Abstract
Purpose
Sickle cell trait is characterized by the presence of both normal and abnormal haemoglobin in red blood cells. The rate of exertional collapse is increased in athletes and military recruits who carry the trait, particularly in stressful environmental conditions. The aim of the present study was to investigate microvascular function and its determinants in response to intense exercise at control and warm environmental temperatures in carriers (AS) and non-carriers (AA) of sickle cell trait.
Methods
Nine AS and 11 AA, all healthy physically active young men, randomly participated in four experimental sessions (rest at 21 °C and 31 °C and cycling at 21 °C and 31 °C). All participants performed three exercises bouts as follows: 18-min submaximal exercise; an incremental test to exhaustion; and three 30-s sprints spaced with 20-s resting intervals.
Results
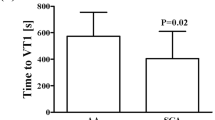
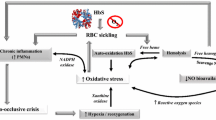
Skin Blood Flow (SkBF) was similar at rest between AA and AS. SkBF for all participants was higher at 31 °C than 21 °C. It was significantly higher in the AS group compared to the AA group immediately after exercise, regardless of the environmental conditions. No significant differences in hemorheological parameters, muscle damage or cardiac injury biomarkers were observed between the two groups. Our data also suggest higher oxidative stress for the AS group, with high superoxide dismutase (P = 0.044 main group effect).
Conclusion
A specific profile is identified in the AS population, with increased microvascular reactivity after maximal exercise in stressful environment and slight pro-/antioxidant imbalance.




Similar content being viewed by others
Abbreviations
- AI%:
-
Aggregation index
- AOPP:
-
Advanced oxidation protein products
- CAT:
-
Catalase
- CL– :
-
Chlorine
- CPK:
-
Creatine phosphokinase
- CPK-MB:
-
Creatine phosphokinase –myoglobin
- GSH:
-
Reduced glutathione
- GSSG:
-
Oxidized glutathione
- HSP70:
-
Heat shock protein 70
- LDH:
-
Lactate dehydrogenase
- K+ :
-
Potassium
- MDA:
-
Malondialdehyde
- MPO:
-
Myeloperoxidase
- NO:
-
Oxide nitric
- NT PROBNP:
-
N-terminal pro-brain natriuretic peptide
- RBC:
-
Red blood cell
- SCT:
-
Sickle cell trait
- SKBF:
-
Skin blood flow
- SOD:
-
Superoxide dismutase
References
Alayash AI (2018) Oxidative Pathways in the Sickle Cell and Beyond. Blood Cells Mol Dis 70:78–86. https://doi.org/10.1016/j.bcmd.2017.05.009
Bilé A, Le Gallais D, Mercier B, Martinez P, Ahmaidi S, Préfaut C (1996) Anaerobic exercise components during the force-velocity test in sickle cell trait. Int J Sports Med 17(4):254–258. https://doi.org/10.1055/s-2007-972842
Buchanan BK, Siebert DM, Suchsland MLZ, Drezner JA, Asif IM, O’Connor FG, Harmon KG (2020) Sudden Death Associated with Sickle Cell Trait before and after Mandatory Screening. Sports Health. https://doi.org/10.1177/1941738120915690
Charkoudian N, Stachenfeld NS (2014) Reproductive Hormone Influences on Thermoregulation in Women. In: Comprehensive Physiology (p. 793‑804). American Cancer Society. https://doi.org/10.1002/cphy.c130029
