Abstract
Purpose
Blood rheology is a key determinant of blood flow and tissue perfusion. There are still large discrepancies regarding the effects of an acute running exercise on blood rheological properties and red blood cell (RBC) physiology. We investigated the effect of a 10 km running trial on markers of blood rheology and RBC physiology in endurance trained athletes.
Methods
Blood was sampled before and after the exercise to measure lactate and glucose, hematological and hemorheological parameters (blood viscosity, RBC deformability, and aggregation), eryptosis markers (phosphatidylserine and CD47 exposure, RBC reactive oxygen species), RBC-derived microparticles (RBC-MPs), and RBC electrophysiological activity. Weight was measured before and after exercise. Peripheral oxygen saturation and heart rate were monitored before and during the trial.
Results
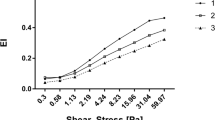
Blood lactate and glucose levels increased after exercise and subjects significantly lost weight. All athletes experienced a significant fall in oxygen saturation. Mean corpuscular volume (MCV) was increased from 95.1 ± 3.2 to 96.0 ± 3.3 and mean corpuscular hemoglobin concentration (MCHC) decreased after exercise suggesting a slight RBC rehydration. Exercise increased RBC deformability from 0.344 ± 0.04 to 0.378 ± 0.07, decreased RBC aggregates strength and blood viscosity, while hematocrit (Hct) remained unaffected. While RBC electrophysiological recording suggested a modulation in RBC calcium content and/or chloride conductance, eryptosis markers and RBC-MPs were not modified by the exercise.
Conclusion
A 10 km acute running exercise had no effect on RBC senescence and membrane blebbing. In contrast, this exercise increased RBC deformability, probably through rehydration process which resulted in a decrease in blood viscosity.





Similar content being viewed by others
Abbreviations
- Ca2+ :
-
Calcium
- CCCP:
-
Carbonyl cyanide m-chlorophenylhydrazone
- EI:
-
Elongation index
- Hb:
-
Hemoglobin
- Hct:
-
Hematocrit
- HR:
-
Heart rate
- Hypermax :
-
Red blood cell maximum hyperpolarization
- MAS:
-
Maximal aerobic speed
- MCH:
-
Mean corpuscular hemoglobin
- MCHC:
-
Mean corpuscular hemoglobin concentration
- MCV:
-
Mean corpuscular volume
- MFI:
-
Median fluorescence intensity
- MPs:
-
Microparticles
- NS:
-
Not significant
- PLA:
-
Platelets
- PS:
-
Phosphatidylserine
- RBC:
-
Red blood cells
- ROS:
-
Reactive oxygen species
- RT:
-
Room temperature
- SPO2 :
-
Peripheral oxygen saturation
- S/V:
-
Surface to volume ratio
- Vm :
-
Membrane potential
- VO2 max :
-
Maximal oxygen consumption
- WBC:
-
White blood cells
References
Baskurt OK, Meiselman HJ (2003) Blood rheology and hemodynamics. Semin Thromb Hemost 29(5):435–450. https://doi.org/10.1055/s-2003-44551
Baskurt OK, Temiz A, Meiselman HJ (1998) Effect of superoxide anions on red blood cell rheologic properties. Free Radic Biol Med 24(1):102–110
Baskurt OK, Yalcin O, Meiselman HJ (2004) Hemorheology and vascular control mechanisms. Clin Hemorheol Microcirc 30(3–4):169–178
Baskurt OK, Boynard M, Cokelet GC, Connes P, Cooke BM, Forconi S, Liao F, Hardeman MR, Jung F, Meiselman HJ, Nash G, Nemeth N, Neu B, Sandhagen B, Shin S, Thurston G, Wautier JL, International Expert Panel for Standardization of Hemorheological M (2009) New guidelines for hemorheological laboratory techniques. Clin Hemorheol Microcirc 42(2):75–97. https://doi.org/10.3233/ch-2009-1202
Baunbaek M, Bennekou P (2008) Evidence for a random entry of Ca2 + into human red cells. Bioelectrochemistry 73(2):145–150. https://doi.org/10.1016/j.bioelechem.2008.04.006
