Abstract
Purpose
The aim of this pilot study was to investigate the effects of a preoperative immune-modulating diet (IMD) before thoracoscopic esophagectomy for patients with esophageal cancer.
Methods
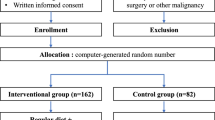
Thirty patients who were diagnosed with resectable esophageal cancer were assigned to two groups: the IMD (n = 15) and the standard liquid diet (SLD; n = 15) groups. We evaluated postoperative parameters, such as the incidence of complications and the postoperative levels of cytokines as the primary endpoints. The secondary endpoint was the length of hospital stay.
Results
The peak levels of interleukin (IL)-6 and tumor necrosis factor (TNF)-α appeared on postoperative day (POD) 1 and POD 2 in the IMD and SLD group, respectively. The peak level of C-reactive protein (CRP) appeared on POD 3 in both groups (IMD 9.9 mg/dL, SLD 15.2 mg/dL).
Overall, TNF-α levels in the IMD group were lower than those in the SLD group (P = 0.033). Furthermore, IL-6 (P = 0.182) and CRP (P = 0.191) levels, and the incidence of postoperative pneumonia (7.1 vs. 26.7%; P = 0.330) tended to be lower in the IMD group than in the SLD group.
Conclusions
Preoperative IMD suppressed the elevation of the TNF-α levels after thoracoscopic esophagectomy in patients with esophageal cancer, although no different tendency was detected in terms of IL-6, CRP or postoperative complications.






Similar content being viewed by others
References
Morita M, Nakanoko T, Fujinaka Y, Kubo N, Yamashita N, Yoshinaga K, Saeki H, Emi Y, Kakeji Y, Shirabe K, Maehara Y (2011) In-hospital mortality after a surgical resection for esophageal cancer: analyses of the associated factors and historical changes. Ann Surg Oncol 18:1757–1765
Takeuchi H, Miyata H, Gotoh M, Kitagawa Y, Baba H, Kimura W, Tomita N, Nakagoe T, Shimada M, Sugihara K, Mori M (2014) A risk model for esophagectomy using data of 5354 patients included in a Japanese nationwide web-based database. Ann Surg 260:259–266
Findlay JM, Gillies RS, Millo J, Sgromo B, Marshall RE, Maynard ND (2014) Enhanced recovery for esophagectomy: a systematic review and evidence-based guidelines. Ann Surg 259:413–431
Weimann A, Braga M, Harsanyi L, Laviano A, Ljungqvist O, Soeters P, DGEM (German Society for Nutritional Medicine), Jauch KW, Kemen M, Hiesmayr JM, Horbach T, Kuse ER, Vestweber KH, ESPEN (European Society for Parenteral and Enteral Nutrition) (2006) ESPEN guidelines on enteral nutrition: surgery including organ transplantation. Clin Nutr 25:224–244
ASPEN (2002) Board of Directors and the Clinical Guidelines Task Force Guidelines for the use of parenteral and enteral nutrition in adult and pediatric patients. JPEN J Parenter Enteral Nutr 26 (1 Suppl):1SA-138SA
Takayanagi T, Sasaki H, Kawashima A, Mizuochi Y, Hirate H, Sugiura T, Azami T, Asai K, Sobue K (2011) A new enteral diet, MHN-02, which contains abundant antioxidants and whey peptide, protects against carbon tetrachloride-induced hepatitis. JPEN J Parenter Enteral Nutr 35:516–522
Nakamura K, Ogawa S, Dairiki K, Fukatsu K, Sasaki H, Kaneko T, Yamaji T (2011) A new immune-modulating diet enriched with whey-hydrolyzed peptide, fermented milk, and isomaltulose attenuates gut ischemia-reperfusion injury in mice. Clin Nutr 30:513–516
Kume H, Okazaki K, Yamaji T, Sasaki H (2012) A newly designed enteral formula containing whey peptides and fermented milk product protects mice against concanavalin A-induced hepatitis by suppressing overproduction of inflammatory cytokines. Clin Nutr 31:283–289
