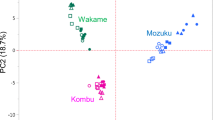
Abstract
Main Conclusion
Among seaweed groups, brown algae had characteristically high concentrations of mannitol, and green algae were characterised by fructose. In red algae, metabolite profiles of individual species should be evaluated.
Seaweeds are metabolically different from terrestrial plants. However, general metabolite profiles of the three major seaweed groups, the brown, red, and green algae, and the effect of various extraction methods on metabolite profiling results have not been comprehensively explored. In this study, we evaluated the water-soluble metabolites in four brown, five red, and two green algae species collected from two sites in northern Japan, located in the Sea of Japan and the Pacific Ocean. Freeze-dried seaweed samples were processed by methanol–water extraction with or without chloroform and analysed by capillary electrophoresis- and liquid chromatography-mass spectrometry for metabolite characterisation. The metabolite concentration profiles showed distinctive characteristic depends on species and taxonomic groups, whereas the extraction methods did not have a significant effect. Taxonomic differences between the various seaweed metabolite profiles were well defined using only sugar metabolites but no other major compound types. Mannitol was the main sugar metabolites in brown algae, whereas fructose, sucrose, and glucose were found at high concentrations in green algae. In red algae, individual species had some characteristic metabolites, such as sorbitol in Pyropia pseudolinearis and panose in Dasya sessilis. The metabolite profiles generated in this study will be a resource and provide guidance for nutraceutical research studies because the information about metabolites in seaweeds is still very limited compared to that of terrestrial plants.








Similar content being viewed by others
Abbreviations
- CE:
-
Capillary electrophoresis
- MS:
-
Mass spectrometry
- LC:
-
Liquid chromatography
- NMR:
-
Nuclear magnetic resonance
- GC:
-
Gas chromatography
- PCA:
-
Principle component analysis
- MeOH:
-
Methanol
- CHCl3 :
-
Chloroform
- H2O:
-
Water
- MES:
-
2-(N-morpholino)ethanesulfonic acid
- CSA:
-
d-camphor-10-sulfonic acid
- IS:
-
Internal standard
- ESI:
-
Electrospray ionisation
- TOF:
-
Time-of-flight
- SD:
-
Standard deviation
- Asp:
-
Aspartate
- Glu:
-
Glutamate
- Asn:
-
Asparagine
- Pro:
-
Proline
- Gly:
-
Glycine
- Phe:
-
Phenylalanine
- Arg:
-
Arginine
- Trp:
-
Tryptophan
- Ala:
-
Alanine
- Ser:
-
Serine
- Gln:
-
Glutamine
- DNA:
-
Deoxyribonucleic acid
- HPLC:
-
High-performance liquid chromatography
- 3PG:
-
3-Phosphoglycerate
- 2PG:
-
2-Phosphoglycerate
- 2AB:
-
2-Aminobutyrate
- G6P:
-
Glucose-6-phosphate
- kV:
-
Kilovolt
- i.d.:
-
Inner diameter
References
Astorga-España MS, Rodríguez-Galdón B, Rodríguez-Rodríguez EM, Díaz-Romero C (2016) Amino acid content in seaweeds from the Magellan Straits (Chile). J Food Compos Anal 53:77–84. https://doi.org/10.1016/j.jfca.2016.09.004
Belghit I, Rasinger JD, Heesch S et al (2017) In-depth metabolic profiling of marine macroalgae confirms strong biochemical differences between brown, red and green algae. Algal Res 26:240–249. https://doi.org/10.1016/j.algal.2017.08.001
Biancarosa I, Espe M, Bruckner CG et al (2017) Amino acid composition, protein content, and nitrogen-to-protein conversion factors of 21 seaweed species from Norwegian waters. J Appl Phycol 29:1001–1009. https://doi.org/10.1007/s10811-016-0984-3
Bolton MD (2009) Primary metabolism and plant defense—fuel for the fire. Mol Plant Microb Interact 22:487–497. https://doi.org/10.1094/MPMI-22-5-0487
Cai J, Henion J, Toxicology A, Drive W (1995) Capillary electrophoresis-mass spectrometry. J Chromatogr A 703:667–692. https://doi.org/10.1016/0021-9673(94)01178-H
Cavalier-Smith T (2000) Membrane heredity and early chloroplast evolution. Trends Plant Sci 5:174–182. https://doi.org/10.1007/978-3-319-06947-0_19
