Abstract
Main conclusion
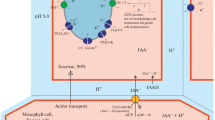
Guard cell- or mesophyll cell-localized phytochromes do not have a predominant direct light sensory role in red- or blue-light-mediated stomatal opening or far-red-light-mediated stomatal closure of Arabidopsis.
Abstract
The role of phytochromes in blue- and red-light-mediated stomatal opening, and far-red-light- mediated decrease in opening, is still under debate. It is not clear whether reduced stomatal opening in a phytochrome B (phyB) mutant line, is due to phytochrome acting as a direct photosensor or an indirect growth effect. The exact tissue localization of the phytochrome photoreceptor important for stomatal opening is also not known. We studied differences in stomatal opening in an Arabidopsis phyB mutant, and lines showing mesophyll cell-specific or guard cell-specific inactivation of phytochromes. Stomatal conductance (gs) of intact leaves was measured under red, blue, and blue + far-red light. Lines exhibiting guard cell-specific inactivation of phytochrome did not show a change in gs under blue or red light compared to Col-0. phyB consistently exhibited a reduction in gs under both blue and red light. Addition of far-red light did not have a significant impact on the blue- or red-light-mediated stomatal response. Treatment of leaves with DCMU (3-(3,4-dichlorophenyl)-1,1-dimethylurea), a photosynthetic electron transport (PET) inhibitor, eliminated the response to red light in all lines, indicating that stomatal opening under red light is controlled by PET, and not directly by phytochrome. Similar to previous studies, leaves of the phyB mutant line had fewer stomata. Overall, phytochrome does not appear have a predominant direct sensory role in stomatal opening under red or blue light. However, phytochromes likely have an indirect effect on the degree of stomatal opening under light through effects on leaf growth and stomatal development.





Similar content being viewed by others
Data availability
All data are available from TDS upon request.
References
Anderson JM, Price DG, Chow WS, Hope AB, Badger MR (1997) Reduced levels of cytochrome bf complex in transgenic tobacco leads to marked photochemical reduction of the plastoquinone pool, without significant change in acclimation to irradiance. Photosynth Res 53(2):215–227. https://doi.org/10.1023/a:1005856615915
Ando E, Kinoshita T (2018) Red light-induced phosphorylation of plasma membrane H+-ATPase in stomatal guard cells. Plant Physiol 178(2):838–849. https://doi.org/10.1104/pp.18.00544
Assmann SM (1988) Enhancement of the stomatal response to blue light by red light, reduced intercellular concentrations of CO2, and low vapor pressure differences. Plant Physiol 87(1):226–231
Berry JA, Beerling DJ, Franks PJ (2010) Stomata: key players in the earth system, past and present. Curr Opin Plant Biol 13(3):233–240. https://doi.org/10.1016/j.pbi.2010.04.013
Busch FA (2014) Opinion: the red-light response of stomatal movement is sensed by the redox state of the photosynthetic electron transport chain. Photosynth Res 119(1–2):131–140. https://doi.org/10.1007/s11120-013-9805-6
Doi M, Shimazaki K-I (2008) The stomata of the fern Adiantum capillus-veneris do not respond to CO2 in the dark and open by photosynthesis in guard cells. Plant Physiol 147(2):922–930. https://doi.org/10.1104/pp.108.118950
Drake PL, Froend RH, Franks PJ (2013) Smaller, faster stomata: scaling of stomatal size, rate of response, and stomatal conductance. J Exp Bot 64(2):495–505. https://doi.org/10.1093/jxb/ers347
Dwyer SA, Chow WS, Yamori W, Evans JR, Kaines S, Badger MR, von Caemmerer S (2012) Antisense reductions in the PsbO protein of photosystem II leads to decreased quantum yield but similar maximal photosynthetic rates. J Exp Bot 63(13):4781–4795