Kaido T, Mori A, Ogura Y, Hata K, Yoshizawa A, Iida T, Yagi S, Uemoto S (2010) Impact of enteral nutrition using a new immune-modulating diet after liver transplantation. Hepato-Gastroenterology 57:1522–1525
Marshall K (2004) Therapeutic applications of whey protein. Altern Med Rev 9:136–156
Arakawa Y, Shimada M, Utsunomiya T, Imura S, Morine Y, Ikemoto T, Hanaoka J (2014) Effects of a whey peptide-based enteral formula diet on liver dysfunction following living donor liver transplantation. Surg Today 44:44–49
Fujiwara T, Naomoto Y, Motoki T, Shigemitsu K, Shirakawa Y, Yamatsuji T et al (2007) Effects of a novel palatinose based enteral formula (MHN-01) carbohydrate-adjusted fluid diet in improving the metabolism of carbohydrates and lipids in patients with esophageal cancer complicated by diabetes mellitus. J Surg Res 138:231–240
Egi M, Toda Y, Katayama H, Yokoyama M, Morita K, Arai H, Yamatsuji T, Bailey M, Naomoto Y (2010) Safer glycemic control using isomaltulose-based enteral formula: a pilot randomized crossover trial. J Crit Care 25:90–96
Kitagawa H, Namikawa T, Munekage M, Fujisawa K, Munekgae E, Kobayashi M, Hanazaki K (2016) Outcomes of thoracoscopic esophagectomy in prone position with laparoscopic gastric mobilization for esophageal cancer. Langenbeck's Arch Surg 401:699–705
Kitagawa H, Akimori T, Okabayashi T, Namikawa T, Sugimoto T, Kobayashi M, Hanazaki K (2009) Total laparoscopic gastric mobilization for esophagectomy. Langenbeck's Arch Surg 394:617–621
Kitagawa H, Namikawa T, Munekage M, Akimori T, Kobayashi M, Hanazaki K (2015) Visualization of the stomach's arterial networks during esophageal surgery using the HyperEye medical system. Anticancer Res 35:6201–6205
Kitagawa H, Yatabe T, Namikawa T, Munekage M, Hanazaki K (2016) Postoperative closed-loop glycemic control using an artificial pancreas in patients after esophagectomy. Anticancer Res 36:4063–4067
Wray CJ, Mammen JM, Hasselgren PO (2002) Catabolic response to stress and potential benefits of nutrition support. Nutrition 18:971–977
Tsujimoto OS, Takahata R, Hiraki S, Yaguchi Y, Kumano I, Matsumoto Y, Yoshida K, Aiko S, Ichikura T, Yamamoto J, Hase K (2012) Systemic inflammatory response syndrome as a predictor of anastomotic leakage after esophagectomy. Surg Today 42:141–146
Sczepanik AM, Scislo L, Scully T, Walewska E, Siedlar M, Kolodziejczyk P, Lenart M, Rutkowska M, Galas A, Czupryna A, Kulig J (2011) IL-6 serum levels predict postoperative morbidity in gastric cancer patients. Gastric Cancer 14:266–273
Rettig TC, Verwijmeren L, Dijkstra IM, Boerma D, van de Garde EM, Noordzij PG (2016) Postoperative interleukin-6 level and early detection of complications after elective major abdominal surgery. Ann Surg 263:1207–1212
Dimopoulou I, Armaganidis A, Douka E, Mavrou I, Augustatou C, Kopterides P, Lyberopoulos P, Tzanela M, Orfanos SE, Pelekanou E, Kostopanagiotou G, Macheras A, Giamarellos-Bourboulis EJ (2007) Tumour necrosis factor-alpha (TNF alpha) and interleukin-10 are crucial mediators in post-operative systemic inflammatory response and determine the occurrence of complications after major abdominal surgery. Cytokine 37:55–61
Mezzasoma L, Cagini L, Antognelli C, Puma F, Pacifico E, Talesa VN (2013) TNF-α regulates natriuretic peptides and aquaporins in human bronchial epithelial cells BEAS-2B. Mediat Inflamm 2013:159349
