Abstract
Purpose
This study investigated the effects of acute hyperthermia and heat acclimation (HA) on maximal and rapid voluntary torque production, and their neuromuscular determinants.
Methods
Ten participants completed 10 days of isothermic HA (50 °C, 50% rh) and had their knee-extensor neuromuscular function assessed in normothermic and hyperthermic conditions, pre-, after 5 and after 10 days of HA. Electrically evoked twitch and octet (300 Hz) contractions were delivered at rest. Maximum voluntary torque (MVT), surface electromyography (EMG) normalised to maximal M-wave, and voluntary activation (VA) were assessed during brief maximal isometric voluntary contractions. Rate of torque development (RTD) and normalised EMG were measured during rapid voluntary contractions.
Results
Acute hyperthermia reduced neural drive (EMG at MVT and during rapid voluntary contractions; P < 0.05), increased evoked torques (P < 0.05), and shortened contraction and relaxation rates (P < 0.05). HA lowered resting rectal temperature and heart rate after 10 days (P < 0.05), and increased sweating rate after 5 and 10 days (P < 0.05), no differences were observed between 5 and 10 days. The hyperthermia-induced reduction in twitch half-relaxation was attenuated after 5 and 10 days of HA, but there were no other effects on neuromuscular function either in normothermic or hyperthermic conditions.
Conclusion
HA-induced favourable adaptations to the heat after 5 and 10 days of exposure, but there was no measurable benefit on voluntary neuromuscular function in normothermic or hyperthermic conditions. HA did reduce the hyperthermic-induced reduction in twitch half-relaxation time, which may benefit twitch force summation and thus help preserve voluntary torque in hot environmental conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Hot environmental conditions impair exercise performance by raising thermal and physiological strain (Ely et al. 2007; Mohr et al. 2012; Morante and Brotherhood 2008; Périard et al. 2014a; Racinais et al. 2015). Repeated exposure to heat stress (heat acclimation (HA)) can lessen this strain (Guy et al. 2015; Chalmers et al. 2014; Tyler et al. 2016), by inducing beneficial adaptations to the heat (Taylor 2014; Sawka et al. 2011). These adaptations include a lower resting core temperature and heart rate (HR) and an increased sweat rate (Sawka et al. 2011). Short-term HA protocols (5 daily exposures) are practical and effective at inducing some of these changes (Garrett et al. 2011; Chalmers et al. 2014); however, ≥ 10 days are required for more complete adaptation (Sawka et al. 2011; Guy et al. 2015; Tyler et al. 2016). The thermoregulatory and cardiovascular responses to HA are well documented (Périard et al. 2015); nevertheless, comparatively little is known about the effect that HA might have on the neuromuscular system, or the time course of those effects. Knowledge of the shortest amount of time to see a benefit to neuromuscular function from HA may be of practical use.
Exercise in hot ambient conditions which leads to a rise in core temperature, can cause reductions in voluntary force, due to reduced neural drive to the muscle (Nybo and Nielsen 2001). A reduction in neural drive resulting in a reduction in voluntary force is also observed during passive heating models (without exercise) (Todd et al. 2005; Morrison et al. 2004; Thomas et al. 2006; Gordon et al. 2021). If the hyperthermia-induced reduction in neural drive could be attenuated, the consequences may be a preservation in voluntary force. Other physiological and thermoregulatory systems have been shown to adapt to the heat, improving exercise performance and capacity (Périard et al. 2015); it therefore seems plausible that neural drive may also adapt following HA. There are data to show 11-day passive HA has a protective effect on voluntary activation and torque production during sustained voluntary contractions in hot conditions (Racinais et al. 2017a), suggesting supraspinal adaptions to HA. Conversely, HA over 28 days using exercise-induced hyperthermia (1 h per day) has shown no protective effect on cognitive performance and fine motor tasks in the heat (Piil et al. 2019). Impairments to cognitive performance during heat stress are distinct to neuromuscular changes (Gaoua et al. 2011); therefore, further investigation on the influence of HA on neural drive is warranted.
Few studies have investigated the influence of HA on neuromuscular function, and their findings have been mixed. Following 5 days of active (exercise) HA, Wingfield et al. (2016) reported no change (high intensity active HA) or a decrease (low intensity active HA) in maximal voluntary torque (MVT) and no change in neural drive. In contrast, Osborne et al. (2021) observed increased MVT without an increase in neural drive, following 5 days of active heat acclimation, suggesting peripheral adaptation to the muscle. However, the active HA protocols used by Wingfield et al. (2016) and Osborne et al. (2021) make it difficult to isolate neuromuscular adaptations to HA from those adaptations to the active component of the HA. Removing the confounding influence of exercise and utilising passive heating, one study has shown benefits of HA on neural drive and MVT (Racinais et al. 2017a), and another none (Brazaitis and Skurvydas 2010). The discrepancy between the findings of Racinais et al. (2017a) and Brazaitis and Skurvydas (2010) is not clear, but may be due to differences in the adaptation stimulus (time at an elevated core temperature; Taylor 2014). In each study, HA was deemed successful [evidenced by reductions in rectal temperature (Tre)], but the adaptation stimulus was lower in Brazaitis and Skurvydas (2010) (~ 20 min Tre ≥ 38.5 °C per session for 7 sessions) compared to Racinais et al. (2017a) (66 ± 8 min Tre ≥ 38.5 °C in 9/11 sessions), which might explain why only Racinais et al. (2017a) observed benefits of HA on neuromuscular function. Thus, a large thermal impulse may be required to induce neuromuscular adaptations, specifically to neural drive generation and transmission to the peripheral nervous system. Providing a large thermal stimulus (daily heating up to a Tre of 39 °C) may help to elucidate the effect of HA on neuromuscular function.
MVT is typically used to assess the capacity of the neuromuscular system. The relevance of MVT is reduced in functional situations, where time to develop torque is limited because MVT takes > 125 ms to achieve when contracting from rest (Tillin et al. 2012, 2018). An alternative assessment to MVT is measuring voluntary RTD, which quantifies the ability to rapidly produce torque throughout the rising torque–time curve from rest (Folland et al. 2014). The determinants of voluntary RTD derive from both central and peripheral mechanisms; however, the relative contributions of these pathways differ throughout the rising torque–time curve (Folland et al. 2014). Moreover, the effects of hyperthermia on voluntary RTD are less well known compared to MVT, with recent data showing there are distinct responses between the two variables in the heat (Gordon et al. 2021). Hyperthermia reduces neural drive at the plateau of an MVC (where MVT is measured). Gordon et al. (2021) also observed reductions in neural drive at the onset of rapid voluntary contractions. Interestingly, whilst these reductions in neural drive translated to reduced MVT, voluntary RTD remained unaffected by hyperthermia, despite neural drive at the onset of a rapid contraction being an important determinant of RTD (Folland et al. 2014; Del Vecchio et al. 2019). RTD appears to be preserved when hyperthermic due to faster intrinsic contractile properties (Gordon et al. 2021). Faster intrinsic contractile properties occur when muscles are warmer (de Ruiter et al. 1999; de Ruiter and de Haan 2000; Dewhurst et al. 2005). Theoretically, if a regime of HA with sufficient thermal stimulus can offer a protective effect on neural drive, in conjunction with the hyperthermia-induced faster intrinsic contractile properties, voluntary RTD may increase.
