Abstract
Purpose
The aim of this study was to follow post-activation potentiation (PAP), low-frequency fatigue (LFF), metabolic-induced fatigue and post-contractile depression (PCD) in response to different isometric muscle contraction modalities.
Methods
Young healthy men (N = 120) were randomly assigned to one of ten exercise modality groups which differed in contraction duration (5–60 s), activation pattern (intermittent or continuous contractions), activation mode (voluntary or stimulated), and intensity [maximal or submaximal (50%)]. Isometric maximal voluntary contraction (MVC), and electrically induced knee extension torque were measured at baseline and at regular intervals for 60 min after exercise.
Results
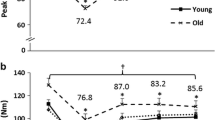
Muscle contraction modalities involving 5 s MVC were the most effective for PAP, whereas the lowest PAP effectiveness was found after the 12 × 5-MVC modality. After all of the 5–15 s MVC and 6 × 5-MVC protocols, the potentiation of the twitch rate was significantly higher than that recorded after continuous 30–60 s protocols (P < 0.001). Tetanic maximal torque (100 Hz) potentiation occurred 5 min after 15–30 s repetitive MVC modalities and after modality involving 15 electrical stimuli (P < 0.05).
Conclusions
The findings demonstrate that post-activation potentiation was most effective after brief duration continuous and repetitive MVC protocols. To understand the resultant warm-up of motor performance, it is necessary to recognize the coexistence of muscle PAP, tetanic maximal force potentiation, rapid recovery of metabolic muscle, and central muscle activation processes, as well as prolonged LFF and prolonged PCD.


Similar content being viewed by others
Abbreviations
- ANOVA:
-
Analysis of variance
- CT:
-
Contraction time
- ES:
-
Electrical stimulation
- LFF:
-
Low-frequency fatigue
- MC:
-
Muscle conditioning protocol
- MVC:
-
Maximal voluntary contraction
- P100:
-
100 Hz electrically induced peak torque
- P20:
-
20 Hz electrically induced peak torque
- PAP:
-
Post-activation potentiation
- PCD:
-
Post-contractile depression
- Pt:
-
Electrically induced twitch peak torque
- Pt/CT:
-
Twitch contraction rate
- Pt/RT½:
-
Twitch half-relaxation rate
- RT½:
-
Half-relaxation time
References
Allen DG, Lamb GD, Westerblad H (2008) Skeletal muscle fatigue: cellular mechanisms. Physiol Rev 88:287–332. https://doi.org/10.1152/physrev.00015.2007
Andrews SK, Horodyski JM, MacLeod DA, Whitten J, Behm DG (2016) The interaction of fatigue and potentiation following an acute bout of unilateral squats. J Sports Sci Med 15:625–632
Bogdanis GC, Tsoukos A, Veligekas P, Tsolakis C, Terzis G (2014) Effects of muscle action type with equal impulse of conditioning activity on postactivation potentiation. J Strength Cond Res 28:2521–2528. https://doi.org/10.1519/JSC.0000000000000444
Brazaitis M, Skurvydas A, Pukėnas K, Daniuseviciūtė L, Mickevicienė D, Solianik R (2012) The effect of temperature on amount and structure of motor variability during 2-minute maximum voluntary contraction. Muscle Nerve 46:799–809. https://doi.org/10.1002/mus.23397
Bruton JD, Wretman C, Katz A, Westerblad H (1997) Increased tetanic force and reduced myoplasmic [Pi] following a brief series of tetani in mouse soleus muscle. Am J Physiol 272:870–874. https://doi.org/10.1152/ajpcell.1997.272.3.C870
Buchanan R, Nielsen OB, Clausen T (2002) Excitation- and beta(2)-agonist induced activation of the Na(+)-K(+) pump in rat soleus muscle. J Physiol 545:229–240
Cheng AJ, Yamada T, Rassier DE, Andersson DC, Westerblad H, Lanner JT (2016) Reactive oxygen/nitrogen species and contractile function in skeletal muscle during fatigue and recovery. J Physiol 594:5149–5160. https://doi.org/10.1113/JP270650
Cheng AJ, Neyroud D, Kayser B, Westerblad H, Place N (2017) Intramuscular contributions to low-frequency force potentiation induced by a high-frequency conditioning stimulation. Front Physiol 8:712. https://doi.org/10.3389/fphys.2017.00712
Esformes JI, Keenan M, Moody J, Bampouras TM (2011) Effect of different types of conditioning contraction on upper body postactivation potentiation. J Strength Cond Res 25:143–148. https://doi.org/10.1519/JSC.0b013e3181fef7f3
Gittings W, Bunda J, Vandenboom R (2017) Myosin phosphorylation potentiated steady state work output without altering contractile economy of mouse fast skeletal muscles. J Exp Biol 221:jeb167742. https://doi.org/10.1242/jeb.167742
Gouvêa AL, Fernandes IA, César EP, Silva WA, Gomes PS (2013) The effects of rest intervals on jumping performance: a meta-analysis on post-activation potentiation studies. J Sports Sci 31:459–467. https://doi.org/10.1080/02640414.2012.738924
Hicks A, McComas AJ (1989) Increased sodium pump activity following repetitive stimulation of rat soleus muscles. J Physiol 414:337–349