Bennekou P, de Franceschi L, Pedersen O, Lian L, Asakura T, Evans G, Brugnara C, Christophersen P (2001) Treatment with NS3623, a novel Cl-conductance blocker, ameliorates erythrocyte dehydration in transgenic SAD mice: a possible new therapeutic approach for sickle cell disease. Blood 97(5):1451–1457
Cabrales P, Martini J, Intaglietta M, Tsai AG (2006) Blood viscosity maintains microvascular conditions during normovolemic anemia independent of blood oxygen-carrying capacity. Am J Physiol Heart Circ Physiol 291(2):H581–H590. https://doi.org/10.1152/ajpheart.01279.2005
Cahalan SM, Lukacs V, Ranade SS, Chien S, Bandell M, Patapoutian A (2015) Piezo1 links mechanical forces to red blood cell volume. Elife. https://doi.org/10.7554/eLife.07370
Chapman KR, D’Urzo A, Rebuck AS (1983) The accuracy and response characteristics of a simplified ear oximeter. Chest 83(6):860–864. https://doi.org/10.1378/chest.83.6.860
Chou SL, Huang YC, Fu TC, Hsu CC, Wang JS (2016) Cycling exercise training alleviates hypoxia-impaired erythrocyte rheology. Med Sci Sports Exerc 48(1):57–65. https://doi.org/10.1249/MSS.0000000000000730
Clark MR, Mohandas N, Shohet SB (1983) Osmotic gradient ektacytometry: comprehensive characterization of red cell volume and surface maintenance. Blood 61(5):899–910
Cokelet GR, Meiselman HJ (2007) Macro- and micro-rheological properties of blood. In: Baskurt OK, Hardeman MR, Rampling MW, Meiselman HJ (eds) Handbook of hemorheology and hemodynamics, pp 45–71. ISBN 978-1-58603-771-0
Connes P, Bouix D, Py G, Caillaud C, Kippelen P, Brun JF, Varray A, Prefaut C, Mercier J (2004a) Does exercise-induced hypoxemia modify lactate influx into erythrocytes and hemorheological parameters in athletes? J Appl Physiol 97(3):1053–1058. https://doi.org/10.1152/japplphysiol.00993.2003
Connes P, Bouix D, Py G, Prefaut C, Mercier J, Brun JF, Caillaud C (2004b) Opposite effects of in vitro lactate on erythrocyte deformability in athletes and untrained subjects. Clin Hemorheol Microcirc 31(4):311–318
Connes P, Frank S, Martin C, Shin S, Aufradet E, Sunoo S, Klara B, Raynaud de Mauverger E, Romana M, Messonnier L, Kang J, Varlet-Marie E, Feasson L, Hardy-Dessources MD, Wilhelm B, Brun JF (2010) New fundamental and applied mechanisms in exercise hemorheology. Clin Hemorheol Microcirc 45(2–4):131–141. https://doi.org/10.3233/CH-2010-1291
Connes P, Pichon A, Hardy-Dessources MD, Waltz X, Lamarre Y, Simmonds MJ, Tripette J (2012) Blood viscosity and hemodynamics during exercise. Clin Hemorheol Microcirc 51(2):101–109. https://doi.org/10.3233/CH-2011-1515
Connes P, Simmonds MJ, Brun JF, Baskurt OK (2013) Exercise hemorheology: classical data, recent findings and unresolved issues. Clin Hemorheol Microcirc 53(1–2):187–199. https://doi.org/10.3233/CH-2012-1643
Connes P, Lamarre Y, Waltz X, Ballas SK, Lemonne N, Etienne-Julan M, Hue O, Hardy-Dessources MD, Romana M (2014) Haemolysis and abnormal haemorheology in sickle cell anaemia. Br J Haematol 165(4):564–572. https://doi.org/10.1111/bjh.12786
El-Sayed MS, Ali N, El-Sayed Ali Z (2005) Haemorheology in exercise and training. Sports Med 35(8):649–670
Gay F, Aguera K, Senechal K, Tainturier A, Berlier W, Maucort-Boulch D, Honnorat J, Horand F, Godfrin Y, Bourgeaux V (2017) Methionine tumor starvation by erythrocyte-encapsulated methionine gamma-lyase activity controlled with per os vitamin B6. Cancer Med 6(6):1437–1452. https://doi.org/10.1002/cam4.1086
Hakim TS (1988) Erythrocyte deformability and segmental pulmonary vascular resistance: osmolarity and heat treatment. J Appl Physiol 65(4):1634–1641
Hierso R, Waltz X, Mora P, Romana M, Lemonne N, Connes P, Hardy-Dessources MD (2014) Effects of oxidative stress on red blood cell rheology in sickle cell patients. Br J Haematol 166(4):601–606. https://doi.org/10.1111/bjh.12912
Juel C, Hellsten Y, Saltin B, Bangsbo J (1999) Potassium fluxes in contracting human skeletal muscle and red blood cells. Am J Physiol 276(1):R184–R188. https://doi.org/10.1152/ajpregu.1999.276.1.R184