Independent of hyperthermia, increases in MVT have been observed in hot and cool conditions following 11 days of passive HA (Racinais et al. 2017b), without modifications to neural drive, suggesting HA may induce adaptations in baseline contractile properties. Evidence for this has been provided in rats by Kodesh and Horowitz (2010) who found increases in peak tetanic force after 30 days’ heat exposure. The same study (Kodesh and Horowitz 2010) also found decreases in the rate of relaxation after HA, suggesting HA modifies the re-uptake of Ca2+ by the sarcoplasmic reticulum. The slower relaxation rate following HA may have increased the tetanic force by enhancing twitch force summation, and a similar effect in humans may result in increased MVT. However, the effects of HA on the contractile properties and, in turn, voluntary torque production in humans remain unclear.
The aim of the current study was to investigate the effects of 5 and 10 days of HA on MVT, voluntary RTD and their neuromuscular determinants measured in normothermic and hyperthermic conditions. It was hypothesised that: (i) a regime of HA would attenuate the decline in neural drive caused by hyperthermia, attenuating any reduction in MVT and enabling an increase in voluntary RTD due to faster contractile properties; (ii) independent of hyperthermia, HA would reduce the relaxation time of muscle, benefiting evoked and voluntary torque output in normothermic and hyperthermic conditions; (iii) neuromuscular adaptations from HA would be more observable after 10, rather than 5 days of heat exposure, giving a time course of adaptation to the neuromuscular system following short- and medium-term HA.
Methods
Participants
Ten (5 females and 5 males) healthy, physically active individuals participated in the study. Mean age, body mass and stature were 35.6 ± 7.2 years; 70.7 ± 9.7 kg, and 175.7 ± 8.6 cm, respectively. An a priori power analysis was performed for sample size estimation (GPower 3.1) based on data from Gordon et al. (2021) (n = 9) comparing acute hyperthermia-induced decreases in EMGMVT at Tre 39.5 °C compared to ~ 37 °C, and a large effect (0.14) using \({\eta }_{p}^{2}\). With an α = 0.05 and β = 0.80, the projected sample size needed for the present study to assess differences between baseline and hyperthermia was approximately n = 10. In addition, a very large effect size (Cohen’s d = 1.2; n = 8) was estimated from similar HA research using a 5-day protocol (Osborne et al. 2021) for the change in EMG between control and HA pre-cycling, at two time points; pre- and post-HA intervention. All participants were informed of any risks and discomforts associated with the experiment before giving their written informed consent, in accordance with the latest version of the Declaration of Helsinki. Experimental procedures were approved by the Ethical Advisory Committee of the University of Roehampton (LSC 19/259). Participants were considered non-heat acclimated because they had not been exposed to ambient temperatures exceeding 25 °C for the 3 weeks prior to participation. Participants were allowed to maintain their normal training routine during the HA days (n = 6 were training to compete in the ultra-endurance running foot race, Marathon des Sables, n = 3 were club-level runners and n = 1 a club-level rower). Participants were instructed to refrain from any strenuous physical activity for 24 h prior to visiting the laboratory for the experimental trials, and from alcohol consumption for the duration of the study. Due to the scheduling requirements of the study, it was not possible to control for the variations in hormone levels associated with the menstrual cycle for female participants. The authors recognise that this may have caused some variability in Tre, contributing to some variability in neuromuscular function; however, ecological validity of the study was increased by not controlling for the menstrual cycle, as such control is not possible in real world/sporting scenarios. Furthermore, recent data suggest menstrual phase does not modulate whole body heat loss in hot conditions (Notley et al. 2019). Data were collected between March and May 2019 (mean outside temperature ~ 11 °C) at the University of Roehampton, London, in the UK.
Overview
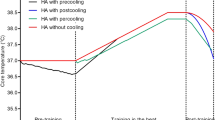
Participants visited the laboratory on 14 separate occasions, completing a familiarisation, three experimental trials, and 10 days of HA. The first experimental trial was completed 3–5 days after the familiarisation, with all remaining visits conducted on consecutive days (Fig. 1). All sessions were completed in a walk-in environmental chamber. The experimental trials and HA sessions were completed at the same time of day for each participant, and in the same ambient conditions (50 °C, 50% rh).
In the three experimental trials, participants completed two sets of the same neuromuscular protocol using their preferred leg as in the protocol of Gordon et al. (2021). The protocol comprised a series of involuntary and voluntary isometric contractions of the knee extensors. Set 1 was completed at a Tre of ~ 37 °C and set 2 at a Tre of ~ 39 °C.
The HA sessions employed an isothermic heat adaptation protocol to a target Tre of ~ 39 °C. There is no consensus on the optimal daily heat exposure, with a meta-analysis concluding research studies investigating HA have used session lengths with a mean duration of 105 ± 62 min (Tyler et al. 2016). To provide sufficient magnitude of thermal impulse (Taylor 2014) and maximum potential for thermal adaptation, we chose 90-min daily heat exposure up to a target Tre of 39 °C, which was adapted from previous research using a progressive protocol initially targeting a Tre of 38.5 °C, increasing to 39 °C (Gibson et al. 2015). After each session, participants were cooled in the laboratory (~ 21 °C) until Tre had returned to 38 °C.
Protocol
Experimental trials
Skin thermistors and EMG electrodes were attached before the participants donned an impermeable rain jacket and trouser combination and entered the environmental chamber. Participants were seated in the isometric strength testing chair ready to complete set 1. To standardise the Tre during set 1 across experimental trials, and account for the expected reduction in resting Tre after HA (Tyler et al. 2016), set 1 was completed at a Tre of 37.0 ± 0.3 °C. If Tre was < 36.7 °C, participants remained seated in the strength chair wearing the jacket and trousers passively heating until Tre was within ~ 0.3 °C of 37.0 °C, and then set 1 commenced. If Tre was greater than 37.3 °C before entering the heat chamber, participants were asked to remain quietly seated in the ambient temperatures of the laboratory (~ 21 °C), until Tre had decreased to within ~ 0.3 °C of 37.0 °C. After completing neuromuscular set 1, participants performed a fixed intensity exercise bout (~ 80 W) on a cycle ergometer, to facilitate heat storage without eliciting exercise-induced fatigue, until a Tre of 38 °C was attained. Participants were then passively heated in either a seated or supine position, before transferring back to the isometric strength chair to perform set 2. The time spent passively heating ranged between 0–45 min (Trial 1), 5–58 min (Trial 2) and 5–60 min (Trial 3). To account for the expected rise in Tre but achieve a mean target Tre of ~ 39 °C during the neuromuscular protocol, set 2 commenced at 38.9 ± 0.1 °C in all trials. If participants had been resting in a supine position, sufficient time was allowed for a gradual change in posture and subsequent transfer back to the isometric strength chair. The impermeable rain jacket was worn continuously throughout the experimental trial. The trousers were removed to perform the neuromuscular sets, but were otherwise worn continuously.
HA sessions
Participants were instrumented with skin thermistors before entering the environmental chamber, sitting quietly on a cycle ergometer for 2 min, and having their baseline thermoregulatory and perceptual measurements recorded. A similar bout of light physical activity to that completed in the experimental trials was performed on the cycle ergometer. Initial work was 100 W, and this was subsequently reduced by 7 W every 5 min to 72 W. Participants cycled at 72 W until Tre reached 38 °C, then transferred to a chair located adjacent to the ergometer, and sat until Tre reached 39 °C. Upon reaching Tre 39 °C, participants removed the jacket and trousers and spent the remainder of the session (HA sessions were 90 min in total) supine or seated according to their preference. Tre was maintained as close as possible to the target 39 °C by donning and doffing the jacket and trouser combination, as necessary, based on real-time Tre data. To improve participant thermal comfort and facilitate the completion of the HA sessions, an electric fan was available if participants requested it for facial fanning only. This option was only provided during the HA sessions. Three participants were unable to complete one HA session each, due to personal circumstances. Overall, 98% of HA sessions were completed.