Chan JC-C, Cheung PC-K, Ang PO (1997) Comparative studies on the effect of three drying methods on the nutritional composition of seaweed Sargassum hemiphyllum (Turn.) C. Ag. J Agric Food Chem 45:3056–3059. https://doi.org/10.1021/jf9701749
Chan CX, Zauner S, Wheeler G et al (2012) Analysis of porphyra membrane transporters demonstrates gene transfer among photosynthetic eukaryotes and numerous sodium-coupled transport systems. Plant Physiol 158:2001–2012. https://doi.org/10.1104/pp.112.193896
Cock JM, Coelho SM, Brownlee C, Taylor AR (2010) The ectocarpus genome sequence: insights into brown algal biology and the evolutionary diversity of the eukaryotes. N Phytol 188:1–4. https://doi.org/10.1111/j.1469-8137.2010.03454.x
Collén J, Porcel B, Carré W et al (2013) Genome structure and metabolic features in the red seaweed Chondrus crispus shed light on evolution of the Archaeplastida. Proc Natl Acad Sci USA 110:5247–5252. https://doi.org/10.1073/pnas.1221259110
Collins K, Fitzgerald G, Stanton C, Ross R (2016) Looking beyond the terrestrial: the potential of seaweed derived bioactives to treat non-communicable diseases. Mar Drugs 14:60. https://doi.org/10.3390/md14030060
Costa LS, Fidelis GP, Cordeiro SL et al (2010) Biological activities of sulfated polysaccharides from tropical seaweeds. Biomed Pharmacother 64:21–28. https://doi.org/10.1016/j.biopha.2009.03.005
Date Y, Sakata K, Kikuchi J (2012) Chemical profiling of complex biochemical mixtures from various seaweeds. Polym J 44:888–894. https://doi.org/10.1038/pj.2012.105
Davis GDJ, Vasanthi AHR (2011) Seaweed metabolite database (SWMD): a database of natural compounds from marine algae. Bioinformation 5:361–364. https://doi.org/10.6026/97320630005361
Dawczynski C, Schubert R, Jahreis G (2007) Amino acids, fatty acids, and dietary fibre in edible seaweed products. Food Chem 103:891–899. https://doi.org/10.1016/j.foodchem.2006.09.041
De Clerck O, Bogaert KA, Leliaert F (2012) Diversity and evolution of algae. In: Piganeau G (ed) Advances in botanical research, vol 64. Elsevier, pp 55–86
de Raad M, Fischer CR, Northen TR (2016) High-throughput platforms for metabolomics. Curr Opin Chem Biol 30:7–13. https://doi.org/10.1016/j.cbpa.2015.10.012
Diniz GS (2011) Gross chemical profile and calculation of nitrogen-to-protein conversion factors for five tropical seaweeds. Am J Plant Sci 02:287–296. https://doi.org/10.4236/ajps.2011.23032
El-Said GF, El-Sikaily A (2013) Chemical composition of some seaweed from Mediterranean Sea coast, Egypt. Environ Monit Assess 185:6089–6099. https://doi.org/10.1007/s10661-012-3009-y
Endo H, Suehiro K, Kinoshita J, Agatsuma Y (2015) Combined effects of temperature and nutrient enrichment on palatability of the brown alga Sargassum yezoense (Yamada) Yoshida & T. Konno. Am J Plant Sci 06:275–282. https://doi.org/10.4236/ajps.2015.62031
Ernst M, Silva DB, Silva RR et al (2014) Mass spectrometry in plant metabolomics strategies: from analytical platforms to data acquisition and processing. Nat Prod Rep 31:784. https://doi.org/10.1039/c3np70086k
Fleurence J (2010) Seaweed proteins: biochemical, nutritional aspects and potential uses. Trends Food Sci Technol 10:25–28. https://doi.org/10.1088/1751-8113/44/8/085201
Garcia-Vaquero M, Rajauria G, O’Doherty JV, Sweeney T (2017) Polysaccharides from macroalgae: recent advances, innovative technologies and challenges in extraction and purification. Food Res Int 99:1011–1020. https://doi.org/10.1016/j.foodres.2016.11.016
Goulitquer S, Potin P, Tonon T (2012) Mass spectrometry-based metabolomics to elucidate functions in marine organisms and ecosystems. Mar Drugs 10:849–880. https://doi.org/10.3390/md10040849
Groisillier A, Shao Z, Michel G et al (2014) Mannitol metabolism in brown algae involves a new phosphatase family. J Exp Bot 65:559–570. https://doi.org/10.1093/jxb/ert405