Kuchel PW, Benga G (2005) Why does the mammalian red blood cell have aquaporins? Biosystems 82(2):189–196. https://doi.org/10.1016/j.biosystems.2005.07.002
Lang E, Lang F (2015) Mechanisms and pathophysiological significance of eryptosis, the suicidal erythrocyte death. Semin Cell Dev Biol 39:35–42. https://doi.org/10.1016/j.semcdb.2015.01.009
Macey RI, Adorante JS, Orme FW (1978) Erythrocyte membrane potentials determined by hydrogen ion distribution. Biochim Biophys Acta 512(2):284–295
Makhro A, Haider T, Wang J, Bogdanov N, Steffen P, Wagner C, Meyer T, Gassmann M, Hecksteden A, Kaestner L, Bogdanova A (2016) Comparing the impact of an acute exercise bout on plasma amino acid composition, intraerythrocytic Ca(2 +) handling, and red cell function in athletes and untrained subjects. Cell Calcium 60(4):235–244. https://doi.org/10.1016/j.ceca.2016.05.005
Mao TY, Fu LL, Wang JS (2011) Hypoxic exercise training causes erythrocyte senescence and rheological dysfunction by depressed Gardos channel activity. J Appl Physiol 111(2):382–391. https://doi.org/10.1152/japplphysiol.00096.2011
McKelvie RS, Lindinger MI, Heigenhauser GJ, Jones NL (1991) Contribution of erythrocytes to the control of the electrolyte changes of exercise. Can J Physiol Pharmacol 69(7):984–993. https://doi.org/10.1139/y91-148
Moersdorf D, Egee S, Hahn C, Hanf B, Ellory C, Thomas S, Bernhardt I (2013) Transmembrane potential of red blood cells under low ionic strength conditions. Cell Physiol Biochem 31(6):875–882. https://doi.org/10.1159/000350105
Nader E, Guillot N, Lavorel L, Hancco I, Fort R, Stauffer E, Renoux C, Joly P, Germain M, Connes P (2018) Eryptosis and hemorheological responses to maximal exercise in athletes: comparison between running and cycling. Scand J Med Sci Sports 28(5):1532–1540. https://doi.org/10.1111/sms.13059
Nebor D, Romana M, Santiago R, Vachiery N, Picot J, Broquere C, Chaar V, Doumdo L, Odievre MH, Benkerrou M, Elion J (2013) Fetal hemoglobin and hydroxycarbamide modulate both plasma concentration and cellular origin of circulating microparticles in sickle cell anemia children. Haematologica 98(6):862–867. https://doi.org/10.3324/haematol.2012.073619
Neuhaus D, Gaehtgens P (1994) Haemorrheology and long term exercise. Sports Med 18(1):10–21. https://doi.org/10.2165/00007256-199418010-00003
Neuhaus D, Behn C, Gaehtgens P (1992) Haemorheology and exercise: intrinsic flow properties of blood in marathon running. Int J Sports Med 13(7):506–511. https://doi.org/10.1055/s-2007-1021307
Nielsen HB (2003) Arterial desaturation during exercise in man: implication for O2 uptake and work capacity. Scand J Med Sci Sports 13(6):339–358
Parrow NL, Violet PC, Tu H, Nichols J, Pittman CA, Fitzhugh C, Fleming RE, Mohandas N, Tisdale JF, Levine M (2018) Measuring deformability and red cell heterogeneity in blood by ektacytometry. J Vis Exp. https://doi.org/10.3791/56910
Parthasarathi K, Lipowsky HH (1999a) Capillary recruitment in response to tissue hypoxia and its dependence on red blood cell deformability. Am J Physiol 277(6):H2145–H2157. https://doi.org/10.1152/ajpheart.1999.277.6.H2145
Parthasarathi K, Lipowsky HH (1999b) Capillary recruitment in response to tissue hypoxia and its dependence on red blood cell deformability. Am J Physiol 277(6 Pt 2):H2145–H2157
Prefaut C, Durand F, Mucci P, Caillaud C (2000) Exercise-induced arterial hypoxaemia in athletes: a review. Sports Med 30(1):47–61
Raj JU, Anderson J (1991) Comparison of dextran and hematocrit effects in the pulmonary microcirculation. Circ Res 68(4):1108–1116. https://doi.org/10.1161/01.res.68.4.1108