Measurements
Torque
Seated in a custom-built strength testing chair (Maffiuletti et al. 2016), participants were securely fastened with a waist belt and shoulder straps with hip and knee angles fixed at 100° and 105°, respectively (180° defined full extension). An ankle strap, in series with a strain gauge load cell (FSB Universal Cell 1.5 kN, Force Logic, Reading, UK), was secured 4 cm proximal to the medial malleolus, with the load cell aligned perpendicular to the tibia during knee extension (Tillin et al. 2010). The force signal was amplified (× 375) and sampled at 2000 Hz via an analogue-to-digital converter (Mirco3 1401, CED, UK) and PC using Spike2 software (Spike 2 Version 8, CED, UK). A computer monitor, placed in view of the participant, provided real-time biofeedback. Off-line, the force signal was filtered using a fourth-order low-pass Butterworth filter with a 500 Hz cutoff frequency. To calculate knee extension torque, the weight of limb was corrected for and multiplied by the external moment arm (measured as the distance between the lateral knee joint space and the centre point of the ankle strap).
EMG
Following preparation of the skin (shaving, light abrasion, and cleaning using 70% ethanol) a bipolar silver–silver chloride gel-electrode configuration (2 cm diameter, and 2 cm inter-electrode distance; Dual Electrode, Noraxon, USA) was placed over the belly of the rectus femoris, vastus lateralis and vastus medialis. Electrode configurations were placed parallel to the presumed orientation of the muscle fibres at specific distances from the greater trochanter to the lateral knee joint space [44 ± 5% (rectus femoris), 71 ± 3% (vastus lateralis), 79 ± 3% (vastus medialis)]. The placement of electrodes was conducted by the same investigator in all trials and established during familiarisation. The position of each electrode was marked on the skin using permanent ink. Participants were instructed not to actively wash these marks off between trials, so electrodes could be placed in the same position at the beginning of each experimental trial. The EMG signal was amplified (× 500; 10–500 Hz bandwidth), transmitted wirelessly to a desktop receiver (TeleMYODTS, Noraxon, USA), and sampled (2000 Hz) in synchronisation with force via the same analogue-to-digital converter using Spike2 software. In off-line analysis, the EMG signals were band-pass-filtered between 6 and 500 Hz using a fourth-order Butterworth digital filter and time corrected (156 ms delay inherent in the TeleMyoDTS system) to align with the force signal.
Electrical stimulation
Electrical square-wave pulses (200 µs duration) delivered over the femoral nerve (DS7AH Constant Current Stimulator, Digitimer, UK) were used to evoke twitch (single pulse), doublet (two pulses at 100 Hz) and octet (eight pulses at 300 Hz) contractions. The anode (rubber electrode 10 × 7 cm, EMS Physio Ltd, UK) was secured by tape (Transpore, 3 M, UK) to the skin over the greater trochanter. The cathode stimulation probe (1 cm diameter tip; S1 Compex Motor PointPen, Digitimer, UK), which protruded 2 cm from the centre of a custom-built plastic base (4 × 3 cm), was placed over the femoral nerve in the femoral triangle. The greatest evoked peak twitch force in response to a submaximal current determined the precise placement of the cathode, where it was taped in place. The electrical current was then increased incrementally by 20 mA until there was a plateau in both twitch peak force and peak-to-peak M-wave amplitude at each EMG site. This current was increased by a further 20% (supramaximal) to ensure all stimulations were eliciting a maximal involuntary response, and this current (122 ± 22 mA) was used for all twitch, doublet, and octet contractions thereafter. The cathode position and supramaximal stimulation intensity were determined for each participant in the familiarisation session and then kept constant for the experimental trials. The cathode position was marked on the skin with permanent ink and maintained by participants to ensure accurate relocation between trials.
Thermoregulatory and perceptual responses
A rectal thermistor (REC-U-VL30, Grant Instruments, UK) was self-inserted ~ 10 cm past the anal sphincter to measure Tre. Wireless skin thermistors (iButton DS1922L; Maxim/Dallas Semiconductor Corp., USA) were applied to the skin with a transparent dressing and secured with surgical tape for the assessment of local skin temperature. Mean weighted skin temperature (\(\overline{T}\)sk) was calculated from four skin sites located on the right side of the body: below the suprasternal notch (chest), flexi carpi radialis (arms), gastrocnemius (legs), and rectus femoris (thighs) (Ramanathan 1964). HR was recorded with a heart rate monitor secured with a strap and worn by the participant in contact with the skin (F3, Polar Electro, UK, Ltd).
Whole body thermal sensation (TS) was rated using a nine-point scale from 0 (unbearably cold) to 8 (unbearably hot) with 4 as comfortable (neutral) (Young et al. 1987). Whole body thermal comfort (TC) was measured using a four-point scale from 1 (comfortable) to 4 (very uncomfortable) (Gagge et al. 1967). All thermoregulatory and perceptual measurements were recorded at 5 min intervals and at the start and end of each of the neuromuscular assessment protocols in the experimental trials.
Fluid loss
Participants consumed 500 ml of water 2 h before each visit to the laboratory. Pre-session hydration status was assessed from a mid-stream urine sample and euhydration was assumed if urine specific gravity was ≤ 1.020. Water (non-chilled) was provided ad libitum throughout and voluntary fluid consumption recorded. Participants were instructed to towel dry themselves, removing any residual sweat on the skin before recording nude body mass, pre- and post-sessions. After correcting for fluid intake and urine output, body mass changes were used to estimate sweat loss.
Neuromuscular set
The series of involuntary and voluntary contractions used in the neuromuscular set in the experimental trials are described below, in the order that they were performed.
Twitch and octet
Two electrically evoked twitch and octet contractions were delivered over the femoral nerve at rest, 20 s apart. The maximal M-wave (Mmax) was calculated as the average M-wave response (peak–peak amplitude of the EMG signal) from the two evoked twitches and used for EMG normalisation. Twitch and octet responses were analysed for: peak torque (PT); rate of torque development during the initial 50 ms from contraction onset (RTD0-50); peak rate of torque development (pRTD); time to peak torque (TPT); and half-relaxation time (½RT). Mean values were calculated for all dependant variables across the two twitch and two octet contractions.
