the taste and quality of tea. In this study, a NAC TF gene CsNAC086 was isolated from tea plants and confirmed its regulatory role in the expression of flavonol synthase which is a key gene involved in the biosynthesis of flavonols in tea plant. Yeast transcription-activity assays showed that CsNAC086 has self-activation activity. The transcriptional activator domain of CsNAC086 is located in the non-conserved C-terminal region (positions 171–550), while the conserved NAC domain (positions 1–170) does not have self-activation activity. Silencing the CsNAC086 gene using antisense oligonucleotides significantly decreased the expression of CsFLS. As a result, the concentration of flavonols decreased significantly. In overexpressing CsNAC086 tobacco leaves, the expression of NtFLS was significantly increased. Compared with wild-type tobacco, the flavonols concentration increased. Yeast one-hybrid assays showed CsNAC086 did not directly regulate the gene expression of CsFLS. These findings indicate that CsNAC086 plays a role in regulating flavonols biosynthesis in tea plants, which has important implications for selecting and breeding of high-flavonols-concentration containing tea-plant cultivars.








Similar content being viewed by others
Data availability
The nucleotide sequence reported in this study have been deposited in the NCBI GenBank with accession number: OQ863021.
Abbreviations
- CHS:
-
Chalcone synthase
- F3H:
-
Flavanone-3-hydroxylase
- FLS:
-
Flavonol synthase
- AsODN:
-
Antisense oligonucleotides
- sODNs:
-
Sense oligodeoxynucleotide
- TF:
-
Transcription factor
References
An JP, Qu FJ, Yao JF, Wang XN, You CX, Wang XF, Hao YJ (2017) The bZIP transcription factor MdHY5 regulates anthocyanin accumulation and nitrate assimilation in apple. Hortic Res 4:17023. https://doi.org/10.1038/hortres.2017.23
Balentine DA, Wiseman SA, Bouwens L (1997) The chemistry of tea flavonoids. Food Sci Nutr 37:693–704. https://doi.org/10.1080/10408399709527797
Chen Q, Wang Q, Xiong L, Lou Z (2011) A structural view of the conserved domain of rice stress-responsive NAC1. Protein Cell 2:55–63. https://doi.org/10.1007/s13238-011-1010-9
Dalman K, Wind JJ, Nemesio-Gorriz M, Hammerbacher A, Lundén K, Ezcurra I, Elfstrand M (2017) Overexpression of PaNAC03, a stress induced NAC gene family transcription factor in Norway spruce leads to reduced flavonol biosynthesis and aberrant embryo development. BMC Plant Biol 17:1–17. https://doi.org/10.1186/s12870-016-0952-8
Ding Y, Lawrence CE (2003) A statistical sampling algorithm for RNA secondary structure prediction. Nucleic Acids Res 31:7280–7301. https://doi.org/10.1093/nar/gkg938
Duval M, Hsieh TF, Kim SY, Thomas TL (2002) Molecular characterization of AtNAM: a member of the Arabidopsis NAC domain superfamily. Plant Mol Biol 50:237–248. https://doi.org/10.1023/a:1016028530943
Dwyer JT, Peterson J (2013) Tea and flavonoids: where we are, where to go next. Am J Clin Nutr 98:1611S-1618S. https://doi.org/10.3945/ajcn.113.059584
Gilbert N (2019) The science of tea’s mood-altering magic. Nature 566:S8–S9. https://doi.org/10.1038/d41586-019-00398-1
Hegedus D, Yu M, Baldwin D, Gruber M, Sharpe A, Parkin I, Whitwill S, Lydiate D (2003) Molecular characterization of Brassica napus NAC domain transcriptional activators induced in response to biotic and abiotic stress. Plant Mol Biol 53:383–397. https://doi.org/10.1023/b:plan.0000006944.61384.11
Jaganath IB, Crozier A (2011) Flavonoid biosynthesis. Plant metabolism and biotechnology. Wiley, New Jersey. https://doi.org/10.1002/9781119991311
