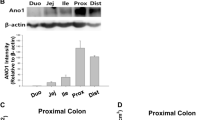
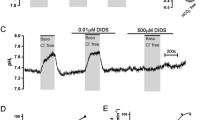
Abstract
The colonic mucosa protects itself from the luminal content by secreting mucus that keeps the bacteria at a distance from the epithelium. For this barrier to be effective, the mucus has to be constantly replenished which involves exocytosis and expansion of the secreted mucins. Mechanisms involved in regulation of mucus exocytosis and expansion are poorly understood, and the aim of this study was to investigate whether epithelial anion secretion regulates mucus formation in the colon. The muscarinic agonist carbachol was used to induce parallel secretion of anions and mucus, and by using established inhibitors of ion transport, we studied how inhibition of epithelial transport affected mucus formation in mouse colon. Anion secretion and mucin exocytosis were measured by changes in membrane current and epithelial capacitance, respectively. Mucus thickness measurements were used to determine the carbachol effect on mucus growth. The results showed that the carbachol-induced increase in membrane current was dependent on NKCC1 co-transport, basolateral K+ channels and Cftr activity. In contrast, the carbachol-induced increase in capacitance was partially dependent on NKCC1 and K+ channel activity, but did not require Cftr activity. Carbachol also induced an increase in mucus thickness that was inhibited by the NKCC1 blocker bumetanide. However, mice that lacked a functional Cftr channel did not respond to carbachol with an increase in mucus thickness, suggesting that carbachol-induced mucin expansion requires Cftr channel activity. In conclusion, these findings suggest that colonic epithelial transport regulates mucus formation by affecting both exocytosis and expansion of the mucin molecules.








Similar content being viewed by others
References
Atuma C, Strugala V, Allen A, Holm L (2001) The adherent gastrointestinal mucus gel layer: thickness and physical state in vivo. Am J Physiol Gastrointest Liver Physiol 280(5):G922–G929
Bachmann O, Reichelt D, Tuo B, Manns MP, Seidler U (2006) Carbachol increases Na+−HCO3- cotransport activity in murine colonic crypts in a M3-, Ca2+/calmodulin-, and PKC-dependent manner. Am J Physiol Gastrointest Liver Physiol 291(4):G650–G657. doi:10.1152/ajpgi.00376.2005
Bachmann O, Wuchner K, Rossmann H, Leipziger J, Osikowska B, Colledge WH, Ratcliff R, Evans MJ, Gregor M, Seidler U (2003) Expression and regulation of the Na+−K+−2Cl- cotransporter NKCC1 in the normal and CFTR-deficient murine colon. J Physiol 549(2):525–536. doi:10.1113/jphysiol.2002.030205
Bertrand CA, Danahay H, Poll CT, Laboisse C, Hopfer U, Bridges RJ (2004) Niflumic acid inhibits ATP-stimulated exocytosis in a mucin-secreting epithelial cell line. Am J Physiol Cell Physiol 286(2):C247–C255. doi:10.1152/ajpcell.00593.200200593.2002
Bertrand CA, Durand DM, Saidel GM, Laboisse C, Hopfer U (1998) System for dynamic measurements of membrane capacitance in intact epithelial monolayers. Biophys J 75(6):2743–2756
Bertrand CA, Laboisse CL, Hopfer U (1999) Purinergic and cholinergic agonists induce exocytosis from the same granule pool in HT29-Cl.16E monolayers. Am J Physiol 276(4 Pt 1):C907–C914