Gu H, Zhang P, Zhu J, Raftery D (2015) Globally optimized targeted mass spectrometry: reliable metabolomics analysis with broad coverage. Anal Chem 87:12355–12362. https://doi.org/10.1021/acs.analchem.5b03812
Guiry MD, Guiry GM (2018) Algaebase: listing the world’s algae. In: World-wide electron. Publ. Natl. Univ. Ireland, Galway, Irel. http://www.algaebase.org/. Accessed 27 Sep 2018
Gupta S, Abu-Ghannam N (2011) Bioactive potential and possible health effects of edible brown seaweeds. Trends Food Sci Technol 22:315–326. https://doi.org/10.1016/j.tifs.2011.03.011
Gupta V, Thakur RS, Reddy CRK, Jha B (2013) Central metabolic processes of marine macrophytic algae revealed from NMR based metabolome analysis. RSC Adv 3:7037. https://doi.org/10.1039/c3ra23017a
Gupta V, Thakur RS, Baghel RS et al (2014) Seaweed metabolomics. Advances in botanical research, vol Serial. Academic Press, Cambridge, pp 31–52
Hamed I, Özogul F, Özogul Y, Regenstein JM (2015) Marine bioactive compounds and their health benefits: a review. Compr Rev Food Sci Food Saf 14:446–465. https://doi.org/10.1111/1541-4337.12136
Hamid SS, Wakayama M, Soga T, Tomita M (2018) Drying and extraction effects on three edible brown seaweeds for metabolomics. J Appl Phycol 30:3335–3350. https://doi.org/10.1007/s10811-018-1614-z
Hartmann A, Becker K, Karsten U et al (2015) Analysis of mycosporine-like amino acids in selected algae and cyanobacteria by hydrophilic interaction liquid chromatography and a novel MAA from the red alga Catenella repens. Mar Drugs 13:6291–6305. https://doi.org/10.3390/md13106291
Heyman HM, Dubery IA (2016) The potential of mass spectrometry imaging in plant metabolomics: a review. Phytochem Rev 15:297–316. https://doi.org/10.1007/s11101-015-9416-2
Hirayama A, Wakayama M, Soga T (2014) Metabolome analysis based on capillary electrophoresis-mass spectrometry. TrAC Trends Anal Chem 61:215–222. https://doi.org/10.1016/j.trac.2014.05.005
Hwang ES, Ki KN, Chung HY (2013) Proximate composition, amino acid, mineral, and heavy metal content of dried laver. Prev Nutr Food Sci 18:139–144. https://doi.org/10.3746/pnf.2013.18.2.139
Kim HK, Verpoorte R (2010) Sample preparation for plant metabolomics. Phytochem Anal 21:4–13. https://doi.org/10.1002/pca.1188
Kim DH, Lee SB, Kim SK et al (2016) Optimization and evaluation of sugars and chemicals production from green macro-algae Enteromorpha intestinalis. Bioenergy Res 9:1155–1166. https://doi.org/10.1007/s12155-016-9759-6
Kumari P, Bijo AJ, Mantri VA et al (2013) Fatty acid profiling of tropical marine macroalgae: an analysis from chemotaxonomic and nutritional perspectives. Phytochemistry 86:44–56. https://doi.org/10.1016/j.phytochem.2012.10.015
La Barre SL, Weinberger F, Kervarec N, Potin P (2004) Monitoring defensive responses in macroalgae—limitations and perspectives. Phytochem Rev 3:371–379. https://doi.org/10.1007/s11101-005-1459-3
Laturnus F (1996) Volatile halocarbons released from Arctic macroalgae. Mar Chem 55:359–366. https://doi.org/10.1016/S0304-4203(97)89401-7
Leal MC, Munro MHG, Blunt JW et al (2013) Biogeography and biodiscovery hotspots of macroalgal marine natural products. Nat Prod Rep 30:1380–1390. https://doi.org/10.1039/c3np70057g
Lei Z, Huhman DV, Sumner LW (2011) Mass spectrometry strategies in metabolomics. J Biol Chem 286:25435–25442. https://doi.org/10.1074/jbc.R111.238691
Lourenço SO, Barbarino E, De-Paula JC et al (2002) Amino acid composition, protein content and calculation of nitrogen-to-protein conversion factors for 19 tropical seaweeds. Phycol Res 50:233–241. https://doi.org/10.1046/j.1440-1835.2002.00278.x
Manns D, Deutschle AL, Saake B, Meyer AS (2014) Methodology for quantitative determination of the carbohydrate composition of brown seaweeds (Laminariaceae). RSC Adv 4:25736–25746. https://doi.org/10.1039/C4RA03537B
Michalak I, Chojnacka K (2015) Algae as production systems of bioactive compounds. Eng Life Sci 15:160–176. https://doi.org/10.1002/elsc.201400191