Mizock BA (2001) Alterations in fuel metabolism in critical illness: hyperglycaemia. Best Pract Res Clin Endocrinol Metab 15:533–551
Shimada H, Okazumi S, Maysubara H, Nabeya Y, Shiratori T, Hayashi H, Shuto K, Akutsu Y, Ochiai T (2007) Is the surgical stress associated with worse survival in patients with esophageal cancer?—Analysis of colon substitution for 37 patients with remnant stomach. Hepato-Gastroenterology 54:791–795
Goh SL, De Silva RP, Dhital K, Gett RM (2015) Is low serum albumin associated with postoperative complications in patients undergoing oesophagectomy for oesophageal malignancies? Interact Cardiovasc Thorac Surg 20:107–113
Beale RJ, Bryg DJ, Bihari DJ (1999) Immunonutrition in the critically ill: a systematic review of clinical outcome. Crit Care Med 27:2799–2805
Heys SD, Walker LG, Smith I, Eremin O (1999) Enteral nutritional supplementation with key nutrients in patients with critical illness and cancer: a meta-analysis of randomized controlled clinical trials. Ann Surg 229:467–477
Braga M, Gianotti L, Radaelli G, Vignali A, Mari G, Gentilini O, Di Carlo V (1999) Perioperative immunonutrition in patients undergoing cancer surgery: results of a randomized double-blind phase 3 trial. Arch Surg 134:428–433
Gianotti L, Braga M, Nespoli L, Radaelli G, Beneduce A, Di Carlo V (2002) A randomized controlled trial of preoperative oral supplementation with a specialized diet in patients with gastrointestinal cancer. Gastroenterology 122:1763–1770
Takeuch H, Ikeuchi S, Kawaguchi Y, Kitagawa Y, Isobe Y, Kubochi K, Kitajima M, Matsumoto S (2007) Clinical significance of perioperative immunonutrition for patients with esophageal cancer. World J Surg 31:2160–2167
Sakurai Y, Masui T, Yoshida I, Tonomura S, Shoji M, Nakamura Y, Isogaki J, Uyama I, Komori Y, Ochiai M (2007) Randomized clinical trial of the effects of perioperative use of immune-enhancing enteral formula on metabolic and immunological status in patients undergoing esophagectomy. World J Surg 31:2150–2157
Rice TW, Wheeler AP, Thompson BT, deBoisblanc BP, Steingrub J, Rock P, NIH NHLBI Acute Respiratory Distress Syndrome Network of Investigators (2011) Enteral omega-3 fatty acid, gamma-linolenic acid, and antioxidant supplementation in acute lung injury. JAMA 306:1574–1581
Sultan J, Griffin SM, Di Franco F, Kirby JA, Shenton BK, Seal CJ, Davis P, Viswanath YK, Preston SR, Hayes N (2012) Randomized clinical trial of omega-3 fatty acid-supplemented enteral nutrition versus standard enteral nutrition in patients undergoing oesophagogastric cancer surgery. Br J Surg 99:346–355
Fujitani K, Tsujinaka T, Fujita J, Miyashiro I, Imamura H, Kimura Y, Kobayashi K, Kurokawa Y, Shimokawa T, Furukawa H, Osaka Gastrointestinal Cancer Chemotherapy Study Group (2012) Prospective randomized trial of preoperative enteral immunonutrition followed by elective total gastrectomy for gastric cancer. Br J Surg 99:621–629
Lobo DN, Williams RN, Welch NT, Aloysius MM, Nunes QM, Padmanabhan J, Crowe JR, Iftikhar SY, Parsons SL, Neal KR, Allison SP, Rowlands BJ (2006) Early postoperative jejunostomy feeding with an immune modulating diet in patients undergoing resectional surgery for upper gastrointestinal cancer: a prospective, randomized, controlled, double-blind study. Clin Nutr 25:716–726
Klek S, Kulig J, Sierzega M, Szybinski P, Szczepanek K, Kubisz A, Kowalczyk T, Gach T, Pach R, Szczepanik AM (2008) The impact of immunostimulating nutrition on infectious complications after upper gastrointestinal surgery: a prospective, randomized, clinical trial. Ann Surg 248:212–220