Cho HJ, Joo NS, Wine JJ (2010) Mucus secretion from individual submucosal glands of the ferret trachea. Am J Physiol Lung Cell Mol Physiol. doi:10.1152/ajplung.00049.2010
Choi JY, Joo NS, Krouse ME, Wu JV, Robbins RC, Ianowski JP, Hanrahan JW, Wine JJ (2007) Synergistic airway gland mucus secretion in response to vasoactive intestinal peptide and carbachol is lost in cystic fibrosis. J Clin Invest 117(10):3118–3127. doi:10.1172/JCI31992
Choi HS, Yang WS, Kim SC, Lee WI, Lee HJ, Choi JY (2010) Functional study of mucus secretion of the eustachian tube in guinea pigs. Otol Neurotol. doi:10.1097/MAO.0b013e3181d35e69
Cuffe JE, Bertog M, Velazquez-Rocha S, Dery O, Bunnett N, Korbmacher C (2002) Basolateral PAR-2 receptors mediate KCl secretion and inhibition of Na+ absorption in the mouse distal colon. J Physiol 539(Pt 1):209–222
Danahay H, Atherton HC, Jackson AD, Kreindler JL, Poll CT, Bridges RJ (2006) Membrane capacitance and conductance changes parallel mucin secretion in the human airway epithelium. Am J Physiol Lung Cell Mol Physiol 290(3):L558–L569
Davis PB (2006) Cystic fibrosis since 1938. Am J Respir Crit Care Med 173(5):475–482. doi:10.1164/rccm.200505-840OE
Dedek K, Waldegger S (2001) Colocalization of KCNQ1/KCNE channel subunits in the mouse gastrointestinal tract. Pflugers Arch 442(6):896–902
Dharmsathaphorn K, Pandol SJ (1986) Mechanism of chloride secretion induced by carbachol in a colonic epithelial cell line. J Clin Invest 77(2):348–354
Dolganov GM, Woodruff PG, Novikov AA, Zhang Y, Ferrando RE, Szubin R, Fahy JV (2001) A novel method of gene transcript profiling in airway biopsy homogenates reveals increased expression of a Na+−K+−Cl- cotransporter (NKCC1) in asthmatic subjects. Genome Res 11(9):1473–1483. doi:10.1101/gr.191301
Ermund A, Schutte A, Johansson ME, Gustafsson JK, Hansson GC (2013) Studies of mucus in mouse stomach, small intestine, and colon. I. Gastrointestinal mucus layers have different properties depending on location as well as over the Peyer’s patches. Am J Physiol Gastrointest Liver Physiol 305(5):G341–G347. doi:10.1152/ajpgi.00046.2013
Fischer H, Illek B, Negulescu PA, Clauss W, Machen TE (1992) Carbachol-activated calcium entry into HT-29 cells is regulated by both membrane potential and cell volume. Proc Natl Acad Sci U S A 89(4):1438–1442
Fischer KG, Leipziger J, Rubini-Illes P, Nitschke R, Greger R (1996) Attenuation of stimulated Ca2+ influx in colonic epithelial (HT29) cells by cAMP. Pflugers Arch 432(4):735–740
French PJ, van Doorninck JH, Peters RH, Verbeek E, Ameen NA, Marino CR, de Jonge HR, Bijman J, Scholte BJ (1996) A delta F508 mutation in mouse cystic fibrosis transmembrane conductance regulator results in a temperature-sensitive processing defect in vivo. J Clin Invest 98(6):1304–1312. doi:10.1172/JCI118917
Fu J, Wei B, Wen T, Johansson ME, Liu X, Bradford E, Thomsson KA, McGee S, Mansour L, Tong M, McDaniel JM, Sferra TJ, Turner JR, Chen H, Hansson GC, Braun J, Xia L (2011) Loss of intestinal core 1-derived O-glycans causes spontaneous colitis in mice. J Clin Invest 121(4):1657–1666. doi:10.1172/JCI4553845538
Garcia MA, Yang N, Quinton PM (2009) Normal mouse intestinal mucus release requires cystic fibrosis transmembrane regulator-dependent bicarbonate secretion. J Clin Invest 119(9):2613–2622. doi:10.1172/JCI38662