Evans L, Allaway W (1972) Action spectrum for the opening of Albizia Julibrissin pinnules, and the role of phytochrome in the closing movements of pinnules and of stomata of Vicla Faba. Aust J Biol Sci 25(5):885–894. https://doi.org/10.1071/BI9720885
Fan L-M, Zhao Z, Assmann SM (2004) Guard cells: a dynamic signaling model. Curr Opin Plant Biol 7:537–546. https://doi.org/10.1016/j.pbi.2004.07.009
Habermann HM (1973) Evidence for two photoreactions and possible involvement of phytochrome in light-dependent stomatal opening. Plant Physiol 51(3):543–548
Habermann HM (1974) Light reactions of stomatal opening: possible involvement of phytochrome. In: Bielski RL, Ferguson AR, Cresswell MM (eds) Mechanisms of Regulation of Plant Growth. Royal Society of New Zealand, Wellington, pp 445–453
Hajirezaei MR, Peisker M, Tschiersch H, Palatnik JF, Valle EM, Carrillo N, Sonnewald U (2002) Small changes in the activity of chloroplastic NADP+-dependent ferredoxin oxidoreductase lead to impaired plant growth and restrict photosynthetic activity of transgenic tobacco plants. Plant J 29(3):281–293
Holalu SV, Reddy SK, Blackman BK, Finlayson SA (2020) Phytochrome interacting factors 4 and 5 regulate axillary branching via bud abscisic acid and stem auxin signalling. Plant Cell Environ 43(9): 2224–2238. https://doi.org/10.1111/pce.13824
Holmes MG, Klein WH (1985) Evidence for phytochrome involvement in light-mediated stomatal movement in Phaseolus vulgaris L. Planta 166(3):348–353. https://doi.org/10.1007/bf00401172
Kami C, Lorrain S, Hornitschek P, Fankhauser C (2010) Light-regulated plant growth and development. Curr Top Dev Biol 91:29–66. https://doi.org/10.1016/s0070-2153(10)91002-8
Kang C-Y, Lian H-L, Wang F-F, Huang J-R, Yang H-Q (2009) Cryptochromes, phytochromes, and COP1 regulate light-controlled stomatal development in Arabidopsis. Plant Cell 21(9):2624–2641. https://doi.org/10.1105/tpc.109.069765
Karlsson PE (1987) The blue light regulation of stomatal movements. Universty of Goteborg Goteborg, Sweden
Karlsson PE (1988) Phytochrome is not involved in the red-light-enhancement of the stomatal blue-light-response in wheat seedlings. Physiol Plant 74(3):544–548. https://doi.org/10.1111/j.1399-3054.1988.tb02016.x
Li J, Li G, Wang H, Deng XW (2011) Phytochrome signaling mechanisms. Arabidopsis Book 9:e0148–e0148. https://doi.org/10.1199/tab.0148
Li N, Bo C, Zhang Y, Wang L (2021) PHYTOCHROME INTERACTING FACTORS PIF4 and PIF5 promote heat stress induced leaf senescence in Arabidopsis. J Exp Bot 72:4577–4589. https://doi.org/10.1093/jxb/erab158
Mott KA, Peak D (2018) Effects of the mesophyll on stomatal responses in amphistomatous leaves. Plant Cell Environ 41(12):2835–2843. https://doi.org/10.1111/pce.13411
Oh S, Kong Q, Montgomery BL (2022) Guard-cell phytochromes impact seedling photomorphogenesis and rosette leaf morphology. MicroPubl Biol. https://doi.org/10.17912/micropub.biology.000521
Quick WP, Schurr U, Scheibe R, Schulze ED, Rodermel SR, Bogorad L, Stitt M (1991) Decreased ribulose-1,5-bisphosphate carboxylase-oxygenase in transgenic tobacco transformed with “antisense” rbcS: I. Impact on photosynthesis in ambient growth conditions. Planta 183(4):542–554
Raschke K, Fellows MP (1971) Transient stomatal responses to red light mediated by phytochrome? In: Filner P, Lamport DTA, Ross JD, Yang N-S, Wolk CP (eds) Plant Research ’71. Plant Research Laboratory. Michiga State University, East Lansing
Roth-Bejerano N, Itai C (1981) Involvement of phytochrome in stomatal movement: effect of blue and red light. Physiol Plant 52(2):201–206. https://doi.org/10.1111/j.1399-3054.1981.tb08494.x
Roth-Bejerano N, Nejidat A, Itai C (1990) Regulation of stomatal opening in epidermal strips of Commelina communis L. by phytochrome. Biochem Physiol Pflanz 186(5):375–379. https://doi.org/10.1016/S0015-3796(11)80236-2