Rapid voluntary contractions
Participants performed 10–15 rapid contractions (~ 1 s) and were instructed to push as “fast and hard” as possible (Tillin et al. 2010), emphasising the “fast” element of the contraction. For each contraction, participants were encouraged to exceed 80% of MVT, as quickly as possible, repeating the effort if this was not achieved, up to a maximum of 15 contractions. Any rapid contractions with discernible countermovement or pre-tension prior to force onset were also repeated. A short recovery (5–10 s) was given between contractions and participants were instructed to relax their leg as quickly as possible before the next effort, which only commenced once force had returned to a stable baseline. Baseline force was displayed on a sensitive scale on a computer monitor in front of the participants to provide biofeedback on the occurrence of any countermovement or pre-tension. The slope of the force–time curve (25 ms time-constant) was also displayed. The three rapid voluntary contractions with the highest pRTD and no discernible countermovement or pre-tension (change in baseline force of > 2 SD of the mean during the 200 ms prior to force onset) were used for analysis. Torque was measured at discrete time points 50 ms (T50), 100 ms (T100) and 150 ms (T150) from torque onset, and RTD measured over three sequential time epochs: 0–50 ms (RTD0-50), 50–100 ms (RTD50-100) and 100–150 ms (RTD100-150). The RMS of the signal at each EMG site was assessed over 0–50 ms (EMG0-50), 0–100 ms (EMG0-100), and 0–150 ms (EMG0-150) from EMG onset, normalised to Mmax at the same EMG site, and averaged across the three EMG sites to give a mean value for the quadriceps muscles. Dependant variables were mean averaged across the three rapid contractions selected for analysis. Torque and EMG signal onsets (voluntary and evoked) were identified using visual identification, which is considered the “gold standard” of signal onset determination compared to automated detection methods (Tillin et al. 2013), using the standardised protocol of Tillin et al. (2010). Mean values were averaged for the two twitch and octet contractions, respectively.
MVCs
Participants performed three MVCs (3–5 s), separated by 30 s rest and were instructed to push as “hard” as possible. At the plateau of the second MVC, two superimposed involuntary doublet stimuli were evoked 2 s apart, followed by a doublet contraction evoked at rest 2–5 s after the MVC. MVT was defined as the greatest voluntary (i.e. not due to superimposed doublet stimulation) torque recorded in any of the rapid contractions or MVCs. To assess neural drive at MVC plateau, the amplitudes of the superimposed doublets were used to determine voluntary activation (VA) using the following formula:
where Dsup represents the superimposed doublet amplitude and Dcon the potentiated doublet amplitude evoked at rest after the MVC. VA was calculated from one of the superimposed doublets (whichever was delivered at the greatest torque value) during the same MVC. Neural drive was also assessed from the RMS amplitude over a 500 ms epoch surrounding MVT (250 ms either side, without influence of artefact from electrical stimulation), normalised to Mmax, and the mean averaged across the three EMG sites to give a value for the whole quadriceps muscle (EMGMVT).
Statistical analyses
All data were assessed for, and met, parametric assumptions prior to analysis. Descriptive data are reported as mean ± SD for n = 10. It was not possible to obtain values past 100 ms during the explosive voluntary contractions for two participants. This was because they were unable to perform the rapid voluntary contractions for the minimum required 1 s duration in enough voluntary efforts, whilst hyperthermic, often beginning to relax the muscle by 150 ms. Therefore, for T150, RTD100–150, and EMG0-150 data are for n = 8. A Two-way repeated measures ANOVA was used to assess the influence of experimental trial (3 trials: pre- [Trial 1]; post-5 days [Trial 2]; and post-10 days [Trial 3]), at two different Tre (37 °C and 39 °C) on all physiological, perceptual, and neuromuscular dependant variables measured during the neuromuscular set. A one-way repeated measures ANOVA was used to assess responses within the HA sessions at HA 1, HA 5, and HA 10. Violations of sphericity were corrected using the Greenhouse–Geisser adjustment, when appropriate. Following a significant F value, pairwise differences were identified using stepwise Bonferroni-corrected paired T tests. Effect sizes for paired comparisons were calculated using Hedge’s g and interpreted as small (0.2), medium (0.5) or large (0.8) (Cohen 1988). The alpha level was set at P < 0.05. Statistical analysis was completed using SPSS version 26 (SPSS Inc., Chicago, IL).
Results
Responses within the HA sessions
Resting Tre (P = 0.010), resting HR (P = 0.003), sessional sweat rate (P = 0.002), mean session Tre (P = 0.003) and mean session HR (P = 0.043) all demonstrated overall improvements as the number of HA sessions increased. Post hoc analysis revealed midway through at HA 5, sweat rate increased (P = 0.024; g = 0.5), while mean session Tre (P = 0.015; g = 0.8) and mean session HR (P = 0.039; g = 0.6) reduced compared to HA 1. Resting Tre (P = 0.148; g = 0.6) and resting HR (P = 0.222; g = 0.6) were not statistically different at HA 5 from HA 1. There were no differences observed for time spent ≥ Tre 38.5 °C (P = 0.404) or ≥ Tre 39 °C (P = 0.795). HA 10 compared to HA 1 showed resting Tre (P = 0.012; g = 1.0), resting HR (P = 0.026; g = 1.2), and mean session Tre (P = 0.026; g = 0.9) decreased, while sweat rate increased (P = 0.021; g = 0.6), providing evidence of successful heat acclimation. Data are presented in Table 1.
Physiological and perceptual strain during the neuromuscular set
Participants were at the desired Tre during all the neuromuscular sets, with no main effects of trial (P = 0.353) or interaction (P = 0.629), but only an effect of Tre (P < 0.001), the latter imposed by study design. There was no main effect of trial on \(\overline{T}\)sk (P = 0.267), but there was an effect of Tre (P < 0.001), in addition to an interaction effect (P = 0.004). Post hoc analysis revealed at Tre 37 °C, \(\overline{T}\)sk was greater in Trial 3 than Trial 1 (P = 0.028; g = 1.0), but it was not statistically different in other between trial comparisons (P ≥ 0.194; g = 0.1–0.7). HR was affected by trial (P = 0.010), Tre (P < 0.001) and there was an interaction effect (P = 0.011). Post hoc analysis showed at Tre 37 °C, HR had increased in Trial 3 compared to Trial 1 (P = 0.019; g = 0.9), likely because some participants had to spend time in the heat passively warming to attain Tre ~ 37 °C before commencing neuromuscular set 1. While at Tre 39 °C HR had decreased in Trial 3 compared to Trial 2 (P = 0.036; g = 0.4). Other between trial comparisons were not statistically different (P ≥ 0.143; g = 0.1–0.6). No main effects of trial (P ≥ 0.147) or interaction (P ≥ 0.062) were observed for TS and TC. However, participants did feel hotter and more uncomfortable at Tre 39 °C compared to 37 °C in all trials (main effect of Tre; P < 0.001). Data are presented in Table 2.
Voluntary torque and RTD
MVT was not affected by experimental trial (P = 0.928), Tre (P = 0.524) or interaction (P = 0.653) (Table 3). Rapid torque production (T50, T100, and T150; Fig. 2) and voluntary RTD (RTD0–50, RTD50–100, and RTD100–150; Fig. 3) were also all unaffected by trial (P ≥ 0.064), Tre (P ≥ 0.071), or interaction (P ≥ 0.493).