Chirico EN, Faës C, Connes P, Canet-Soulas E, Martin C, Pialoux V (2016) Role of exercise-induced oxidative stress in sickle cell trait and disease. Sports Med 46(5):629–639. https://doi.org/10.1007/s40279-015-0447-z
Connes P, Sara F, Hardy-Dessources M-D, Marlin L, Etienne F, Larifla L, Saint-Martin C, Hue O (2006) Effects of short supramaximal exercise on hemorheology in sickle cell trait carriers. Eur J Appl Physiol 97(2):143–150. https://doi.org/10.1007/s00421-006-0155-3
Das SK, Hinds JE, Hardy RE, Collins JC, Mukherjee S (1993) Effects of physical stress on peroxide scavengers in normal and sickle cell trait erythrocytes. Free Radical Biol Med 14(2):139–147. https://doi.org/10.1016/0891-5849(93)90004-e
Diaw M, Samb A, Diop S, Sall ND, Ba A, Cissé F, Connes P (2014) Effects of hydration and water deprivation on blood viscosity during a soccer game in sickle cell trait carriers. Br J Sports Med 48(4):326–331. https://doi.org/10.1136/bjsports-2012-091038
Diaw M, Pialoux V, Martin C, Samb A, Diop S, Faes C, Mury P, Sall Diop N, Diop S-N, Ranque B, Mbaye MN, Key NS, Connes P (2015) Sickle cell trait worsens oxidative stress, abnormal blood rheology, and vascular dysfunction in Type 2 Diabetes. Diabetes Care 38(11):2120–2127. https://doi.org/10.2337/dc15-0699
Francisco MA, Brunt VE, Jensen KN, Lorenzo S, Minson CT (2017) Ten days of repeated local forearm heating does not affect cutaneous vascular function. J Appl Physiol 123(2):310–316. https://doi.org/10.1152/japplphysiol.00966.2016 ((Bethesda, Md.: 1985))
Gozal D, Thiriet P, Mbala E, Wouassi D, Gelas H, Geyssant A, Lacour JR (1992) Effect of different modalities of exercise and recovery on exercise performance in subjects with sickle cell trait. Med Sci Sports Exerc 24(12):1325–1331
Harmon KG, Drezner JA, Klossner D, Asif IM (2012) Sickle cell trait associated with a RR of death of 37 times in national collegiate athletic association football athletes: a database with 2 million athlete-years as the denominator. Br J Sports Med 46(5):325–330. https://doi.org/10.1136/bjsports-2011-090896
Hebbel RP, Morgan WT, Eaton JW, Hedlund BE (1988) Accelerated autoxidation and heme loss due to instability of sickle hemoglobin. Proc Natl Acad Sci USA 85(1): 237‑241. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC279519/
Hedreville M, Connes P, Romana M, Magnaval G, David T, Hardy-Dessources M-D, Belloy M-S, Etienne-Julan M, Hue O (2009) Central retinal vein occlusion in a sickle cell trait carrier after a cycling race. Med Sci Sports Exerc 41(1):14–18. https://doi.org/10.12s49/MSS.0b013e31818313d0
Hierso R, Waltz X, Mora P, Romana M, Lemonne N, Connes P, Hardy-Dessources M-D (2014) Effects of oxidative stress on red blood cell rheology in sickle cell patients. Br J Haematol 166(4):601–606. https://doi.org/10.1111/bjh.12912
Ighodaro OM, Akinloye OA (2018) First line defence antioxidants-superoxide dismutase (SOD), catalase (CAT) and glutathione peroxidase (GPX): their fundamental role in the entire antioxidant defence grid. Alex J Med 54(4):287–293. https://doi.org/10.1016/j.ajme.2017.09.001
Kark JA, Posey DM, Schumacher HR, Ruehle CJ (1987) Sickle-cell trait as a risk factor for sudden death in physical training. N Engl J Med 317(13):781–787. https://doi.org/10.1056/NEJM198709243171301
