Abstract
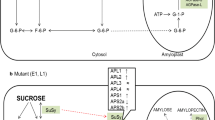
Two glucanotransferases, disproportionating enzyme 1 (StDPE1) and disproportionating enzyme 2 (StDPE2), were repressed using RNA interference technology in potato, leading to plants repressed in either isoform individually, or both simultaneously. This is the first detailed report of their combined repression. Plants lacking StDPE1 accumulated slightly more starch in their leaves than control plants and high levels of maltotriose, while those lacking StDPE2 contained maltose and large amounts of starch. Plants repressed in both isoforms accumulated similar amounts of starch to those lacking StDPE2. In addition, they contained a range of malto-oligosaccharides from maltose to maltoheptaose. Plants repressed in both isoforms had chlorotic leaves and did not grow as well as either the controls or lines where only one of the isoforms was repressed. Examination of photosynthetic parameters suggested that this was most likely due to a decrease in carbon assimilation. The subcellular localisation of StDPE2 was re-addressed in parallel with DPE2 from Arabidopsis thaliana by transient expression of yellow fluorescent protein fusions in tobacco. No translocation to the chloroplasts was observed for any of the fusion proteins, supporting a cytosolic role of the StDPE2 enzyme in leaf starch metabolism, as has been observed for Arabidopsis DPE2. It is concluded that StDPE1 and StDPE2 have individual essential roles in starch metabolism in potato and consequently repression of these disables regulation of leaf malto-oligosaccharides, starch content and photosynthetic activity and thereby plant growth possibly by a negative feedback mechanism.







Similar content being viewed by others
Abbreviations
- At:
-
Arabidopsis thaliana
- CBM20:
-
Carbohydrate binding module 20
- CLSM:
-
Confocal laser scanning microscopy
- DPE:
-
Disproportionating enzyme
- ISA3:
-
Isoamylase 3
- LDA:
-
Limit dextrinase
- MEX1:
-
Maltose excess 1
- MOS:
-
Malto-oligosaccharides
- St:
-
Solanum tuberosum
- PSI:
-
Photosystem I
- PSII:
-
Photosystem II
- YFP:
-
Yellow fluorescent protein
References
Baunsgaard L, Lütken H, Mikkelsen R, Glaring MA, Pharm TT, Blennow A (2005) A novel isoform of glucan, water dikinase phosphorylates pre-phosporylated α-glucans and is involved in starch degradation. Plant J 41:595–605
Blennow A, Bay-Smidt AM, Wischmann B, Olsen CE, Møller BL (1998) The degree of starch phosphorylation is related to the chain length distribution of the neutral and the phosphorylated chains of amylopectin. Carbohydr Res 307:45–54
Bradford MM (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Chia T, Thorneycroft D, Chapple A, Messerli G, Chen J, Zeeman SC, Smith SM, Smith AM (2004) A cytosolic glucosyltransferase is required for conversion of starch to sucrose in Arabidopsis leaves at night. Plant J 37:853–863
Christiansen C, Hachem MA, Janeček Š, Viksø-Nielsen A, Blennow A, Svensson B (2009) The carbohydrate-binding module family 20—diversity, structure, and function. FEBS J 276:5006–5029
Critchley JH, Zeeman SC, Takaha T, Smith AM, Smith SM (2001) A critical role for disproportionating enzyme in starch breakdown is revealed by a knock-out mutation in Arabidopsis. Plant J 26:89–100
Delatte T, Umhang M, Trevisan M, Eicke S, Thorneycroft D, Smith SM, Zeeman SC (2006) Evidence for distinct mechanisms of starch granule breakdown in plants. J Biol Chem 281:12050–12059
Fettke J, Poeste S, Eckermann N, Tiessen A, Pauly M, Geigenberger P, Steup M (2005) Analysis of cytosolic heteroglycans from leaves of transgenic potato (Solanum tuberosum L.) plants that under- or overexpress the Pho 2 phosphorylase isozyme. Plant Cell Physiol 46:1987–2004
Fettke J, Chia T, Eckermann N, Smith A, Steup M (2006) A transglucosidase necessary for starch degradation and maltose metabolism in leaves at night acts on cytosolic heteroglycans (SHG). Plant J 46:668–684
Fettke J, Hejazi M, Smirnova J, Höchel E, Stage M, Steup M (2009) Eukaryotic starch degradation: integration of plastidial and cytosolic pathways. J Exp Bot 60:2907–2922
Fulton DC, Stettler M, Mettler T, Vaughan CK, Li J, Francisco P, Gil M, Reinhold H, Eicke S, Messerli G, Dorken G, Halliday K, Smith AM, Smith SM, Zeeman SC (2008) β-AMYLASE4, a noncatalytic protein required for starch breakdown, acts upstream of three active β-amylases in Arabidopsis chloroplasts. Plant Cell 20:1040–1058