Murtagh F, Legendre P (2014) Ward’s hierarchical agglomerative clustering method: Which algorithms implement ward’s criterion? J Classif 31:274–295. https://doi.org/10.1007/s00357-014-9161-z
Nakamura Y, Mochamad Afendi F, Kawsar Parvin A et al (2014) KNApSAcK metabolite activity database for retrieving the relationships between metabolites and biological activities. Plant Cell Physiol 55:1–9. https://doi.org/10.1093/pcp/pct176
Narsai R, Rocha M, Geigenberger P et al (2011) Comparative analysis between plant species of transcriptional and metabolic responses to hypoxia. N Phytol 190:472–487. https://doi.org/10.1111/j.1469-8137.2010.03589.x
Nishitsuji K, Arimoto A, Iwai K et al (2016) A draft genome of the brown alga, Cladosiphon okamuranus, S-strain: a platform for future studies of ‘mozuku’ biology. DNA Res 23:561–570. https://doi.org/10.1093/dnares/dsw039
Norziah MH, Ching CY (2000) Nutritional composition of edible seaweed Gracilaria changgi. Food Chem 68:69–76
Omar MMA, Elbashir AA, Schmitz OJ (2017) Capillary electrophoresis method with UV-detection for analysis of free amino acids concentrations in food. Food Chem 214:300–307. https://doi.org/10.1016/j.foodchem.2016.07.060
Ortiz-Tena JG, Rühmann B, Schieder D, Sieber V (2016) Revealing the diversity of algal monosaccharides: fast carbohydrate fingerprinting of microalgae using crude biomass and showcasing sugar distribution in Chlorella vulgaris by biomass fractionation. Algal Res 17:227–235. https://doi.org/10.1016/j.algal.2016.05.008
Paiva L, Lima E, Neto AI, Baptista J (2018) Seasonal variability of the biochemical composition and antioxidant properties of Fucus spiralis at two Azorean Islands. Mar Drugs. https://doi.org/10.3390/md16080248
Palanisamy SK, Arumugam V, Rajendran S et al (2018) Chemical diversity and anti-proliferative activity of marine algae. Nat Prod Res. https://doi.org/10.1080/14786419.2018.1488701
Parjikolaei BR, Bruhn A, Eybye KL et al (2016) Valuable biomolecules from nine north atlantic red macroalgae: amino acids, fatty acids, carotenoids, minerals and metals. Nat Resour 07:157–183. https://doi.org/10.4236/nr.2016.74016
Peinado I, Girón J, Koutsidis G, Ames JM (2014) Chemical composition, antioxidant activity and sensory evaluation of five different species of brown edible seaweeds. Food Res Int 66:36–44. https://doi.org/10.1016/j.foodres.2014.08.035
Percival E (1979) The polysaccharides of green, red and brown seaweeds: their basic structure, biosynthesis and function. Br Phycol J 14:103–117. https://doi.org/10.1080/00071617900650121
Qasim R (1991) Amino acid composition of some common seaweeds. Pak J Pharm Sci 4:49–54
Ramautar R, Somsen GW, de Jong GJ (2009) CE–MS in metabolomics. Electrophoresis 30:276–291. https://doi.org/10.1002/elps.200800512
Robin A, Chavel P, Chemodanov A et al (2017) Diversity of monosaccharides in marine macroalgae from the Eastern Mediterranean Sea. Algal Res 28:118–127. https://doi.org/10.1016/j.algal.2017.10.005
Robledo D, Freile Pelegrín Y (1997) Chemical and mineral composition of six potentially edible seaweed species of Yucatán. Bot Mar 40:301–306. https://doi.org/10.1515/botm.1997.40.1-6.301
Rodrigues D, Freitas AC, Pereira L et al (2015) Chemical composition of red, brown and green macroalgae from Buarcos bay in central west coast of Portugal. Food Chem 183:197–207. https://doi.org/10.1016/j.foodchem.2015.03.057
Rousvoal S, Groisillier A, Dittami SM et al (2011) Mannitol-1-phosphate dehydrogenase activity in Ectocarpus siliculosus, a key role for mannitol synthesis in brown algae. Planta 233:261–273. https://doi.org/10.1007/s00425-010-1295-6
Ryall K, Harper JT, Keeling PJ (2003) Plastid-derived type II fatty acid biosynthetic enzymes in chromists. Gene 313:139–148. https://doi.org/10.1016/S0378-1119(03)00671-1
Skriptsova AV, Shevchenko NM, Zvyagintseva TN, Imbs TI (2010) Monthly changes in the content and monosaccharide composition of fucoidan from Undaria pinnatifida (Laminariales, Phaeophyta). J Appl Phycol 22:79–86. https://doi.org/10.1007/s10811-009-9438-5