Reithmeier RA (2001) A membrane metabolon linking carbonic anhydrase with chloride/bicarbonate anion exchangers. Blood Cells Mol Dis 27(1):85–89. https://doi.org/10.1006/bcmd.2000.0353
Renoux C, Faivre M, Bessaa A, Da Costa L, Joly P, Gauthier A, Connes P (2019) Impact of surface-area-to-volume ratio, internal viscosity and membrane viscoelasticity on red blood cell deformability measured in isotonic condition. Sci Rep 9(1):6771. https://doi.org/10.1038/s41598-019-43200-y
Robach P, Boisson RC, Vincent L, Lundby C, Moutereau S, Gergele L, Michel N, Duthil E, Feasson L, Millet GY (2014) Hemolysis induced by an extreme mountain ultra-marathon is not associated with a decrease in total red blood cell volume. Scand J Med Sci Sports 24(1):18–27. https://doi.org/10.1111/j.1600-0838.2012.01481.x
Smith JA, Telford RD, Kolbuch-Braddon M, Weidemann MJ (1997) Lactate/H + uptake by red blood cells during exercise alters their physical properties. Eur J Appl Physiol Occup Physiol 75(1):54–61
Strobaek D, Teuber L, Jorgensen TD, Ahring PK, Kjaer K, Hansen RS, Olesen SP, Christophersen P, Skaaning-Jensen B (2004) Activation of human IK and SK Ca2 + -activated K + channels by NS309 (6,7-dichloro-1H-indole-2,3-dione 3-oxime). Biochim Biophys Acta 1665(1–2):1–5. https://doi.org/10.1016/j.bbamem.2004.07.006
Suvorava T, Cortese-Krott MM (2018) Exercise-induced cardioprotection via eNOS: a putative role of red blood cell signaling. Curr Med Chem 25(34):4457–4474. https://doi.org/10.2174/0929867325666180307112557
Tomschi F, Bizjak D, Bloch W, Latsch J, Predel HG, Grau M (2018) Deformability of different red blood cell populations and viscosity of differently trained young men in response to intensive and moderate running. Clin Hemorheol Microcirc 69(4):503–514. https://doi.org/10.3233/CH-189202
Tripette J, Hardy-Dessources MD, Beltan E, Sanouiller A, Bangou J, Chalabi T, Chout R, Hedreville M, Broquere C, Nebor D, Dotzis G, Hue O, Connes P (2011) Endurance running trial in tropical environment: a blood rheological study. Clin Hemorheol Microcirc 47(4):261–268. https://doi.org/10.3233/CH-2011-1388
Tsai AG, Cabrales P, Intaglietta M (2005) Blood viscosity: a factor in tissue survival? Crit Care Med 33(7):1662–1663. https://doi.org/10.1097/01.ccm.0000170177.64555.db
Waltz X, Hardy-Dessources MD, Lemonne N, Mougenel D, Lalanne-Mistrih ML, Lamarre Y, Tarer V, Tressieres B, Etienne-Julan M, Hue O, Connes P (2015) Is there a relationship between the hematocrit-to-viscosity ratio and microvascular oxygenation in brain and muscle? Clin Hemorheol Microcirc 59(1):37–43. https://doi.org/10.3233/CH-131742
Zarychanski R, Schulz VP, Houston BL, Maksimova Y, Houston DS, Smith B, Rinehart J, Gallagher PG (2012) Mutations in the mechanotransduction protein PIEZO1 are associated with hereditary xerocytosis. Blood 120(9):1908–1915. https://doi.org/10.1182/blood-2012-04-422253
Funding
No funding was received for this study.
Author information
Authors and Affiliations
Contributions
All the authors approved the final version of the manuscript. EN, PC, DM, SE, MR, SS performed research, analyzed the data, and wrote the manuscript. ES, JH, and MG performed research. CR, PJ, AC, and AS edited the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
None of the authors have any conflict of interest.
Additional information
Communicated by Michael Lindinger.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Philippe Connes and Stéphane Égée are co-last author
Rights and permissions
About this article
Cite this article
Nader, E., Monedero, D., Robert, M. et al. Impact of a 10 km running trial on eryptosis, red blood cell rheology, and electrophysiology in endurance trained athletes: a pilot study. Eur J Appl Physiol 120, 255–266 (2020). https://doi.org/10.1007/s00421-019-04271-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-019-04271-x