Abstract
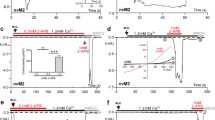
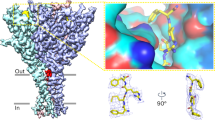
FMRFamide-gated Na+ channel (FaNaC) is a member of the DEG/ENaC family. Amino acid sequence of the second transmembrane region (TM2) of FaNaC is quite similar to that of the acid-sensing ion channels (ASIC) of the same family. In the upper part of TM2, there are two aspartate residues (D552 and D556 in Aplysia FaNaC, AkFaNaC) which construct two negative rings in the external vestibule. In the present study, we examined the function of D552/D556 mutants of AkFaNaC in Xenopus oocytes with special interest in Ca2+ sensitivity of FaNaC. The FMRFamide-evoked current through AkFaNaC was depressed by submillimolar Ca2+ such that the current in Ca2+-free condition was 2–3-fold larger than that in the control solution which contained 1.8 mM CaCl 2. Both D552 and D556 were found to be indispensable for the sensitivity of FaNaC to submillimolar Ca2+. Unexpectedly, however, both acidic residues were not essential for the inhibition by millimolar Ca2+ concentrations. The Ca2+-sensitive gating of FaNaC was recapitulated by an allosteric model in which Ca2+-bound channels are more difficult to open. The desensitization of FaNaC was also inhibited by Ca2+, which was abolished in some D552/D556 mutants. Structural models of FaNaC made by homology modeling showed that the distance between oxygen atoms of D552 and D556 on the adjacent subunits is close enough to coordinate Ca2+ in the nonconducting desensitized channel but not in the open channel. The results suggest that Ca2+ coordination between oxygen atoms of D552 and D556 disturbs the opening transition as well as the desensitization of FaNaC.








Similar content being viewed by others
References
Babini E, Paukert M, Geisler HS, Gründer S (2002) Alternative splicing and interaction with di- and polyvalent cations control the dynamic range of acid-sensing ion channel 1 (ASIC1). J Biol Chem 277(44):41597–41603
Baconguis I, Bohlen CJ, Goehring A, Julius D, Gouaux E (2014) X-ray structure of acid-sensing ion channel 1-snake toxin complex reveals open state of a Na+-selective channel. Cell 156(4):717–729
Boscardin E, Alijevic O, Hummler E, Frateschi S, Kellenberger S (2016) The function and regulation of acid-sensing ion channels (ASICs) and the epithelial Na+ channel (ENaC): IUPHAR review 19. Br J Pharmacol 173(18):2671–2701
Cottrell GA, Green KA, Davies NW (1990) The neuropeptide Phe-Met-Arg-Phe-NH 2 (FMRFamide) can activate a ligand-gated ion channel in Helix neurones. Pflugers Arch 416(5):612–614
Driscoll M, Chalfie M (1991) The mec-4 gene is a member of a family of Caenorhabditis elegans genes that can mutate to induce neuronal degeneration. Nature 349(6310):588–593
Dürrnagel S, Kuhn A, Tsiairis CD, Williamson M, Kalbacher H, Grimmelikhuijzen CJ, Holstein TW, Gründer S (2010) Three homologous subunits form a high affinity peptide-gated ion channel in Hydra. J Biol Chem 285(16):11958–11965
Dürrnagel S, Falkenburger BH, Gründer S (2012) High Ca2+ permeability of a peptide-gated DEG/ENaC from Hydra. J Gen Physiol 140(4):391–402
Furukawa Y, Miyawaki Y, Abe G (2006) Molecular cloning and functional characterization of the Aplysia FMRFamide-gated na + channel. Pflugers Arch 451(5):646–656
Golubovic A, Kuhn A, Williamson M, Kalbacher H, Holstein TW, Grimmelikhuijzen CJ, Gründer S (2007) A peptide-gated ion channel from the freshwater polyp Hydra. J Biol Chem 282(48):35098–35103
Gonzales EB, Kawate T, Gouaux E (2009) Pore architecture and ion sites in acid-sensing ion channels and P2X receptors. Nature 460(7255):599–604
Gründer S, Assmann M (2015) Peptide-gated ion channels and the simple nervous system of hydra. J Exp Biol 218(Pt 4):551–561
Hidalgo P, MacKinnon R (1995) Revealing the architecture of a K + channel pore through mutant cycles with a peptide inhibitor. Science 268(5208):307–310
Horovitz A (1996) Double-mutant cycles: a powerful tool for analyzing protein structure and function. Fold Des 1(6):R121–R126
Immke DC, McCleskey EW (2003) Protons open acid-sensing ion channels by catalyzing relief of Ca2+ blockade. Neuron 37:75–84
Jasti J, Furukawa H, Gonzales EB, Gouaux E (2007) Structure of acid-sensing ion channel 1 at 1.9 Å resolution and low pH. Nature 449(7160):316–323
Jeziorski MC, Green KA, Sommerville J, Cottrell GA (2000) Cloning and expression of a FMRFamide-gated Na+ channel from Helisoma trivolvis and comparison with the native neuronal channel. J Physiol (Lond.) 526 Pt 1:13–25