Soga T, Heiger DN (2000) Amino acid analysis by capillary electrophoresis electrospray ionization mass spectrometry. Anal Chem 72:1236-1241. https://doi.org/10.1021/ac990976y
Soga T, Igarashi K, Ito C et al (2009) Metabolomic profiling of anionic metabolites by capillary electrophoresis mass spectrometry. Anal Chem 81:6165–6174. https://doi.org/10.1021/ac900675k
Sugimoto M, Kawakami M, Robert M et al (2012) Bioinformatics tools for mass spectroscopy-based metabolomic data processing and analysis. Curr Bioinform 7:96–108. https://doi.org/10.2174/157489312799304431
Tabarsa M, Rezaei M, Ramezanpour Z, Waaland JR (2012) Chemical compositions of the marine algae Gracilaria salicornia (Rhodophyta) and Ulva lactuca (Chlorophyta) as a potential food source. J Sci Food Agric 92:2500–2506. https://doi.org/10.1002/jsfa.5659
Taboada MC, Millán R, Miguez MI (2013) Nutritional value of the marine algae wakame (Undaria pinnatifida) and nori (Porphyra purpurea) as food supplements. J Appl Phycol 25:1271–1276. https://doi.org/10.1007/s10811-012-9951-9
Thompson GA (1996) Lipids and membrane function in green algae. Biochim Biophys Acta Lipids Lipid Metab 1302:17–45. https://doi.org/10.1016/0005-2760(96)00045-8
Wahbeh MI (1997) Amino acid and fatty acid profiles of four species of macroalgae from Aqaba and their suitability for use in fish diets. Aquaculture 159:101–109. https://doi.org/10.1016/S0044-8486(97)00183-X
Wakayama M, Aoki N, Sasaki H, Ohsugi R (2010) Simultaneous analysis of amino acids and carboxylic acids by capillary electrophoresis–mass spectrometry using an acidic electrolyte and uncoated fused-silica capillary. Anal Chem 82:9967–9976. https://doi.org/10.1021/ac1019039
Wakayama M, Hirayama A, Soga T (2015) Capillary electrophoresis-mass spectrometry. In: Bjerrum JT (ed) Metabonomics: methods and protocols, methods in molecular biology. Springer, New York, pp 113–122
West J, Calumpong HP, Martin G (2016) Seaweeds. In: Nations U (ed) The first global integrated marine assessment. Cambridge University Press, Cambridge, pp 223–228
Wong K, Cheung PC (2001a) Nutritional evaluation of some subtropical red and green seaweeds part II. In vitro protein digestibility and amino acid profiles of protein concentrates. Food Chem 72:11–17. https://doi.org/10.1016/S0308-8146(00)00176-X
Wong K, Cheung PC (2001b) Influence of drying treatment on three Sargassum species 2. Protein extractability, in vitro protein digestibility and amino acid profile of protein concentrates. J Appl Phycol 13:51–58
Wu X, Jiang W, Lu J et al (2014) Analysis of the monosaccharide composition of water-soluble polysaccharides from Sargassum fusiforme by high performance liquid chromatography/electrospray ionisation mass spectrometry. Food Chem 145:976–983. https://doi.org/10.1016/j.foodchem.2013.09.019
Zhou AY, Robertson J, Hamid N et al (2015) Changes in total nitrogen and amino acid composition of New Zealand Undaria pinnatifida with growth, location and plant parts. Food Chem 186:319–325. https://doi.org/10.1016/j.foodchem.2014.06.016
Acknowledgements
For samples collection and species identification, the authors would like to thank all those who have contributed directly or indirectly in this research; all staffs of Muroran Marine Station, Yamagata Prefecture Fisheries Experiment Station, and Mr. Yohichi Homma, fishermen from Wasada port (Tsuruoka city). This study was funded by the Yamagata prefectural government and Tsuruoka city. It was also supported by a scholarship from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT), Japan, for an international student (Hamid, S.S).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hamid, S.S., Wakayama, M., Ichihara, K. et al. Metabolome profiling of various seaweed species discriminates between brown, red, and green algae. Planta 249, 1921–1947 (2019). https://doi.org/10.1007/s00425-019-03134-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-019-03134-1