Jiang X, Huang K, Zheng G, Hou H, Wang P, Jiang H, Zhao X, Li M, Zhang S, Liu Y, Gao L, Zhao L, XiaT, (2018) CsMYB5a and CsMYB5e from Camellia sinensis differentially regulate anthocyanin and proanthocyanidin biosynthesis. Plant Sci 270:209–220. https://doi.org/10.1016/j.plantsci.2018.02.009
Jiang G, Li Z, Song Y, Zhu H, Lin S, Huang R, Jiang Y, Duan X (2019) LcNAC13 physically interacts with LcR1MYB1 to coregulate anthocyanin biosynthesis-related genes during litchi fruit ripening. Biomolecules 9:135. https://doi.org/10.3390/biom9040135
Jiang W, Xia Y, Su X, Pang Y (2022) ARF2 positively regulates flavonols and proanthocyanidins biosynthesis in Arabidopsis thaliana. Planta 256(2):44. https://doi.org/10.1007/s00425-022-03936-w
Khan N, Mukhtar H (2013) Tea and health: studies in humans. Curr Pharm 19:6141–6147. https://doi.org/10.2174/1381612811319340008
Kikuchi K, Ueguchi-Tanaka M, Yoshida K, Nagato Y, Matsusoka M, Hirano HY (2000) Molecular analysis of the NAC gene family in rice. Mol Gen Genet 262:1047–1051. https://doi.org/10.1007/pl00008647
Kim S-Y, Kim S-G, Kim Y-S, Seo PJ, Bae M, Yoon H-K, Park C-M (2007) Exploring membrane-associated NAC transcription factors in Arabidopsis: implications for membrane biology in genome regulation. Nucleic Acids Res 35:203–213. https://doi.org/10.1093/nar/gkl1068
Lee SH, Hannink M (2003) Molecular mechanisms that regulate transcription factor localization suggest new targets for drug development. Adv Drug Deliv Rev 55:717–731. https://doi.org/10.1016/s0169-409x(03)00052-8
Lescot M, Déhais P, Thijs G, Marchal K, Moreau Y, Van de Peer Y, Rouzé P, Rombauts S (2002) PlantCARE, a database of plant cis-acting regulatory elements and a portal to tools for in silico analysis of promoter sequences. Nucleic Acids Res 30:325–327. https://doi.org/10.1093/nar/30.1.325
Li C, Bai Y, Li S, Chen H, Han X, Zhao H, Shao J, Park SU, Wu Q (2012) Cloning, characterization, and activity analysis of a flavonol synthase gene FtFLS1 and its association with flavonoid content in tartary buckwheat. J Agric Food Chem 60:5161–5168. https://doi.org/10.1021/jf205192q
Li CF, Zhu Y, Yu Y, Zhao QY, Wang SJ, Wang XC, Yao MZ, Luo D, Li X, Chen L, Yang YJ (2015) Global transcriptome and gene regulation network for secondary metabolite biosynthesis of tea plant (Camellia sinensis). BMC Genomics 16(1):560. https://doi.org/10.1186/s12864-015-1773-0
Li H, Guo L, Yan M, Hu J, Lin Q, Wang P, Wang M, Zhao H, Wang Y, Ni D (2022) A rapid and efficient transient expression system for gene function and subcellular localization studies in the tea plant (Camellia sinensis) leaves. Sci Hortic 297:110927. https://doi.org/10.1016/j.scienta.2022.110927
Li F, Deng X, Huang Z, Zhao Z, Li C, Song Q, He Y, Niu S (2023) Integrated transcriptome and metabolome provide insights into flavonoid biosynthesis in “P113”, a new purple tea of Camellia tachangensis. Beverage Plant Res. 3:3. https://doi.org/10.48130/BPR-2023-0003
Liu L, White MJ, MacRae TH (1999) Transcription factors and their genes in higher plants: functional domains, evolution and regulation. Eur J Biochem 262:247–257. https://doi.org/10.1046/j.1432-1327.1999.00349.x
Liu C, Long J, Zhu K, Liu L, Yang W, Zhang H, Li L, Xu Q, Deng X (2016) Characterization of a citrus R2R3-MYB transcription factor that regulates the flavonol and hydroxycinnamic acid biosynthesis. Sci Rep 6:25352. https://doi.org/10.1038/srep25352
Liu Y, Hou H, Jiang X, Wang P, Dai X, Chen W, Gao L, Xia T (2018) A WD40 repeat protein from Camellia sinensis regulates anthocyanin and proanthocyanidin accumulation through the formation of MYB–bHLH–WD40 ternary complexes. Int J Mol Sci 19:1686. https://doi.org/10.3390/ijms19061686