Kellenberger S, Gautschi I, Schild L (1999) A single point mutation in the pore region of the epithelial Na+ channel changes ion selectivity by modifying molecular sieving. Proc Natl Acad Sci USA 96(7):4170–4175
Kellenberger S, Hoffmann-Pochon N, Gautschi I, Schneeberger E, Schild L (1999) On the molecular basis of ion permeation in the epithelial Na+ channel, vol 114
Kodani Y, Furukawa Y (2010) Position 552 in a FMRFamide-gated Na+ channel affects the gating properties and the potency of FMRFamide. Zool Sci 27(5):440–448
Kodani Y, Furukawa Y (2014) Electrostatic charge at position 552 affects the activation and permeation of FMRFamide-gated Na+ channels. J Physiol Sci 64(2):141–150
Krissinel E, Henrick K (2007) Inference of macromolecular assemblies from crystalline state. J Mol Biol 372(3):774–797
Lefevre CM, Diakov A, Haerteis S, Korbmacher C, Gründer S, Wiemuth D (2014) Pharmacological and electrophysiological characterization of the human bile acid-sensitive ion channel (hBASIC). Pflugers Arch 466 (2):253–263
Li T, Yang Y, Canessa CM (2011a) Asp 433 in the closing gate of ASIC1 determines stability of the open state without changing properties of the selectivity filter or Ca2+ block. J Gen Physiol 137(3):289–297
Li T, Yang Y, Canessa CM (2011b) Outlines of the pore in open and closed conformations describe the gating mechanism of ASIC1. Nat Commun 2:399
Lingueglia E, Champigny G, Lazdunski M, Barbry P (1995) Cloning of the amiloride-sensitive FMRFamide peptide-gated sodium channel. Nature 378(6558):730–733
Lingueglia E, Deval E, Lazdunski M (2006) FMRFAmide-gated sodium channel and ASIC channels: a new class of ionotropic receptors for FMRFamide and related peptides. Peptides 27(5):1138–1152
Monod J, Wyman J, Changeux JP (1965) On the nature of allosteric transitions: a plausible model. J Mol Biol 12:88–118
Paukert M, Babini E, Pusch M (2004) Gründer S Identification of the Ca2+ blocking site of acid-sensing ion channel (ASIC) 1: implications for channel gating. J Gen Physiol 124:383–394
Paukert M, Chen X, Polleichtner G, Schindelin H, Gründer S (2008) Candidate amino acids involved in H + gating of acid-sensing ion channel 1a. J Biol Chem 283(1):572–581
Perry SJ, Straub VA, Schofield MG, Burke JF, Benjamin PR (2001) Neuronal expression of an FMRFamide-gated Na+ channel and its modulation by acid pH. J Neurosci 21(15):5559–5567
(2001) R: a language and environmentfor statistical computing. r foundation for statistical computing. Vienna, Austria URL http://www.R-project.org/. ISBN 3-900051-07-0
Sakai H, Lingueglia E, Champigny G, Mattei MG, Lazdunski M (1999) Cloning and functional expression of a novel degenerin-like Na+ channel gene in mammals. J Physiol (Lond) 519 Pt 2:323–333
Šali A, Blundell TL (1993) Comparative protein modelling by satisfaction of spatial restraints. J Mol Biol 234(3):779–815
Wang X, Kirberger M, Qiu F, Chen G, Yang JJ (2009) Towards predicting Ca2+-binding sites with different coordination numbers in proteins with atomic resolution. Proteins 75(4):787–798
Wiemuth D, Gründer S (2010) A single amino acid tunes Ca2+ inhibition of brain liver intestine Na+ channel (BLINaC). J Biol Chem 285(40):30404–30410
Wiemuth D, Assmann M, Gründer S (2014) The bile acid-sensitive ion channel (BASIC), the ignored cousin of ASICs and ENaC. Channels (Austin) 8(1):29–34
Zhainazarov AB, Cottrell GA (1998) Single-channel currents of a peptide-gated sodium channel expressed in Xenopus oocytes. J Physiol (Lond) 513(Pt 1):19–31
Zhang P, Sigworth FJ, Canessa CM (2006) Gating of acid-sensitive ion channel-1: release of Ca2+ block vs. allosteric mechanism. J Gen Physiol 127(2):109–117
Acknowledgments
This work was supported by JSPS KAKENHI Grant Number JP15K07149.
Author contributions
YF conceived research; YF designed experiments; AF and YK performed experiments; AF, YK, and YF analyzed data; YF performed the simulation and the homology modeling; YF wrote a manuscript; AF, YK, and YF approved the final version of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
This article is part of the Topical Collection on Ion channels, receptors and transporters
Rights and permissions
About this article
Cite this article
Fujimoto, A., Kodani, Y. & Furukawa, Y. Modulation of the FMRFamide-gated Na+ channel by external Ca2+ . Pflugers Arch - Eur J Physiol 469, 1335–1347 (2017). https://doi.org/10.1007/s00424-017-2021-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-017-2021-z