Geibel JP (2005) Secretion and absorption by colonic crypts. Annu Rev Physiol 67:471–490
Gopel S, Zhang Q, Eliasson L, Ma XS, Galvanovskis J, Kanno T, Salehi A, Rorsman P (2004) Capacitance measurements of exocytosis in mouse pancreatic alpha-, beta- and delta-cells within intact islets of Langerhans. J Physiol 556(Pt 3):711–726
Gouyer V, Gottrand F, Desseyn JL (2011) The extraordinarily complex but highly structured organization of intestinal mucus-gel unveiled in multicolor images. PLoS One 6(4):e18761. doi:10.1371/journal.pone.0018761
Greig ER, Boot-Handford RP, Mani V, Sandle GI (2004) Decreased expression of apical Na+ channels and basolateral Na+, K+−ATPase in ulcerative colitis. J Pathol 204(1):84–92
Greig E, Sandle GI (2000) Diarrhea in ulcerative colitis. The role of altered colonic sodium transport. Ann N Y Acad Sci 915:327–332
Grootjans J, Hundscheid IH, Lenaerts K, Boonen B, Renes IB, Verheyen FK, Dejong CH, von Meyenfeldt MF, Beets GL, Buurman WA (2013) Ischaemia-induced mucus barrier loss and bacterial penetration are rapidly counteracted by increased goblet cell secretory activity in human and rat colon. Gut 62(2):250–258. doi:10.1136/gutjnl-2011-301956
Gustafsson JK, Ermund A, Ambort D, Johansson ME, Nilsson HE, Thorell K, Hebert H, Sjovall H, Hansson GC (2012) Bicarbonate and functional CFTR channel are required for proper mucin secretion and link cystic fibrosis with its mucus phenotype. J Exp Med 209(7):1263–1272. doi:10.1084/jem.20120562
Gustafsson JK, Ermund A, Johansson ME, Schutte A, Hansson GC, Sjovall H (2012) An ex vivo method for studying mucus formation, properties, and thickness in human colonic biopsies and mouse small and large intestinal explants. Am J Physiol Gastrointest Liver Physiol 302(4):G430–G438. doi:10.1152/ajpgi.00405.2011.10.1152/ajpgi.00405.2011
Gustafsson JK, Hansson GC, Sjovall H (2012) Ulcerative colitis patients in remission have an altered secretory capacity in the proximal colon despite macroscopically normal mucosa. Neurogastroenterol Motil. doi:10.1111/j.1365-2982.2012.01958.x
Gustafsson JK, Hansson GC, Sjovall H (2012) Ulcerative colitis patients in remission have an altered secretory capacity in the proximal colon despite macroscopically normal mucosa. Neurogastroenterol Motil 24(8):e381–e391. doi:10.1111/j.1365-2982.2012.01958.x
Halm DR, Halm ST (2000) Secretagogue response of goblet cells and columnar cells in human colonic crypts. Am J Physiol Cell Physiol 278(1):C212–C233
Halm ST, Liao T, Halm DR (2006) Distinct K+ conductive pathways are required for Cl- and K+ secretion across distal colonic epithelium. Am J Physiol Cell Physiol 291(4):C636–C648
Hardcastle J, Hardcastle PT, Taylor CJ, Goldhill J (1991) Failure of cholinergic stimulation to induce a secretory response from the rectal mucosa in cystic fibrosis. Gut 32(9):1035–1039
Hecht G (1999) Innate mechanisms of epithelial host defense: spotlight on intestine. Am J Physiol 277(3 Pt 1):C351–C358
Hemlin M, Jodal M, Lundgren O, Sjovall H, Stage L (1988) The importance of the subepithelial resistance for the electrical properties of the rat jejunum in vitro. Acta Physiol Scand 134(1):79–88
Jakab RL, Collaco AM, Ameen NA (2011) Physiological relevance of cell-specific distribution patterns of CFTR, NKCC1, NBCe1, and NHE3 along the crypt-villus axis in the intestine. Am J Physiol Gastrointest Liver Physiol 300(1):G82–G98. doi:10.1152/ajpgi.00245.2010