Rapid voluntary torque at; 50 ms (T50; A), 100 ms (T100; B) and 150 ms (T150; C) from contraction onset. Measurements were taken at two different rectal temperatures: 37 °C (grey bars and open circles) and 39 °C (open bars and open squares), and pre- (Trial 1), post-5 days (Trial 2) and post-10 days (Trial 3) of heat acclimation. Individual data points and group means presented for n = 10 at T50 and T100. Data are n = 8 for T150 because it was not possible to obtain torque values after 100 ms for two participants. For clarity, error bars have been omitted
Rate of torque development at sequential time epochs, 0–50 ms (RTD0–50; A), 50–100 ms (RTD50–100; B) and 100–150 ms (RTD100–150; C). Measurements were taken at two different rectal temperatures: 37 °C (grey bars and open circles) and 39 °C (open bars and open squares), and pre- (Trial 1), post-5 days (Trial 2) and post-10 days (Trial 3) of heat acclimation. Individual data points and group means presented for n = 10 at RTD0–50 and RTD50–100. Data are n = 8 for RTD100–150 because it was not possible to obtain RTD values after 100 ms for two participants. For clarity, error bars have been omitted
Neural drive
EMGMVT (Table 3) decreased with the rise in Tre (P = 0.001), but was not affected by experimental trial (P = 0.509), or an interaction effect (P = 0.564). Post hoc analysis revealed at Tre 39 °C EMGMVT had decreased compared to Tre 37 °C in all three trials (P ≤ 0. 021; g = 1.1–1.6). There were no main effects of HA (P = 0.146), Tre (P = 0.060), or interaction effect (P = 0.790) for VA (Table 3).
No main effect of trial (P = 0.816), Tre (P = 0.101), or interaction (P = 0.097) was observed for EMG0–50 (Fig. 4A). No effects of trial (P ≥ 0.467) or interaction (P ≥ 0.326) were observed for EMG0–100 (Fig. 4B) or EMG0–150 (Fig. 4C), but the rise in Tre did result in a decrease (P ≤ 0.016) in both these variables. Post hoc analysis revealed at Tre 39 °C EMG0–100 had decreased compared to Tre 37 °C in Trial 2 (P = 0.003; g = 0.9), but was not statistically different in the other trials (P ≥ 0.051; g = 0.8–1.4). EMG0–150 also decreased at Tre 39 °C compared to Tre 37 °C in Trial 2 (P = 0.020; g = 0.8), but was not statistically different in Trial 1 or 3 (P ≥ 0.055; g = 0.9–1.3).
Surface EMG RMS normalised to Mmax at 0–50 ms (EMG0–50; A), 0–100 ms (EMG0–100; B) and 0–150 ms (EMG0–150; C). Measurements were taken at two different rectal temperatures: 37 °C (grey bars and open circles) and 39 °C (open bars and open squares), and pre- (Trial 1), post-5 days (Trial 2) and post-10 days (Trial 3) of heat acclimation. Individual data points and group means presented for n = 10 at EMG0–50 and EMG0–100. Data are n = 8 for EMG0–150 because it was not possible to obtain EMG values after 100 ms for two participants. For clarity error bars have been omitted. Post hoc significant difference is denoted by: † (P < 0.05), †† (< 0.005), different from 37 °C
Intrinsic contractile properties
No main effect of experimental trial was observed on the twitch dependant variables (P ≥ 0. 116). There was an interaction effect for ½ RT (P = 0.032), but not for any other parameter (P ≥ 0. 155). There was a main effect of Tre, and the increase in Tre caused greater (PT, RTD0–50, pRTD) and faster (TPT and ½ RT) twitch responses (P ≤ 0.027). Post hoc analysis revealed at Tre 39 °C twitch PT had increased compared to Tre 37 °C in Trial 1 and 2 (P ≤ 0.037, g = 0.5), but was not statistically different in Trial 3 (P = 0.059; g = 0.3). Twitch RTD0–50 and pRTD were greater at Tre 39 °C in all trials (P ≤ 0.013; g = 0.4–0.7). Twitch TPT was faster at Tre 39 °C compared to Tre 37 °C in Trial 1 and Trial 2 (P ≤ 0.018; g = 1.5), but not in Trial 3 (P = 0.133; g = 0.9). Twitch ½ RT was also faster at Tre 39 °C in Trial 1 (P = 0.002; g = 1.3), but not statistically different in the other trials (P ≥ 0.098; g = 0.6). Data are presented in Table 4.
No main effect of experimental trial (P ≥ 0.059) or interaction effects (P ≥ 0. 062) were observed on the octet dependant variables. A main effect and rise in Tre caused greater (PT, RTD0–50, pRTD) and faster (TPT and ½ RT) octet responses (P ≤ 0.019). Post hoc analysis revealed at Tre 39 °C octet PT had increased compared to Tre 37 °C in trial 3 (P = 0.004, g = 0.2), but was not statistically different in the other trials (P ≥ 0.082; g = 0.1–0.2). Octet RTD0–50 and pRTD were greater at Tre 39 °C in all trials (P ≤ 0.002; g = 0.4–0.6), while octet TPT and ½ RT were faster at Tre 39 °C in all trials (P ≤ 0.044; g = 0.9–2.2). Data are presented in Table 4.
Discussion
This study investigated the effect of 5 and 10 days of isothermal HA on the neural and contractile mechanisms that determine rapid and maximal torque capacity, whilst normothermic and hyperthermic. Consistent with our previous observations (Gordon et al. 2021), acute hyperthermia (independent of HA) reduced neural drive at MVT and during rapid voluntary contractions. The reductions in neural drive did not reduce MVT or RTD, potentially due to warmer muscles increasing intrinsic contractile (twitch and octet) torques and the speed of torque development (Table 4). HA induced favourable physiological adaptations to the heat after 10 days of heat exposure, with successful manipulation of the “classic” markers of HA (e.g. reduction in resting Tre and HR and an increased sweat rate). However, there was no evidence that HA, independent of hyperthermia, improved any measured aspect of neuromuscular function, nor did HA attenuate the observed hyperthermia-induced reductions in neural drive. Nevertheless, there was an interaction effect on ½ RT, in which the reduction in ½ RT caused by hyperthermia in Trial 1 was not observable in Trial 2 or 3. Speculatively, this preservation of ½ RT when the muscle is hot, following HA, may have functional benefits for exercise in the heat, as the faster ½ RT typically observed with warmer muscles is thought to negatively affect twitch force summation, necessitating a greater neural drive to obtain the same absolute force (Todd et al. 2005; Périard et al. 2014b).
Effects of acute hyperthermia
During the experimental trial sessions, heat exposure increased all thermoregulatory, cardiovascular, and perceptual responses, while Tre was successfully clamped across the different trials at pre-, post-5 days, and post-10 days of HA (Table 2). Similarly, subjective perceptual ratings of the heat (TS) and comfort (TC) were not altered by HA, with participants feeling “very hot” and “uncomfortable” in all trials. Taken together, these data demonstrate that the neuromuscular set was performed under similar levels of actual and perceived thermal strain.
MVT (Table 3) was not affected by hyperthermia. Despite no change in MVT, neural drive did decrease with hyperthermia, evidenced by lower EMGMVT (main effect of Tre; P < 0.05; Table 3). The reduction in neural drive at MVC plateau with acute hyperthermia corroborates previous research from our laboratory which used the same neuromuscular assessment protocol (Gordon et al. 2021). However, our previous study (Gordon et al. 2021) found MVT to decrease concomitantly with neural drive due to acute hyperthermia. It is unclear why MVT did not decrease with hyperthermia in the current study, but it is possibly linked to the hyperthermia-induced increased torque capacity of the muscle, evidenced by the acute increases in twitch and octet PT (Table 4). Specifically, the increased torque capacity may have countered a reduction in neural drive, maintaining MVT. Furthermore, the training status of participants used in this study (endurance trained amateur athletes) and that of Gordon et al. (2021) (recreationally trained males) was different and data suggests that more aerobically trained individuals are better able to tolerate high heat stress, leading to smaller performance decrements in the heat (Cheung and McLellan 1998; Guy et al. 2015). Additionally, closer post hoc inspection of the individual data revealed a speculative effect of sex on neuromuscular function during acute hyperthermia. Specifically, in Trial 1, acute hyperthermia caused a 6.2% decline in MVT in the males (paired t test; P = 0.205; g = 0.6), but only a 1.1% decline in females (paired t test; P = 0.765; g = 0.1). Thus, males may be more susceptible to declines in MVT than females which if true, would reduce the chances of observing significant declines in MVT across a mixed-sex sample. Our previous study (Gordon et al. 2021) and that of others who observed reduced MVT with hyperthermia, have included only male participants (Morrison et al. 2004; Périard et al. 2014a), which might explain why their results contrast with ours. On the other hand, other studies have used mixed male and female cohorts and observed reduced MVT with hyperthermia (Todd et al. 2005; Thomas et al. 2006), but did not assess the effects of sex. The current study is under-powered for considering between-sex differences and so we recommend future research considers the effect of sex on neuromuscular responses to hyperthermia.