Mahmood K, Xu Z, El-Kereamy A, Casaretto JA, Rothstein SJ (2016) The Arabidopsis transcription factor ANAC032 represses anthocyanin biosynthesis in response to high sucrose and oxidative and abiotic stresses. Front Plant Sci 7:1548. https://doi.org/10.3389/fpls.2016.01548
Matsui K, Oshima Y, Mitsuda N, Sakamoto S, Nishiba Y, Walker AR, Ohme-Takagi M, Robinson SP, Yasui Y, Mori M, Takami H (2018) Buckwheat R2R3 MYB transcription factor FeMYBF1 regulates flavonol biosynthesis. Plant Sci 274:466–475. https://doi.org/10.1016/j.plantsci.2018.06.025
Morishita T, Kojima Y, Maruta T, Nishizawa-Yokoi A, Yabuta Y, Shigeoka S (2009) Arabidopsis NAC transcription factor, ANAC078, regulates flavonoid biosynthesis under high-light. Plant Cell Physiol 50:2210–2222. https://doi.org/10.1093/pcp/pcp159
Naik J, Rajput R, Pucker B, Stracke R, Pandey A (2021) The R2R3-MYB transcription factor MtMYB134 orchestrates flavonol biosynthesis in Medicago truncatula. Plant Mol Biol 106:157–172. https://doi.org/10.1007/s11103-021-01135-x
Ning G, Xiao X, Lv H, Li X, Zuo Y, Bao M (2012) Shortening tobacco life cycle accelerates functional gene identification in genomic research. Plant Biol 14:934–943. https://doi.org/10.1111/j.1438-8677.2012.00571.x
Olsen AN, Ernst HA, Leggio LL, Skriver K (2005) NAC transcription factors: structurally distinct, functionally diverse. Trends Plant Sci 10:79–87. https://doi.org/10.1016/j.tplants.2004.12.010
Ooka H, Satoh K, Doi K, Nagata T, Otomo Y, Murakami K, Matsubara K, Osato N, Kawai J, Carninci P, Hayashizaki Y, Suzuki K, Kojima K, Takahara Y, Yamamoto K, Kikuchi S (2003) Comprehensive analysis of NAC family genes in Oryza sativa and Arabidopsis thaliana. DNA Res 10:239–247. https://doi.org/10.1093/dnares/10.6.239
Osterlund MT, Hardtke CS, Wei N, Deng XW (2000) Targeted destabilization of HY5 during light-regulated development of Arabidopsis. Nature 405:462–466. https://doi.org/10.1038/35013076
Puranik S, Sahu PP, Srivastava PS, Prasad M (2012) NAC proteins: regulation and role in stress tolerance. Trends Plant Sci 17:369–381. https://doi.org/10.1016/j.tplants.2012.02.004
Quattrocchio F, Verweij W, Kroon A, Spelt C, Mol J, Koes R (2006) PH4 of Petunia is an R2R3 MYB protein that activates vacuolar acidification through interactions with basic-helix-loop-helix transcription factors of the anthocyanin pathway. Plant Cell 18:1274–1291. https://doi.org/10.1105/tpc.105.034041
Song S, Tao Y, Gao L, Liang H, Tang D, Lin J, Wang Y, Gmitter FG, Li C (2022) An integrated metabolome and transcriptome analysis reveal the regulation mechanisms of flavonoid biosynthesis in a purple tea plant cultivar. Front Plant Sci 13:880227. https://doi.org/10.3389/fpls.2022.880227
Sun Q, Jiang S, Zhang T, Xu H, Fang H, Zhang J, Su M, Wang Y, Zhang Z, Wang N (2019) Apple NAC transcription factor MdNAC52 regulates biosynthesis of anthocyanin and proanthocyanidin through MdMYB9 and MdMYB11. Plant Sci 289:110286. https://doi.org/10.1016/j.plantsci.2019.110286
Wang YX, Liu ZW, Wu ZJ, Li H, Zhuang J (2016) Transcriptome-wide identification and expression analysis of the NAC gene family in tea plant [Camellia sinensis (L.) O. Kuntze]. PLoS ONE 11:e0166727. https://doi.org/10.1371/journal.pone.0166727
Wang J, Lian W, Cao Y, Wang X, Wang G, Qi C, Liu L, Qin S, Yuan X, Li X, Ren S, Guo YD (2018) Overexpression of BoNAC019, a NAC transcription factor from Brassica oleracea, negatively regulates the dehydration response and anthocyanin biosynthesis in Arabidopsis. Sci Rep 8:13349. https://doi.org/10.1038/s41598-018-31690-1
