Abstract
Main conclusion
Transcriptome analysis and biochemical characterization of the putative l-3,4-dihydroxyphenylalanine (l-DOPA) pathway in Mucuna pruriens (L.) DC have been performed. Spatio-temporal quantification of the putative l-DOPA biosynthetic pathway genes and its correlation with respective metabolites was established. l-tyrosine, l-DOPA, and dopamine from all plant parts were quantified.
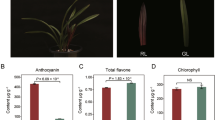
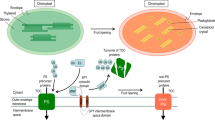
The de novo transcriptome analysis was performed using leaves of the selected M. pruriens mutant T-IV-9 during maturity. The putative l-DOPA pathway and its regulatory genes were retrieved from transcriptome data and the l-DOPA pathway was biochemically characterized. The spatial and temporal gene expression for the l-DOPA pathway was identified with respect to the chemical constituents. l-tyrosine, l-DOPA, and dopamine contents were highest in leaves during maturity (about 170-day-old plants). The polyphenol oxidase (PPO) was highly expressed in tender stems (230-fold higher as compared to seeds) as well as a high l-DOPA content. The PPO gene was highly expressed in leaves (3367.93 in FPKM) with a 79-fold increase compared to control plants during maturity. l-DOPA was found in every part with varied levels. The highest l-DOPA content was found in mature dried seed (3.18–5.8%), whereas the lowest amount was recorded in mature and dried leaves. The reproductive parts of the plant had a higher amount of l-DOPA content (0.9–5.8%) compared to the vegetative parts (0.2–0.91%). Various amino acid transporters and permeases were expressed in M. pruriens. The transcripts of dopa decarboxylase (DDC) were found in almost all parts of the plant, but its higher content was limited to the leaf.





Similar content being viewed by others
Abbreviations
- AD:
-
Arogenate dehydrogenase
- DDC:
-
Dopa/tyrosine decarboxylase
- L-DOPA:
-
l-3, 4-dihydroxyphenylalanine
- PPO:
-
Polyphenol oxidase
References
Agbafor KN, Nwachukwu N, Agbafor KN, Nwachukwu N (2011) Phytochemical analysis and antioxidant property of leaf extracts of Vitex doniana and Mucuna pruriens. Biochem Res Int 2011:e459839. https://doi.org/10.1155/2011/459839
Altschul SF, Gish W, Miller W et al (1990) Basic local alignment search tool. J Mol Biol 215:403–410. https://doi.org/10.1016/S0022-2836(05)80360-2
Daxenbichler ME, VanEtten CH, Earle FR, Tallent WH (1972) L-DOPA recovery from Mucuna seed. J Agric Food Chem 20:1046–1047
Dhawan SS, Rai GK, Darokar MP et al (2011) Comparative genetic analysis of trichome-less and normal pod genotypes of Mucuna pruriens (Fabaceae). Genet Mol Res 10:2049–2056. https://doi.org/10.4238/vol10-3gmr1264
Dhawan SS, Shukla P, Gupta P, Lal RK (2016) A cold-tolerant evergreen interspecific hybrid of Ocimum kilimandscharicum and Ocimum basilicum: analyzing trichomes and molecular variations. Protoplasma 253:845–855. https://doi.org/10.1007/s00709-015-0847-9
Eze JI, Ndukwe S (2011) Effect of methanolic extract of Mucuna pruriens seed on the immune response of mice. Comp Clin Pathol 21:1343–1347. https://doi.org/10.1007/s00580-011-1294-4
Fischer W-N, André B, Rentsch D et al (1998) Amino acid transport in plants. Trends Plant Sci 3:188–195. https://doi.org/10.1016/S1360-1385(98)01231-X
Fujii Y, Shibuya T, Yasuda T (1991) l-3,4-Dihydroxyphenylalanine as an allelochemical candidate from Mucuna pruriens (L.) DC. var. utilis. Agric Biol Chem 55:617–618. https://doi.org/10.1080/00021369.1991.10870627
Gaines CG, Byng GS, Whitaker RJ, Jensen RA (1982) l-tyrosine regulation and biosynthesis via arogenate dehydrogenase in suspension-cultured cells of Nicotiana silvestris Speg. et Comes. Planta 156:233–240. https://doi.org/10.1007/BF00393730
Gomes BR, de Siqueira-Soares RC, Dos Santos WD et al (2014) The effects of dopamine on antioxidant enzymes activities and reactive oxygen species levels in soybean roots. Plant Signal Behav 9:e977704. https://doi.org/10.4161/15592324.2014.977704
Grabherr MG, Haas BJ, Yassour M et al (2011) Trinity: reconstructing a full-length transcriptome without a genome from RNA-Seq data. Nat Biotechnol 29:644–652. https://doi.org/10.1038/nbt.1883
Ito S, Wakamatsu K (2008) Chemistry of mixed melanogenesis—pivotal roles of dopaquinone. Photochem Photobiol 84:582–592. https://doi.org/10.1111/j.1751-1097.2007.00238.x
Kumar A, Gupta C, Nair DT, Salunke DM (2016a) MP-4 contributes to snake venom neutralization by Mucuna pruriens seeds through an indirect antibody-mediated mechanism. J Biol Chem 291:11373–11384. https://doi.org/10.1074/jbc.M115.699173
Kumar S, Stecher G, Tamura K (2016b) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol Evol 33:1870–1874. https://doi.org/10.1093/molbev/msw054
Lampariello LR, Cortelazzo A, Guerranti R et al (2012) The magic velvet bean of Mucuna pruriens. J Tradit Complement Med 2:331–339