Similar to MVT, voluntary RTD was unaffected by acute hyperthermia, despite a reduction in neural drive evidenced by a main effect for decreased EMG0–100 (Fig. 4B) and EMG0–150 (Fig. 4C), which is consistent with what we observed in our previous study (Gordon et al. 2021). The preservation of rapid voluntary torque is likely linked to the faster contractile properties in hyperthermic conditions (Table 4), which increase the intrinsic capacity for RTD and counter the reduction in neural drive. The faster contractile properties with hyperthermia are likely due to increased muscle temperature. Although muscle temperature was not measured in the present study, research reporting similar magnitudes of change in Tre to the present study when heating the whole body have observed concomitant increases in muscle temperature. The muscle temperatures were reported from resting pre-heating values of ~ 35 °C (Tre ~ 37 °C) up to end of trial values of ~ 39.4 °C (Tre ~ 39.5 °C) (Périard et al. 2014b; Racinais and Girard, 2012). Greater muscle temperatures improve muscle fibre conduction velocity (Gray et al. 2006; Farina et al. 2005) and excitation contraction coupling (Brody 1976). This is due to the faster rate of myosin–actin attachment during cross-bridge cycling (Davies et al. 1982) and ATPase activity (Bárány 1967; Stein et al. 1982). Additionally, elevated core temperature appears to shorten the electromechanical delay between muscle activation and force production onsets, reflecting the faster excitation contraction coupling (Gordon et al. 2021). These electromechanical mechanisms observed with increased muscle temperature would contribute to the faster contractile responses observed in the present study. Our data align with others that have found faster twitch (Gordon et al. 2021; Périard et al. 2014b) and octet ½ RT (Gordon et al. 2021) during moderate (Tre 38.5 °C) and severe (Tre 39.5 °C) hyperthermia.
Effects of HA
The HA protocol employed in the present study successfully induced adaptation to repeated heat exposures (Table 1). Sweat rate was greater at HA 5 (+ 0.4 L·h−1), whilst reductions in resting Tre (− 0.5 °C) and HR (− 10 beat·min−1) and an increase in the sweating rate (+ 0.6 L·h−1) were observed by HA 10 relative to HA 1, the magnitude of which are all consistent with the HA literature (Sawka et al. 2011; Tyler et al. 2016). These data provide evidence that participants improved heat storage capacity, lowered cardiovascular strain, and subsequently ameliorated the heat loss mechanisms for more efficient thermoregulation.
We hypothesised that a regime of HA would attenuate the expected hyperthermia-induced reduction in neural drive, and thus attenuate a decline in MVT and enable faster RTD due to the faster contractile properties of the muscle. However, HA did not attenuate reductions in neural drive whilst hyperthermic, and in agreement with others who found no influence centrally mediated mechanisms (Wingfield et al. 2016; Osborne et al. 2021; Brazaitis and Skurvydas 2010). It is therefore not surprising that the effects (or lack thereof) of acute hyperthermia on MVT and rapid voluntary torque were unchanged following HA. To the author’s knowledge, the effect of 10 days of HA on rapid voluntary torque has not previously been investigated. However, our findings on MVT contrast with others. Racinais et al. (2017b) reported plantar flexion MVC torque in hyperthermic conditions to be 16% (P < 0.05) greater after 11 days of passive HA. Osborne et al. (2021) similarly observed increased knee extension torque (P < 0.05) in hyperthermic conditions following 5 days of HA. Nevertheless, the above increases in MVC torque whilst hyperthermic, following HA, were not attributed to increased neural drive to the muscle. We therefore speculate that for constant levels of hyperthermia and thermal sensation (as observed in this study; Table 2), reduced neural drive during whole-body hyperthermia is inevitable, and this effect cannot be ameliorated through HA.
Independent of hyperthermia, we did not observe any change in MVT (Table 3), RTD (Figs. 2, 3, 4), or any other neuromuscular variable (Table 4), following 5 or 10 days of HA. In contrast Racinais et al. (2017b) reported an 11% increase (P < 0.05) in maximal plantar flexion MVT in temperate (24 °C) conditions, following 11 days of passive HA. The increased baseline MVT was not due to changes in neural drive which remained unchanged following HA. Instead, the same study (Racinais Wilson and Périard 2017b) observed increased twitch peak torque in in temperate conditions following HA and suggested this HA-induced improvement in intrinsic contractile function likely caused the improvement in MVT. In contrast, our data showed the twitch and octet responses in temperate conditions (independent of hyperthermia) were unaffected by HA, which may explain why we did not observe improved MVT in cool conditions, whereas Racinais et al. (2017b) did. In addition to increased peak twitch torque, the same study (Racinais et al. 2017b) did observe a slowing of twitch ½ RT (P < 0.05) during the controlled background muscle contraction at 10% of MVC. Whilst our current study did not find evidence of slower ½ RT in baseline temperate conditions following HA, we did observe an interaction effect (P < 0.05) of acute hyperthermia and HA on twitch ½ RT (Table 4), providing some supporting evidence that HA affects the intrinsic contractile properties of muscle.
To the author’s knowledge, the interaction effect of hyperthermia and HA on twitch ½ RT (caused by an attenuation of the hyperthermia-induced reduction of ½ RT following HA) is a novel finding (Table 4). The mechanism and implication of this effect are unclear. Half RT is a measure of skeletal muscle relaxation after a single twitch or tetanic contraction initiated by reductions in Ca2+ concentration in the sarcoplasmic reticulum. The efficiency of this process is modulated by the dissociation of Ca2+ from troponin, translocation of the Ca2+ to the sarcoplasmic reticulum and then its subsequent active re-uptake (Bennett 1985). We speculate that the maintenance of ½ RT whilst hyperthermic post-HA may indicate phenotypic changes to the release and re-uptake of Ca2+. Attenuating the hyperthermic-induced reduction of ½ RT may serve to benefit force summation, by minimising the rightward shift in the force–frequency relationship that occurs with elevated core, and in turn muscle temperature (Périard et al. 2014b; Todd et al. 2005). The residual effect could be to help maintain MVT during whole-body hyperthermia.
The authors recognise some limitations of the study. The study design attempted to clamp Tre to standardise when the neuromuscular measurements were completed, which in Trial 3 led to elevated \(\overline{T}\)sk and HR values when baseline values were measured (Table 2). This is because successful HA lowered resting Tre, meaning participants spent longer time passively heating prior to the first neuromuscular set to attain a Tre of 37 °C. Elevated HR and \(\overline{T}\)sk (due to cardiovascular drift) suggest greater cardiovascular strain (Rowell 1974). However, because of the passive nature of the heating protocol used, the influence of moderately increased cardiovascular strain was likely to have been minimal. In addition, voluntary and involuntary evoked torque responses were similar at Tre of 37 °C pre- and post-HA, suggesting despite raised cardiovascular strain and \(\overline{T}\)sk, this did not affect neuromuscular responses.