Jakab RL, Collaco AM, Ameen NA (2012) Lubiprostone targets prostanoid signaling and promotes ion transporter trafficking, mucus exocytosis, and contractility. Dig Dis Sci 57(11):2826–2845. doi:10.1007/s10620-012-2352-8
Johansson ME (2012) Fast renewal of the distal colonic mucus layers by the surface goblet cells as measured by in vivo labeling of mucin glycoproteins. PLoS One 7(7):e41009. doi:10.1371/journal.pone.0041009
Johansson ME, Phillipson M, Petersson J, Velcich A, Holm L, Hansson GC (2008) The inner of the two Muc2 mucin-dependent mucus layers in colon is devoid of bacteria. Natl Acad Sci U S A 105(39):15064–15069. doi:10.1073/pnas.0803124105
Laburthe M, Augeron C, Rouyer-Fessard C, Roumagnac I, Maoret JJ, Grasset E, Laboisse C (1989) Functional VIP receptors in the human mucus-secreting colonic epithelial cell line CL.16E. Am J Physiol 256(3 Pt 1):G443–G450
Liao T, Wang L, Halm ST, Lu L, Fyffe RE, Halm DR (2005) K+ channel KVLQT1 located in the basolateral membrane of distal colonic epithelium is not essential for activating Cl- secretion. Am J Physiol Cell Physiol 289(3):C564–C575
Linden SK, Driessen KM, McGuckin MA (2007) Improved in vitro model systems for gastrointestinal infection by choice of cell line, pH, microaerobic conditions, and optimization of culture conditions. Helicobacter 12(4):341–353. doi:10.1111/j.1523-5378.2007.00509.x
Mall M, Bleich M, Schurlein M, Kuhr J, Seydewitz HH, Brandis M, Greger R, Kunzelmann K (1998) Cholinergic ion secretion in human colon requires coactivation by cAMP. Am J Physiol 275(6 Pt 1):G1274–G1281
Mall M, Wissner A, Seydewitz HH, Kuehr J, Brandis M, Greger R, Kunzelmann K (2000) Defective cholinergic Cl(−) secretion and detection of K(+) secretion in rectal biopsies from cystic fibrosis patients. Am J Physiol Gastrointest Liver Physiol 278(4):G617–G624
Matthews JB (2002) Molecular regulation of Na+−K+−2Cl- cotransporter (NKCC1) and epithelial chloride secretion. World J Surg 26(7):826–830
McCormick DA, Horton LW, Mee AS (1990) Mucin depletion in inflammatory bowel disease. J Clin Pathol 43(2):143–146
Muchekehu RW, Quinton PM (2010) A new role for bicarbonate secretion in cervico-uterine mucus release. J Physiol 588(13):2329–2342. doi:10.1113/jphysiol.2010.187237
Navabi N, McGuckin MA, Linden SK (2013) Gastrointestinal cell lines form polarized epithelia with an adherent mucus layer when cultured in semi-wet interfaces with mechanical stimulation. PLoS One 8(7):e68761. doi:10.1371/journal.pone.0068761
Neutra MR, O’Malley LJ, Specian RD (1982) Regulation of intestinal goblet cell secretion. II. A survey of potential secretagogues. Am J Physiol 242(4):G380–G387
Osbak PS, Bindslev N, Poulsen SS, Kaltoft N, Tilotta MC, Hansen MB (2007) Colonic epithelial ion transport is not affected in patients with diverticulosis. BMC Gastroenterol 7:37. doi:10.1186/1471-230X-7-37
Ostedgaard LS, Rogers CS, Dong Q, Randak CO, Vermeer DW, Rokhlina T, Karp PH, Welsh MJ (2007) Processing and function of CFTR-DeltaF508 are species-dependent. Proc Natl Acad Sci U S A 104(39):15370–15375. doi:10.1073/pnas.0706974104
Schulzke JD, Fromm M, Hegel U (1986) Epithelial and subepithelial resistance of rat large intestine: segmental differences, effect of stripping, time course, and action of aldosterone. Pflugers Arch 407(6):632–637