Key NS, Connes P, Derebail VK (2015) Negative health implications of sickle cell trait in high income countries: from the football field to the laboratory. Br J Haematol 170(1):5–14. https://doi.org/10.1111/bjh.13363
Marlin L, Connes P, Antoine-Jonville S, Tripette J, Montout-Hedreville M, Sanouiller A, Etienne-Julan M, Hue O (2008) Cardiorespiratory responses during three repeated incremental exercise tests in sickle cell trait carriers. Eur J Appl Physiol 102(2):181–187. https://doi.org/10.1007/s00421-007-0570-0
Messonnier L, Samb A, Tripette J, Gogh BD, Loko G, Sall ND, Féasson L, Hue O, Lamothe S, Bogui P, Connes P (2012) Moderate endurance exercise is not a risk for rhabdomyolysis or renal failure in sickle cell trait carriers. Clin Hemorheol Microcirc 51(3):193–202. https://doi.org/10.3233/CH-2011-1524
Nader E, Skinner S, Romana M, Fort R, Lemonne N, Guillot N, Gauthier A, Antoine-Jonville S, Renoux C, Hardy-Dessources M-D, Stauffer E, Joly P, Bertrand Y, Connes P (2019) Blood rheology: key parameters, impact on blood flow, role in sickle cell disease and effects of exercise. Front Physiol. https://doi.org/10.3389/fphys.2019.01329
Nelson DA, Deuster PA, O’Connor FG, Kurina LM (2018) Sickle cell trait and heat injury among US Army soldiers. Am J Epidemiol 187(3):523–528. https://doi.org/10.1093/aje/kwx285
O’Connor FG, Bergeron MF, Cantrell J, Connes P, Harmon KG, Ivy E, Kark J, Klossner D, Lisman P, Meyers BK, O’Brien K, Ohene-Frempong K, Thompson AA, Whitehead J, Deuster PA (2012) ACSM and CHAMP summit on sickle cell trait: mitigating risks for warfighters and athletes. Med Sci Sports Exerc 44(11):2045–2056. https://doi.org/10.1249/MSS.0b013e31826851c2
Pedersen ES, Tengesdal S, Radtke M, Langlo KAR (2019) Major increase in creatine kinase after intensive exercise. Tidsskrift for Den Norske Laegeforening: Tidsskrift for Praktisk Medicin, Ny Raekke. https://doi.org/10.4045/tidsskr.18.0120
Quattrone RD, Eichner ER, Beutler A, Adams WB, O’Connor FG (2015) Exercise collapse associated with sickle cell trait (ECAST): case report and literature review. Curr Sports Med Rep 14(2):110–116. https://doi.org/10.1249/JSR.0000000000000137
Reminy K, Hue O, Antoine-Jonville S (2020) Effect of warm environment on the skin blood flow response to food intake. Int J Hyperth 37(1):836–842. https://doi.org/10.1080/02656736.2020.1788174
Romero SA, Gagnon D, Adams AN, Cramer MN, Kouda K, Crandall CG (2017) Acute limb heating improves macro- and microvascular dilator function in the leg of aged humans. Am J Physiol Heart Circ Physiol 312(1):H89–H97. https://doi.org/10.1152/ajpheart.00519.2016
Roustit M, Blaise S, Millet C, Cracowski JL (2010) Reproducibility and methodological issues of skin post-occlusive and thermal hyperemia assessed by single-point laser Doppler flowmetry. Microvasc Res 79(2):102–108. https://doi.org/10.1016/j.mvr.2010.01.001
Sara F, Hardy-Dessources M-D, Marlin L, Connes P, Hue O (2006) Lactate distribution in the blood compartments of sickle cell trait carriers during incremental exercise and recovery. Int J Sports Med 27(6):436–443. https://doi.org/10.1055/s-2005-865844
Saxena P, Chavarria C, Thurlow J (2016) Rhabdomyolysis in a Sickle Cell Trait Positive Active Duty Male Soldier. U.S. Army Med Dep J 20–23.