Glaring MA, Zygadlo A, Thorneycroft D, Schulz A, Smith SA, Blennow A, Baunsgaard Lone (2007) An extra-plastidial α-glucan, water dikinase from Arabidopsis phosphorylates amylopectin in vitro and is not necessary for transient starch degradation. J Exp Bot 58:3949–3960
Haldrup A, Naver H, Scheller HV (1999) The interaction between plastocyanin and photosystem I is inefficient in transgenic Arabidopsis plants lacking the PSI-N subunit of photosystem I. Plant J 17:689–698
Haldrup A, Simpson DJ, Scheller HV (2000) Down-regulation of the PSI-F subunit of photosystem I (PSI) in Arabidopsis thaliana. The PSI-F subunit is essential for photoautotrophic growth and contributes to antenna function. J Biol Chem 275:31211–31218
Kaplan F, Guy CL (2005) RNA interference of Arabidopsis beta-amylase8 prevents maltose accumulation upon cold shock and increases sensitivity of PSII photochemical efficiency to freezing stress. Plant J 44:730–743
Kötting O, Pusch K, Tiessen A, Geigenberger P, Steup M, Ritte G (2005) Identification of a novel enzyme required for starch metabolism in Arabidopsis leaves. The phosphoglucan, water dikinase. Plant Physiol 137:242–252
Kötting O, Kossmann J, Zeeman SC, Lloyd JR (2010) Regulation of starch metabolism: the age of enlightenment? Curr Opin Plant Biol. doi:10.1016/j.pbi.2010.01.003
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Methods Enzymol 148:350–382
Lloyd JR, Blennow A, Burhenne K, Kossmann J (2004) Repression of a novel isoform of disproportionating enzyme (stDPE2) in potato leads to inhibition of starch degradation in leaves but not tubers stored at low temperature. Plant Physiol 134:1347–1354
Lloyd JR, Kossmann J, Ritte G (2005) Leaf starch degradation comes out of the shadows. Trends Plant Sci 10:130–137
Lorberth R, Ritte G, Willmitzer L, Kossmann J (1998) Inhibition of a starch-granule-bound protein leads to modified starch and repression of cold sweetening. Nat Biotechnol 16:473–477
Lu Y, Sharkey TD (2004) The role of amylomaltase in maltose metabolism in the cytosol of photosynthetic cells. Planta 218:466–473
Monte E, Ludevid D, Prat S (1999) Leaf C40.4: a carotenoid-associated protein involved in the modulation of photosynthetic efficiency? Plant J 19:399–410
Müller-Röber B, Kossmann J, Hannah LC, Willmitzer L, Sonnewald U (1990) One of two different ADP-glucose pyrophosphorylase genes from potato responds strongly to elevated levels of sucrose. Mol Gen Genet 224:136–146
Müller-Röber B, Sonnewald U, Willmitzer L (1992) Inhibition of the ADP-glucose pyrophosphorylase in transgenic potatoes leads to sugar-storing tubers and influences tuber formation and expression of tuber storage proteins. EMBO J 11:1229–1238
Nielsen TH, Skjærbæk HC, Karlsen P (1991) Carbohydrate metabolism during fruit development in sweet pepper (Capsicum annuum) plants. Physiol Plant 82:311–319
Nielsen TH, Deiting U, Stitt M (1997) A β-amylase in potato tubers is induced by storage at low temperature. Plant Physiol 113:503–510
Niittylä T, Messerli G, Trevisan M, Chen J, Smith AM, Zeeman S (2004) A previously unknown maltose transporter essential for starch degradation in leaves. Science 303:87–89
Nour-Eldin HH, Hansen BG, Nørholm MHH, Jensen JK, Halkier BA (2006) Advancing uracil-excision based cloning towards an ideal technique for cloning PCR fragments. Nucleic Acids Res 34:e122
Quick WP, Stitt M (1989) An examination of factors contributing to non-photochemical quenching of chlorophyll in barley leaves. Biochim Biophys Acta 977:287–296
Scheidig A, Fröhlich A, Schulze S, Lloyd JR, Kossmann J (2002) Downregulation of a chloroplast-targeted β-amylase leads to a starch excess phenotype in leaves. Plant J 30:581–591
Schneider A, Hausler RE, Kolukisaoglu U, Kunze R, van der Graaf E, Schwacke R, Catoni E, Desimone M, Flügge U-I (2002) An Arabidopsis thaliana knock-out mutant of the triose phosphate/phosphate translocator is severely compromised only when starch synthesis, but not starch mobilisation is abolished. Plant J 32:685–699
Smith AM, Zeeman SC, Smith SM (2005) Starch degradation. Annu Rev Plant Biol 56:73–98
Soitamo AJ, Zhou G, Clarke AK, Oquist G, Aro EM, Gustafsson P (1994) Over-production of the D1 protein of photosystem II reaction centre in the cyanobacterium Synechococcus sp. PCC 7942. Plant Mol Biol 26:709–721