Specian RD, Neutra MR (1980) Mechanism of rapid mucus secretion in goblet cells stimulated by acetylcholine. J Cell Biol 85(3):626–640
Tabcharani JA, Harris RA, Boucher A, Eng JW, Hanrahan JW (1994) Basolateral K channel activated by carbachol in the epithelial cell line T84. J Membr Biol 142(2):241–254
Tousson A, Fuller CM, Benos DJ (1996) Apical recruitment of CFTR in T-84 cells is dependent on cAMP and microtubules but not Ca2+ or microfilaments. J Cell Sci 109(Pt 6):1325–1334
TranVan Nhieu G, Clair C, Grompone G, Sansonetti P (2004) Calcium signalling during cell interactions with bacterial pathogens. Biol Cell 96(1):93–101. doi:10.1016/j.biolcel.2003.10.006
Van der Sluis M, De Koning BA, De Bruijn AC, Velcich A, Meijerink JP, Van Goudoever JB, Buller HA, Dekker J, Van Seuningen I, Renes IB, Einerhand AW (2006) Muc2-deficient mice spontaneously develop colitis, indicating that MUC2 is critical for colonic protection. Gastroenterology 131(1):117–129. doi:10.1053/j.gastro.2006.04.020
van Doorninck JH, French PJ, Verbeek E, Peters RH, Morreau H, Bijman J, Scholte BJ (1995) A mouse model for the cystic fibrosis delta F508 mutation. EMBO J 14(18):4403–4411
Velcich A, Yang W, Heyer J, Fragale A, Nicholas C, Viani S, Kucherlapati R, Lipkin M, Yang K, Augenlicht L (2002) Colorectal cancer in mice genetically deficient in the mucin Muc2. Science 295(5560):1726–1729
Weber WM, Cuppens H, Cassiman JJ, Clauss W, Van Driessche W (1999) Capacitance measurements reveal different pathways for the activation of CFTR. Pflugers Arch 438(4):561–569
Wilke M, Buijs-Offerman RM, Aarbiou J, Colledge WH, Sheppard DN, Touqui L, Bot A, Jorna H, de Jonge HR, Scholte BJ (2011) Mouse models of cystic fibrosis: phenotypic analysis and research applications. J Cyst Fibros 10(2):S152–S171. doi:10.1016/S1569-1993(11)60020-9
Yu K, Lujan R, Marmorstein A, Gabriel S, Hartzell HC (2010) Bestrophin-2 mediates bicarbonate transport by goblet cells in mouse colon. J Clin Invest 120(5):1722–1735. doi:10.1172/JCI41129
Acknowledgments
This work was supported by the Swedish Research Council (nos. 7461, 8288, 21027, 21372, 20693, 2195501, 2009-2420, 521-2011-2370 and 342-2004-4434), The Knut and Alice Wallenberg Foundation, IngaBritt and Arne Lundberg Foundation, Sahlgren’s University Hospital (LUA-ALF), Wilhelm and Martina Lundgren’s Foundation, Åke Wibergs Foundation, Torsten and Ragnar Söderbergs Foundations, The Swedish Foundation for Strategic Research-The Mucosal Immunobiology and Vaccine Center (MIVAC) and the Mucus-Bacteria-Colitis Center (MBC) of the Innate Immunity Program. We acknowledge Dr. Anna Velcich for the Muc2−/− animals and Maria Sapnara for the technical assistance.
Ethical standards
All experiments were performed according to the ethical guidelines of the University of Gothenburg.
Conflict of interest
The authors declare that they have no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gustafsson, J.K., Lindén, S.K., Alwan, A.H. et al. Carbachol-induced colonic mucus formation requires transport via NKCC1, K+ channels and CFTR. Pflugers Arch - Eur J Physiol 467, 1403–1415 (2015). https://doi.org/10.1007/s00424-014-1595-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00424-014-1595-y