Singer DE, Byrne C, Chen L, Shao S, Goldsmith J, Niebuhr DW (2018) Risk of Exertional Heat Illnesses Associated with Sickle Cell Trait in U.S. Military. Military Medicine, 183(7‑8), e310–e317. https://doi.org/10.1093/milmed/usx085
Skinner S, Connes P, Sigaudo-Roussel D, Lo M, Liu KL, Nader E, Josset-Lamaugarny A, Charrin E, Martin C, Romanet-Faes C, Diaw M, Pialoux V, Fromy B (2019) Altered blood rheology and impaired pressure-induced cutaneous vasodilation in a mouse model of combined type 2 diabetes and sickle cell trait. Microvasc Res 122:111–116. https://doi.org/10.1016/j.mvr.2018.11.014
Skinner S, Liu KL, Lo M, Josset-Lamaugarny A, Charrin E, Martin C, Pialoux V, Fromy B, Connes P, Sigaudo-Roussel D (2020) Alterations in vascular reactivity in a transgenic mouse model of sickle cell trait. Br J Haematol 189(4):e154–e157. https://doi.org/10.1111/bjh.16577
Tripette J, Alexy T, Hardy-Dessources M-D, Mougenel D, Beltan E, Chalabi T, Chout R, Etienne-Julan M, Hue O, Meiselman HJ, Connes P (2009) Red blood cell aggregation, aggregate strength and oxygen transport potential of blood are abnormal in both homozygous sickle cell anemia and sickle-hemoglobin C disease. Haematologica 94(8):1060–1065. https://doi.org/10.3324/haematol.2008.005371
Tripette J, Connes P, Beltan E, Chalabi T, Marlin L, Chout R, Baskurt OK, Hue O, Hardy-Dessources M-D (2010a) Red blood cell deformability and aggregation, cell adhesion molecules, oxidative stress and nitric oxide markers after a short term, submaximal, exercise in sickle cell trait carriers. Clin Hemorheol Microcirc 45(1):39–52. https://doi.org/10.3233/CH-2010-1325
Tripette J, Loko G, Samb A, Gogh BD, Sewade E, Seck D, Hue O, Romana M, Diop S, Diaw M, Brudey K, Bogui P, Cissé F, Hardy-Dessources M-D, Connes P (2010b) Effects of hydration and dehydration on blood rheology in sickle cell trait carriers during exercise. Am J Physiol-Heart Circ Physiol 299(3):H908–H914. https://doi.org/10.1152/ajpheart.00298.2010
Vincent L, Féasson L, Oyono-Enguéllé S, Banimbek V, Denis C, Guarneri C, Aufradet E, Monchanin G, Martin C, Gozal D, Dohbobga M, Wouassi D, Garet M, Thiriet P, Messonnier L (2010) Remodeling of skeletal muscle microvasculature in sickle cell trait and alpha-thalassemia. Am J Physiol Heart Circ Physiol 298(2):375–384. https://doi.org/10.1152/ajpheart.00812.2009
Walløe L (2015) Arterio-venous anastomoses in the human skin and their role in temperature control. Temp Multidiscip Biomed J 3(1):92–103. https://doi.org/10.1080/23328940.2015.1088502
Wang Y, Branicky R, Noë A, Hekimi S (2018) Superoxide dismutases: Dual roles in controlling ROS damage and regulating ROS signaling. J Cell Biol 217(6):1915–1928. https://doi.org/10.1083/jcb.201708007
Witko-Sarsat V, Friedlander M, Capeillère-Blandin C et al (1996) Advanced oxidation protein products as a novel marker of oxidative stress in uremia. Kidney Int 49:1304–13. https://doi.org/10.1038/ki.1996.186
Zimmerman JL, Shen MC (2013) Rhabdomyolysis. Chest 144(3):1058–1065. https://doi.org/10.1378/chest.12-2016
Acknowledgements
We thank the participants, nurses (coordinated by Franceline Falla), and physicians (Patrick Chérubin, Nicolas Benoit) for their great involvement.
Funding
Funding for this work was provided by European Regional Development Fund & Région Guadeloupe 2015-FED-213.
Author information
Authors and Affiliations
Contributions
KR: formal analysis, investigation, methodology, writing– original draft, writing review & editing. ETNS: data curation, formal analysis, investigation, review & editing. MR: investigation, resources, writing-review & editing. PC: conceptualization, formal analysis, resources, writing – review & editing. SR: investigation, methodology. SH: investigation, methodology. OH: original draft, review & editing. MHD: investigation, methodology, writing – review & editing. SAJ: conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, project administration, resources, supervision, writing – original draft, writing – review & editing.
Corresponding author
Ethics declarations
Conflict interest
The author declares no financial or other conflict of interest that might bias this article.
Ethical approval
The participants were informed of the procedures and purposes of the study, which were in accordance with the institutional guidelines and the Helsinki Declaration of 2013 and was approved by the National Ethics Committee (CPP 17.10.10). This protocol described in this article is part of a larger project registered in EudraCT (TDEX: 2017-A02226-47) and Clinical Trials (NCT04028791).
Additional information
Communicated by Narihiko kondo.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Reminy, K., Ngo Sock, E.T., Romana, M. et al. Strenuous exercise in warm environment is associated with improved microvascular function in sickle cell trait. Eur J Appl Physiol 122, 185–197 (2022). https://doi.org/10.1007/s00421-021-04821-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-021-04821-2