Conclusion
In conclusion, neural drive was reduced during both maximal and rapid voluntary contractions during hyperthermia, but this did not affect MVT or rapid voluntary torque production. The preservation of voluntary torque when hyperthermic is likely due to improvements in contractile function mitigating the decline in neural drive. Neither 5 nor 10 days of HA mitigated the hyperthermia-induced decline in neural drive. Similarly, HA exhibited no beneficial effects on measures of voluntary torque production, independent of acute hyperthermia. Nevertheless, a novel finding from this study was the interaction between hyperthermia and HA on twitch ½ RT, which reduced the hyperthermia-induced speeding-up of rate of muscle relaxation, following HA. This could theoretically benefit force summation when exercising in the heat.
Availability of data and material
The datasets generated and analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- \(\overline{T}\) s k :
-
Mean weighted skin temperature (°C)
- ½ RT:
-
Half-relaxation time (ms)
- ANOVA:
-
Analysis of variance
- g :
-
Hedge’s g effect size
- EMG:
-
Electromyography
- HA:
-
Heat acclimation
- HR:
-
Heart rate (beat·min−1)
- MVC:
-
Maximal voluntary contraction
- MVT:
-
Maximal voluntary torque (Nm)
- pRTD:
-
Peak rate of torque development
- PT:
-
Peak torque (Nm)
- Rh:
-
Relative humidity
- RTD:
-
Rate of torque development (Nm·s−1)
- TC:
-
Thermal comfort
- TPT:
-
Time to peak torque (ms)
- Tre :
-
Rectal temperature (°C)
- TS:
-
Thermal sensation
- VA:
-
Voluntary activation (%)
References
Bárány M (1967) ATPase activity of myosin correlated with speed of muscle shortening. J Gen Physiol 50(6):197–218. https://doi.org/10.1085/jgp.50.6.197
Bennett AF (1985) Temperature and muscle. J Exp Biol 115:333–344
Brazaitis M, Skurvydas A (2010) Heat acclimation does not reduce the impact of hyperthermia on central fatigue. Eur J Appl Physiol 109(4):771–778. https://doi.org/10.1007/s00421-010-1429-3
Brody IA (1976) Regulation of isometric contraction in skeletal muscle. Exp Neurol 50(3):673–683. https://doi.org/10.1016/0014-4886(76)90036-4
Chalmers S et al (2014) Short-term heat acclimation training improves physical performance: a systematic review, and exploration of physiological adaptations and application for team sports. Sports Med (auckland, NZ) 44(7):971–988. https://doi.org/10.1007/s40279-014-0178-6
Cheung SS, McLellan TM (1998) Heat acclimation, aerobic fitness, and hydration effects on tolerance during uncompensable heat stress. J Appl Physiol (bethesda, MD) 84(5):1731–1739. https://doi.org/10.1152/jappl.1998.84.5.1731
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd edn. Erlbaum, Hillsdale
Davies CT, Mecrow IK, White MJ (1982) Contractile properties of the human triceps surae with some observations on the effects of temperature and exercise. Eur J Appl Physiol 49(2):255–269. https://doi.org/10.1007/BF02334074
de Ruiter CJ, de Haan A (2000) Temperature effect on the force/velocity relationship of the fresh and fatigued human adductor pollicis muscle. Pflugers Arch 440(1):163–170. https://doi.org/10.1007/s004240000284
de Ruiter CJ et al (1999) Temperature effect on the rates of isometric force development and relaxation in the fresh and fatigued human adductor pollicis muscle. Exp Physiol 84(6):1137–1150 (PHY_1895[pii])
Del Vecchio A et al (2019) You are as fast as your motor neurons: speed of recruitment and maximal discharge of motor neurons determine the maximal rate of force development in humans. J Physiol 597(9):2445–2456. https://doi.org/10.1113/JP277396
Dewhurst S et al (2005) Temperature dependence of soleus H-reflex and M wave in young and older women. Eur J Appl Physiol 94(5–6):491–499. https://doi.org/10.1007/s00421-005-1384-6
Ely MR et al (2007) Impact of weather on marathon-running performance. Med Sci Sports Exerc 39(3):487–493 (00005768-200703000-00012[pii])
Farina D, Arendt-Nielsen L, Graven-Nielsen T (2005) Effect of temperature on spike-triggered average torque and electrophysiological properties of low-threshold motor units. J Appl Physiol (bethesda, MD) 99(1):197–203 (00059.2005 [pii])
Folland JP, Buckthorpe MW, Hannah R (2014) Human capacity for explosive force production: neural and contractile determinants. Scand J Med Sci Sports 24(6):894–906. https://doi.org/10.1111/sms.12131
Gagge AP, Stolwijk JA, Hardy JD (1967) Comfort and thermal sensations and associated physiological responses at various ambient temperatures. Environ Res 1(1):1–20 (0013-9351(67)90002-3[pii])
Gaoua N, Grantham J, El Massioui F, Girard O, Racinais S (2011) Cognitive decrements do not follow neuromuscular alterations during passive heat exposure. Int J Hyperth 27(1):10–19. https://doi.org/10.3109/02656736.2010.519371
Garrett AT, Rehrer NJ, Patterson MJ (2011) Induction and decay of short-term heat acclimation in moderately and highly trained athletes. Sports Med (auckland, NZ) 41(9):757–771. https://doi.org/10.2165/11587320-000000000-00000
Gibson OR et al (2015) Isothermic and fixed intensity heat acclimation methods induce similar heat adaptation following short and long-term timescales. J Therm Biol 49–50:55–65 (S0306-4565(15)00023-6 [pii])
Gordon RJFH et al (2021) Progressive hyperthermia elicits distinct responses in maximum and rapid torque production. J Sci Med Sport. https://doi.org/10.1016/j.jsams.2021.03.007. (S1440-2440(21)00059-1[pii])
Gray SR et al (2006) Skeletal muscle ATP turnover and muscle fiber conduction velocity are elevated at higher muscle temperatures during maximal power output development in humans. Am J Physiol Regul Integr Comp Physiol 290(2):376 (00291.2005[pii])
Guy JH et al (2015) Adaptation to hot environmental conditions: an exploration of the performance basis, procedures and future directions to optimise opportunities for elite athletes. Sports Med (auckland, NZ) 45(3):303–311. https://doi.org/10.1007/s40279-014-0277-
Kodesh E, Horowitz M (2010) Soleus adaptation to combined exercise and heat acclimation: physiogenomic aspects. Med Sci Sports Exerc 42(5):943–952. https://doi.org/10.1249/MSS.0b013e3181c3ac3f
Maffiuletti NA et al (2016) Rate of force development: physiological and methodological considerations. Eur J Appl Physiol 116(6):1091–1116. https://doi.org/10.1007/s00421-016-3346-6
Mohr M et al (2012) Physiological responses and physical performance during football in the heat. PLoS ONE 7(6):e39202. https://doi.org/10.1371/journal.pone.0039202
Morante SM, Brotherhood JR (2008) Autonomic and behavioural thermoregulation in tennis. Brit J Sports Med 42(8):679–685 (discussion 685. bjsm.2007.042499 [pii])
Morrison S, Sleivert GG, Cheung SS (2004) Passive hyperthermia reduces voluntary activation and isometric force production. Eur J Appl Physiol 91(5–6):729–736. https://doi.org/10.1007/s00421-004-1063-z