Steichen JM, Petty RV, Sharkey TD (2008) Domain characterization of a 4-α-glucanotransferase essential for maltose metabolism in photosynthetic leaves. J Biol Chem 283:20797–20804
Stensballe A, Hald S, Bauw G, Blennow A, Welinder KG (2008) The amyloplast proteome of potato tuber. FEBS J 275:1723–1741
Stettler M, Eicke S, Mettler T, Messerli G, Hörtensteiner S, Zeeman SC (2009) Blocking the metabolism of starch breakdown products in Arabidopsis leaves triggers chloroplast degradation. Mol Plant 2:1233–1246
Steup M (1990) Starch degrading enzymes. In: Lea PJ (ed) Methods in plant biochemistry, vol 3. Academic Press, London, pp 103–128
Takaha T, Yanase M, Okada S, Smith SM (1993) Disproportionating enzyme (4-α-glucanotransferase, EC 2.4.1.25) of potato. Purification, molecular cloning, and potential role in starch metabolism. J Biol Chem 268:1391–1396
Takaha T, Critchley J, Okada S, Smith SM (1998) Normal starch content and composition in tubers of antisense potato plants lacking D-enzyme (4-α-glucanotransferase). Planta 205:445–451
Voinnet O, Rivas S, Mestre P, Baulcombe D (2003) An enhanced transient expression system in plants based on suppression of gene silencing by the p19 protein of tomato bushy stunt virus. Plant J 33:949–956
von Schaewen A, Stitt M, Schmidt R, Sonnewald U, Willmitzer L (1990) Expression of a yeast-derived invertase in the cell wall of tobacco and Arabidopsis plants leads to accumulation of carbohydrate and inhibition of photosynthesis and strongly influences growth and phenotype of transgenic tobacco plants. EMBO J 9:3033–3044
Weise SE, Schrader SM, Kleinbeck KR, Sharkey TD (2006) Carbon balance and circadian regulation of hydrolytic and phosphorolytic breakdown of transitory starch. Plant Physiol 14:879–886
Wesley AV, Helliwell CA, Smith NA, Wang M, Rouse DT, Liu Q, Gooding PS, Singh SP, Abbott D, Stoutjesdijk PA, Robinson SP, Gleave AP, Green AG, Waterhouse PM (2001) Construct design for efficient, effective and high-throughput gene silencing in plants. Plant J 27:581–590
Wolf S (1993) Effect of leaf age on photosynthesis, carbon transport and carbon allocation in potato plants. Potato Res 36:253–262
Yu T-S, Kofler H, Häusler RE, Flügge U-I, Zeeman SC, Smith AM, Kossmann J, Lloyd JR, Ritte G, Steup M, Lue W-L, Chen J, Weber A (2001) The Arabidopsis sex1 mutant is defective in the R1 protein, a general regulator of starch degradation in plants, and not in the chloroplast hexose transporter. Plant Cell 13:1907–1918
Yu T-S, Zeeman SC, Thorneycroft D, Fulton DC, Dunstan H, Lue W-L, Hegemann B, Tung S-Y, Umemoto T, Chapple A, Tsai D-L, Wang SM, Smith AM, Chen J, Smith SM (2005) α-amylase is not required for breakdown of transitory starch in Arabidopsis leaves. J Biol Chem 280:9773–9779
Zeeman SC, Northrop F, Smith AM, ap Rees T (1998) A starch-accumulating mutant of Arabidopsis thaliana deficient in chloroplastic starch-hydrolysing enzyme. Plant J 15:357–365
Zeeman SC, Thorneycroft D, Schupp N, Chapple A, Weck M, Dunstan H, Haldimann P, Bechtold N, Smith AM, Smith SM (2004) Plastidial α-glucan phosphorylase is not required for starch degradation in Arabidopsis leaves but has a role in the tolerance of abiotic stress. Plant Physiol 135:849–858
Zeeman SC, Smith SM, Smith AM (2007) The diurnal metabolism of leaf starch. Biochem J 401:13–28
Zeeman SC, Kossmann J, Smith AM (2010) Starch: its metabolism, evolution and biotechnological modification in plants. Annu Rev Plant Biol 61:209–234
Acknowledgments
We thank Lis B. Møller, Per Lassen Nielsen and Camilla Falk for technical assistance as well as Claus Frohberg (Bayer Crop Science) for arranging the potato transformations. Poul Erik Jensen is acknowledged for fruitful discussions. This project was supported by The Danish National Research Foundation, The Danish Biotechnology Program and a stipend from the Danish Research School for Biotechnology (H.L.).
Conflict of interest
The authors declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
H. Lütken and J. R. Lloyd contributed equally to the work.
Rights and permissions
About this article
Cite this article
Lütken, H., Lloyd, J.R., Glaring, M.A. et al. Repression of both isoforms of disproportionating enzyme leads to higher malto-oligosaccharide content and reduced growth in potato. Planta 232, 1127–1139 (2010). https://doi.org/10.1007/s00425-010-1245-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00425-010-1245-3