Lieu CA, Venkiteswaran K, Gilmour TP et al (2012) The antiparkinsonian and antidyskinetic mechanisms of Mucuna pruriens in the MPTP-treated nonhuman primate. Evid Based Compl Alt Med 2012:e840247. https://doi.org/10.1155/2012/840247
Luthra PM, Singh S (2010) Identification and optimization of tyrosine hydroxylase activity in Mucuna pruriens DC. var. utilis. Planta 231:1361–1369. https://doi.org/10.1007/s00425-010-1140-y
Marchler-Bauer A, Derbyshire MK, Gonzales NR et al (2015) CDD: NCBI’s conserved domain database. Nucleic Acids Res 43:D222–D226. https://doi.org/10.1093/nar/gku1221
Matsumoto H (2011) The mechanisms of phytotoxic action and selectivity of non-protein aromatic amino acids L-DOPA and m-tyrosine. J Pestic Sci 36:1–8. https://doi.org/10.1584/jpestics.R10-15
Mishra A, Lal RK, Chanotiya CS, Dhawan SS (2016) Genetic elaborations of glandular and non-glandular trichomes in Mentha arvensis genotypes: assessing genotypic and phenotypic correlations along with gene expressions. Protoplasma 254:1045–1061. https://doi.org/10.1007/s00709-016-1011-x
Misra L, Wagner H (2007) Extraction of bioactive principles from Mucuna pruriens seeds. Indian J Biochem Biophys 44:56–60
Mushtaq MN, Sunohara Y, Matsumoto H (2013) Bioactive l-DOPA induced quinoprotein formation to inhibit root growth of cucumber seedlings. J Pestic Sci 38:68–73. https://doi.org/10.1584/jpestics.D13-005
Nair P, Misra A, Singh A et al (2013) Differentially expressed genes during contrasting growth stages of Artemisia annua for artemisinin content. PLoS One 8:e60375. https://doi.org/10.1371/journal.pone.0060375
Ortiz-Lopez A, Chang H-C, Bush DR (2000) Amino acid transporters in plants. Biochim Biophys Acta BBA Biomembr 1465:275–280. https://doi.org/10.1016/S0005-2736(00)00144-9
Raghavendra S, Kumar V, Ramesh CK, Khan MHM (2012) Enhanced production of L-DOPA in cell cultures of Mucuna pruriens L. and Mucuna prurita H. Nat Prod Res 26:792–801. https://doi.org/10.1080/14786419.2011.553721
Sanchita Chauhan R, Soni G et al (2013) Docking and molecular dynamics studies of peptide inhibitors of ornithine decarboxylase: a rate-limiting enzyme for the metabolism of Fusarium solani. J Biomol Struct Dyn 31:874–887. https://doi.org/10.1080/07391102.2012.718526
Sathyanarayana N, Pittala RK, Tripathi PK et al (2017) Transcriptomic resources for the medicinal legume Mucuna pruriens: de-novo transcriptome assembly, annotation, identification and validation of EST-SSR markers. BMC Genom 18:409. https://doi.org/10.1186/s12864-017-3780-9
Scire A, Tanfani F, Bertoli E et al (2011) The belonging of gpMuc, a glycoprotein from Mucuna pruriens seeds, to the Kunitz-type trypsin inhibitor family explains its direct anti-snake venom activity. Phytomedicine 18:887–895. https://doi.org/10.1016/j.phymed.2011.02.004
Singh SK, Dhawan SS (2017) Analyzing trichomes and spatio-temporal expression of a cysteine protease gene Mucunain in Mucuna pruriens L. (DC). Protoplasma 255:575–584. https://doi.org/10.1007/s00709-017-1164-2
Singh AP, Sarkar S, Tripathi M, Rajender S (2013) Mucuna pruriens and its major constituent l-DOPA recover spermatogenic loss by combating ROS, loss of mitochondrial membrane potential and apoptosis. PLoS One 8:e54655. https://doi.org/10.1371/journal.pone.0054655
Singh SK, Yadav D, Lal RK et al (2016) Inducing mutations through γ-irradiation in seeds of Mucuna pruriens for developing high l-DOPA-yielding genotypes. Int J Radiat Biol 93:426–432. https://doi.org/10.1080/09553002.2016.1254832
Smith TA (1977) Phenethylamine and related compounds in plants. Phytochemistry 16:9–18. https://doi.org/10.1016/0031-9422(77)83004-5
Trapnell C, Williams BA, Pertea G et al (2010) Transcript assembly and quantification by RNA-Seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat Biotechnol 28:511–515. https://doi.org/10.1038/nbt.1621
Wichers HJ, Visser JF, Huizing HJ, Pras N (1993) Occurrence of l-DOPA and dopamine in plants and cell cultures of Mucuna pruriens and effects of 2,4-D and NaCl on these compounds. Plant Cell Tissue Organ Cult 33:259–264. https://doi.org/10.1007/BF02319010
Acknowledgements
We gratefully acknowledge the support and facilities provided by the Director CSIR-CIMAP Lucknow, India and senior research fellowship to the first author from ICMR (Indian Council Medical Research), New Delhi, India. The funding for this work was acknowledged from AGTEC (BSC0110- a CSIR network programme). We are highly thankful to Dr. M.M. Gupta for his kind support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare no conflict of interests.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Singh, S.K., Dhawan, S.S., Lal, R.K. et al. Biochemical characterization and spatio-temporal analysis of the putative l-DOPA pathway in Mucuna pruriens. Planta 248, 1277–1287 (2018). https://doi.org/10.1007/s00425-018-2978-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-018-2978-7