Notley SR et al (2019) Menstrual cycle phase does not modulate whole body heat loss during exercise in hot, dry conditions. J Appl Physiol (bethesda, MD) 126(2):286–293. https://doi.org/10.1152/japplphysiol.00735.2018
Nybo L, Nielsen B (2001) Hyperthermia and central fatigue during prolonged exercise in humans. J Appl Physiol (bethesda, MD) 91(3):1055–1060. https://doi.org/10.1152/jappl.2001.91.3.1055
Osborne JO et al (2021) Short-term heat acclimation preserves knee extensor torque but does not improve 20 km self-paced cycling performance in the heat. Eur J Appl Physiol. https://doi.org/10.1007/s00421-021-04744-y
Périard JD et al (2014a) Voluntary muscle and motor cortical activation during progressive exercise and passively induced hyperthermia. Exp Physiol 99(1):136–148. https://doi.org/10.1113/expphysiol.2013.074583
Périard JD, Racinais S, Thompson MW (2014b) Adjustments in the force-frequency relationship during passive and exercise-induced hyperthermia. Muscle Nerve 50(5):822–829. https://doi.org/10.1002/mus.24228
Périard JD, Racinais S, Sawka MN (2015) Adaptations and mechanisms of human heat acclimation: applications for competitive athletes and sports. Scand J Med Sci Sports 25(Suppl 1):20–38. https://doi.org/10.1111/sms.12408
Piil JF et al (2019) Heat acclimation does not protect trained males from hyperthermia-induced impairments in complex task performance. Int J Environ Res Public Health 16(5):716. https://doi.org/10.3390/ijerph16050716
Racinais S, Girard O (2012) Neuromuscular failure is unlikely to explain the early exercise cessation in hot ambient conditions. Psychophysiology 49(6):853–865. https://doi.org/10.1111/j.1469-8986.2012.01360.x
Racinais S et al (2015) Effect of heat and heat acclimatization on cycling time trial performance and pacing. Med Sci Sports Exerc 47(3):601–606. https://doi.org/10.1249/MSS.0000000000000428
Racinais S et al (2017a) Heat acclimation has a protective effect on the central but not peripheral nervous system. J Appl Physiol (bethesda, MD) 123(4):816–824. https://doi.org/10.1152/japplphysiol.00430.2017
Racinais S, Wilson MG, Périard JD (2017b) Passive heat acclimation improves skeletal muscle contractility in humans. Am J Physiol Regul Integr Comp Physiol 312(1):R101–R107. https://doi.org/10.1152/ajpregu.00431.2016
Ramanathan NL (1964) A new weighting system for mean surface temperature of the human body. J Appl Physiol 19:531–533. https://doi.org/10.1152/jappl.1964.19.3.531
Rowell LB (1974) Human cardiovascular adjustments to exercise and thermal stress. Physiol Rev 54(1):75–159. https://doi.org/10.1152/physrev.1974.54.1.75
Sawka MN et al (2011) Integrated physiological mechanisms of exercise performance, adaptation, and maladaptation to heat stress. Compr Physiol 1(4):1883–1928. https://doi.org/10.1002/cphy.c100082
Stein RB, Gordon T, Shriver J (1982) Temperature dependence of mammalian muscle contractions and ATPase activities. Biophys J 40(2):97–107 (S0006-3495(82)84464-0[pii])
Taylor NA (2014) Human heat adaptation. Compr Physiol 4(1):325–365. https://doi.org/10.1002/cphy.c130022
Thomas MM et al (2006) Voluntary muscle activation is impaired by core temperature rather than local muscle temperature. J Appl Physiol (bethesda, MD) 100(4):1361–1369 (00945.2005 [pii])
Tillin NA et al (2010) Neuromuscular performance of explosive power athletes versus untrained individuals. Med Sci Sports Exerc 42(4):781–790. https://doi.org/10.1249/MSS.0b013e3181be9c7e
Tillin NA, Pain MT, Folland JP (2012) Contraction type influences the human ability to use the available torque capacity of skeletal muscle during explosive efforts. Proc Biol Sci 279(1736):2106–2115. https://doi.org/10.1098/rspb.2011.2109
Tillin NA, Pain MT, Folland JP (2013) Identification of contraction onset during explosive contractions. Response to thompson et al. “ consistency of rapid muscle force characteristics: Influence of muscle contraction onset detection methodology” [J electromyogr kinesiol 2012;22(6):893–900]. J Electromyogr Kinesiol 23(4):991–994 (S1050-6411(13)00113-2 [pii])
Tillin NA, Pain MTG, Folland JP (2018) Contraction speed and type influences rapid utilisation of available muscle force: neural and contractile mechanisms. J Exp Biol. https://doi.org/10.1242/jeb.193367. (jeb193367 [pii])
Todd G et al (2005) Hyperthermia: a failure of the motor cortex and the muscle. J Physiol 563(Pt 2):621–631 (jphysiol.2004.077115[pii])
Tyler CJ et al (2016) The effects of heat adaptation on physiology, perception and exercise performance in the heat: a meta-analysis. Sports Med (auckland, NZ) 46(11):1699–1724. https://doi.org/10.1007/s40279-016-0538-5
Wingfield, et al (2016) The effect of high versus low intensity heat acclimation on performance and neuromuscular responses. J Therm Biol 58:50–59 (S0306-4565(15)30164-9[pii])
Young AJ et al (1987) Cooling different body surfaces during upper and lower body exercise. J Appl Physiol (bethesda, Md) 63(3):1218–1223. https://doi.org/10.1152/jappl.1987.63.3.1218
Acknowledgements
The authors would like to thank all the participants who took part in this research for their time and effort and Professor Lewis Halsey for his assistance with data collection.
Funding
No financial assistance was provided to conduct the study described in the manuscript or used to assist with the preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
The present investigation was conducted at the Sports and Exercise Science Research Centre physiology laboratory, located on the Whitelands campus at the University of Roehampton. RG, CD, CT and NT contributed to the conception and design of the study. RG, JM, FC, TR, CD, CT and NT contributed to the acquisition, analysis and interpretation of the data. RG drafted the manuscript and JM, FC, TR, CD, CT and NT made critical revisions. RG, JM, FC, TR, CD, CT and NT have approved the final version of the manuscript. RG, JM, FC, TR, CD, CT and NT agree to be accountable for all aspects of the presented work. RG, JM, FC, TR, CD, CT and NT qualify for authorship.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to disclose.
Ethical approval
Experimental procedures were approved by the Ethical Advisory Committee of the University of Roehampton (LSC 19/259), in accordance with the latest iteration of the Declaration of Helsinki.
Consent to participate
All participants were informed of any risks and discomforts associated with the experiment before giving their written informed consent.
Additional information
Communicated by George Havenith.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Gordon, R.J.F.H., Moss, J.N., Castelli, F. et al. Heat acclimation reduces the effects of whole-body hyperthermia on knee-extensor relaxation rate, but does not affect voluntary torque production. Eur J Appl Physiol 123, 1067–1080 (2023). https://doi.org/10.1007/s00421-022-05127-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-022-05127-7