Klek S, Kulig J, Sierzega M, Szczepanek K, Szybiński P, Scislo L, Walewska E, Kubisz A, Szczepanik AM (2008) Standard and immunomodulating enteral nutrition in patients after extended gastro-intestinal surgery: a prospective, randomized, controlled clinical trial. Clin Nutr 27:504–512
Kaya SO, Akcam TI, Ceylan KC, Samancılar O, Ozturk O, Usluer O (2016) Is preoperative protein-rich nutrition effective on postoperative outcome in non-small cell lung cancer surgery? A prospective randomized study. J Cardiothorac Surg 11:14
Takesue T, Takeuchi H, Ogura M, Fukuda K, Nakamura R, Takahashi T, Wada N, Kawakubo H, Kitagawa Y (2015) A prospective randomized trial of enteral nutrition after thoracoscopic esophagectomy for esophageal cancer. Ann Surg Oncol 22:802–809
Ata A, Lee J, Bestle SL, Desemone J, Stain SC (2010) Postoperative hyperglycemia and surgical site infection in general surgery patients. Arch Surg 145:858–864
NICE-SUGAR Study Investigators (2009) Intensive versus conventional glucose control in critically ill patients. N Engl J Med 360:1283–1297
Casaer MP, Wilmer A, Hermans G, Wouters PJ, Mesotten D, Van den Berghe G (2013) Role of disease and macronutrient dose in the randomized controlled EPaNIC trial: a post hoc analysis. Am J Respir Crit Care Med 187:247–255
Singer P, Anbar R, Cohen J, Shapiro H, Shalita-Chesner M, Lev S, Grozovski E, Theilla M, Frishman S, Madar Z (2011) The tight calorie control study (TICACOS): a prospective, randomized, controlled pilot study of nutritional support in critically ill patients. Intensive Care Med 37:601–609
Heidegger CP, Berger MM, Graf S, Zingg W, Darmon P, Costanza MC, Thibault R, Pichard C (2013) Optimization of energy provision with supplemental parenteral nutrition in critically ill patients: a randomized controlled clinical trial. Lancet 381:385–393
Hanazaki K, Kitagawa H, Yatabe T, Munekage M, Dabanaka K, Takezaki Y, Tsukamoto Y, Asano T, Kinoshita Y, Namikawa T (2014) Perioperative intensive insulin therapy using an artificial endocrine pancreas with closed-loop glycemic control system: the effect of no hypoglycemia. Am J Surg 207:935–941
Egi M, Bellomo R, Stachowski E, French CJ, Hart G (2006) Variability of blood glucose concentration and short-term mortality in critically ill patients. Anesthesiology 105:244–252
Acknowledgements
Authors’ contributions
H. K and T. N contributed to the study conception and design. H. K, T. Y, and M. M contributed to the acquisition of the data. H. K and F. Y did the analysis and interpretation of the data. H. K contributed to the drafting of the manuscript. The critical revision of the manuscript was done by T. N, M. K, and K. H.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Preoperative MHN-02 and MEIBARANCE® were offered by Meiji Dairies Co., Tokyo, Japan.
Ethical board approval
The study protocol was approved by the Institutional Review Board of Kochi Medical School, registered to UMIN 000014321, and was performed in accordance with the ethics standards laid down in the 1964 Declaration of Helsinki and its later amendments.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Kitagawa, H., Namikawa, T., Yatabe, T. et al. Effects of a preoperative immune-modulating diet in patients with esophageal cancer: a prospective parallel group randomized study. Langenbecks Arch Surg 402, 531–538 (2017). https://doi.org/10.1007/s00423-016-1538-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00423-016-1538-5