Sharkey TD, Raschke K (1981a) Effect of light quality on stomatal opening in leaves of Xanthium strumarium L. Plant Physiol 68(5):1170–1174
Sharkey TD, Raschke K (1981b) Separation and measurement of direct and indirect effects of light on stomata. Plant Physiol 68(1):33–40
Shimazaki K, Doi M, Assmann SM, Kinoshita T (2007) Light regulation of stomatal movement. Annu Rev Plant Biol 58:219–247
Sokolskaya SV, Sveshnikova NV, Kochetova GV, Solovchenko AE, Gostimski SA, Bashtanova OB (2003) Involvement of phytochrome in regulation of transpiration: red-/far red-induced responses in the chlorophyll-deficient mutant of pea. Funct Plant Biol 30(12):1249–1259
Suetsugu N, Takami T, Ebisu Y, Watanabe H, Iiboshi C, Doi M, Shimazaki K (2014) Guard cell chloroplasts are essential for blue light-dependent stomatal opening in Arabidopsis. PLoS ONE 9(9):e108374
Talbott LD, Zhu J, Han SW, Zeiger E (2002) Phytochrome and blue light-mediated stomatal opening in the orchid, Paphiopedilum. Plant Cell Physiol 43(6):639–646
Talbott LD, Shmayevich IJ, Chung Y, Hammad JW, Zeiger E (2003) Blue light and phytochrome-mediated stomatal opening in the npq1and phot1 phot2 mutants of Arabidopsis. Plant Physiol 133(4):1522–1529. https://doi.org/10.1104/pp.103.029587
Wang FF, Lian HL, Kang CY, Yang HQ (2010) Phytochrome B is involved in mediating red light-induced stomatal opening in Arabidopsis thaliana. Mol Plant 3(1):246–259
Wang Y, Noguchi K, Terashima I (2011) Photosynthesis-dependent and -independent responses of stomata to blue, red and green monochromatic light: differences between the normally oriented and inverted leaves of sunflower. Plant Cell Physiol 52(3):479–489. https://doi.org/10.1093/pcp/pcr005
Wang W, Liu Z, Bao L-J, Zhang S-S, Zhang C-G, Li X, Li H-X, Zhang X-L, Bones AM, Yang Z-B, Chen Y-L (2017) The RopGEF2-ROP7/ROP2 pathway activated by phyb suppresses red light-induced stomatal opening. Plant Physiol 174:717–731. https://doi.org/10.1104/pp.16.01727
Weraduwage SM, Campos ML, Yoshida Y, Major I, Kim Y-S, Kim S-J, Renna L, Anozie FC, Brandizzi F, Thomashow MF, Howe GA, Sharkey TD (2018) Molecular mechanisms determining leaf architecture. In: Adams WW, Terashima I (eds) The leaf: a platform for performing photosynthesis. Advances in photosynthesis and respiration (including bioenergy and related processes), vol 44. Springer, Cham,
Acknowledgements
We wish to thank Prof. Beronda Montgomery of the Plant Research Laboratory, Michigan State University, for generously sharing Arabidopsis seeds of all plant lines used in the current study, and for the tremendous support and guidance given to us throughout this research. We are also grateful to Dr. Sookyung Oh (Montgomery Lab) for her assistance. We thank Jim Klug and Cody Keilen (Growth Chamber Facility, Michigan State University) for their assistance, undergraduate student Lydia Sanchez (Department of Biochemistry and Molecular Biology, Michigan State University) for helping with growth and maintenance of plants, and all members of the Sharkey lab for their support.
Funding
Funding for participation of S.M.W. in this research was supported by the Chemical Sciences, Geosciences and Biosciences Division, Office of Basic Energy Sciences, Office of Science, U.S. Department of Energy (award no. DE-FG02-91ER 20021). Partial salary support for T.D.S. came from Michigan AgBioResearch.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Communicated by Anastasios Melis.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Weraduwage, S.M., Frame, M.K. & Sharkey, T.D. Role of guard cell- or mesophyll cell-localized phytochromes in stomatal responses to blue, red, and far-red light. Planta 256, 55 (2022). https://doi.org/10.1007/s00425-022-03967-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00425-022-03967-3