Wang Z, Wang S, Xiao Y, Li Z, Wu M, Xie X, Li H, Mu W, Li F, Liu P, Wang R, Yang J (2020) Functional characterization of a HD-ZIP IV transcription factor NtHDG2 in regulating flavonols biosynthesis in Nicotiana tabacum. Plant Physiol Biochem 146:259–268. https://doi.org/10.1016/j.plaphy.2019.11.033
Xie Q, Sanz-Burgos AP, Guo H, García JA, Gutiérrez C (1999) GRAB proteins, novel members of the NAC domain family, isolated by their interaction with a geminivirus protein. Plant Mol Biol 39:647–656. https://doi.org/10.1023/a:1006138221874
Yasumasa M, Miho S, Atsushi H, Eiji N, Shigeru I (2006) Isolation of cDNAs for R2R3-MYB, bHLH and WDR transcriptional regulators and identification of c and ca mutations conferring white flowers in the Japanese morning glory. Plant Cell Physiol 4:457. https://doi.org/10.1093/pcp/pcj012
Ye D, Chan CY, Lawrence CE (2005) RNA secondary structure prediction by centroids in a Boltzmann weighted ensemble. RNA 11:1157–1166. https://doi.org/10.1261/rna.2500605
Zhao X, Zeng X, Lin N, Yu S, Fernie AR, Zhao J (2021) CsbZIP1-CsMYB12 mediates the production of bitter-tasting flavonols in tea plants (Camellia sinensis) through a coordinated activator-repressor network. Hortic Res 8:110. https://doi.org/10.1038/s41438-021-00545-8
Zhou H, Lin-Wang K, Wang H, Gu C, Dare AP, Espley RV, He H, Allan AC, Han Y (2015) Molecular genetics of blood-fleshed peach reveals activation of anthocyanin biosynthesis by NC transcription factors. Plant J 82:105–121. https://doi.org/10.1111/tpj.12792
Zhuang J, Dai X, Zhu M, Zhang S, Dai Q, Jiang X, Liu Y, Gao L, Xia T (2020) Evaluation of astringent taste of green tea through mass spectrometry-based targeted metabolic profiling of polyphenols. Food Chem 305:125507. https://doi.org/10.1016/j.foodchem.2019.125507
Acknowledgements
This work was financially supported by the Zhejiang Provincial Natural Science Foundation of China (No. LY19C160003), open fund project of Key Laboratory of Biology, Genetics and Breeding of Special Economic Animals and Plants, Ministry of Agriculture and Rural Affairs, P. R. China (No. TZDZW202203), and the Scientific Research Project of Zhejiang Education Department (Y202353282). This work was supported by the Public Research Platform of the College of Horticultural Science, Zhejiang A&F University.
Funding
Natural Science Foundation of Zhejiang Province, LY19C160003, Chun-Fang Li, Key Laboratory of Biology, Genetics and Breeding of Special Economic Animals and Plants, Ministry of Agriculture and Rural Affairs, P. R. China, TZDZW202203, Chun-Fang Li. The Scientific Research Project of Zhejiang Education Department, Y202353282, Wei-Xi Ran.
Author information
Authors and Affiliations
Contributions
CFL and LC designed the experiments and coordinated the project. SSS and WXR conducted the AsODN analyses, metabolite concentration analysis, and gene expression level analysis. LHG performed gene cloning, transcriptional activity analysis, and gene overexpression in tobacco. YCW and WYL performed the yeast one-hybrid assays. YT carried out the subcellular analysis. CFL, LC, and SSS were responsible for writing and editing the manuscript. All authors have reviewed and approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by De-Yu Xie.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Song, SS., Ran, WX., Gao, LH. et al. A functional study reveals CsNAC086 regulated the biosynthesis of flavonols in Camellia sinensis. Planta 259, 147 (2024). https://doi.org/10.1007/s00425-024-04426-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-024-04426-x