Jones DA (1996) High-and low-frequency fatigue revisited. Acta Physiol Scand 156:265–270. https://doi.org/10.1046/j.1365-201X.1996.192000.x
Kamandulis S, Skurvydas A, Masiulis N, Mamkus G, Westerblad H (2010) The decrease in electrically evoked force production is delayed by a previous bout of stretch-shortening cycle exercise. Acta Physiol (Oxf) 198:91–98. https://doi.org/10.1111/j.1748-1716.2009.02041.x
Kent-Braun JA (1999) Central and peripheral contributions to muscle fatigue in humans during sustained maximal effort. Eur J Appl Physiol Occup Physiol 80:57–63. https://doi.org/10.1007/s004210050558
Kilduff LP, Cunningham DJ, Owen NJ, West DJ, Bracken RM, Cook CJ (2011) Effect of postactivation potentiation on swimming starts in international sprint swimmers. J Strength Cond Res 25:2418–2423. https://doi.org/10.1519/JSC.0b013e318201bf7a
Lännergren J, Larsson L, Westerblad H (1989) A novel type of delayed tension reduction observed in rat motor units after intense activity. J Physiol 412:267–276
Macintosh BR, Robillard ME, Tomaras EK (2012) Should postactivation potentiation be the goal of your warm-up? Appl Physiol Nutr Metab 37:546–550. https://doi.org/10.1139/h2012-016
Miyamoto N, Kanehisa H, Kawakami Y (2012) Potentiation of maximal voluntary concentric torque in human quadriceps femoris. Med Sci Sports Exerc 44:1738–1746. https://doi.org/10.1249/MSS.0b013e318256b813
Miyamoto N, Wakahara T, Ema R, Kawakami Y (2013) Further potentiation of dynamic muscle strength after resistance training. Med Sci Sports Exerc 45:1323–1330. https://doi.org/10.1249/MSS.0b013e3182874c0e
Needham RA, Morse CI, Degens H (2009) The acute effect of different warm-up protocols on anaerobic performance in elite youth soccer players. J Strength Cond Res 23:2614–2620. https://doi.org/10.1519/JSC.0b013e3181b1f3ef
Nibali ML, Chapman DW, Robergs RA, Drinkwater EJ (2015) Considerations for determining the time course of post-activation potentiation. Appl Physiol Nutr Metab 40:1163–1170. https://doi.org/10.1139/apnm-2015-0175
Place N, Ivarsson N, Venckunas T, Neyroud D, Brazaitis M, Cheng AJ et al (2015) Ryanodine receptor fragmentation and sarcoplasmic reticulum Ca2+ leak after one session of high-intensity interval exercise. Proc Natl Acad Sci USA 112:15492–15497. https://doi.org/10.1073/pnas.1507176112
Rassier DE, Macintosh BR (2000) Coexistence of potentiation and fatigue in skeletal muscle. Braz J Med Biol Res 33:499–508. https://doi.org/10.1590/S0100-879X2000000500003
Requena B, Gapeyeva H, García I, Ereline J, Pääsuke M (2008) Twitch potentiation after voluntary versus electrically induced isometric contractions in human knee extensor muscles. Eur J Appl Physiol 104:463–472. https://doi.org/10.1007/s00421-008-0793-8
Rodriguez-Falces J, Place N (2017) New insights into the potentiation of the first and second phases of the M-wave after voluntary contractions in the quadriceps muscle. Muscle Nerve 55:35–45. https://doi.org/10.1002/mus.25186
Sale DG (2002) Postactivation potentiation: role in human performance. Exerc Sport Sci Rev 30:138–143
Sasaki K, Tomioka Y, Ishii N (2012) Activation of fast-twitch fibers assessed with twitch potentiation. Muscle Nerve 46:218–227. https://doi.org/10.1002/mus.23290
Seitz LB, Haff GG (2016) Factors modulating post-activation potentiation of jump, sprint, throw, and upper-body ballistic performances: a systematic review with meta-analysis. Sports Med 46:231–240. https://doi.org/10.1007/s40279-015-0415-7
Shushakov V, Stubbe C, Peuckert A, Endeward V, Maassen N (2007) The relationships between plasma potassium, muscle excitability and fatigue during voluntary exercise in humans. Exp Physiol 92:705–715. https://doi.org/10.1113/expphysiol.2006.036384
Skurvydas A, Zachovajevas P (1998) Is post-tetanic potentiation, low frequency fatigue (LFF) and pre-contractile depression (PCD) coexistent in intermittent isometric exercises of maximal intensity? Acta Physiol Scand 164:127–133. https://doi.org/10.1046/j.1365-201X.1998.00415.x
Skurvydas A, Mamkus G, Kamandulis S, Dudoniene V, Valanciene D, Westerblad H (2016) Mechanisms of force depression caused by different types of physical exercise studied by direct electrical stimulation of human quadriceps muscle. Eur J Appl Physiol 116:2215–2224. https://doi.org/10.1007/s00421-016-3473-0
Smith IC, Vandenboom R, Tupling AR (2017) Contraction-induced enhancement of relaxation during high force contractions of mouse lumbrical muscle at 37 °C. J Exp Biol 220:2870–2873. https://doi.org/10.1242/jeb.158998
Stull JT, Kamm KE, Vandenboom R (2011) Myosin light chain kinase and the role of myosin light chain phosphorylation in skeletal muscle. Arch Biochem Biophys 510:120–128. https://doi.org/10.1016/j.abb.2011.01.017
Thomas K, Toward A, West DJ, Howatson G, Goodall S (2017) Heavy-resistance exercise-induced increases in jump performance are not explained by changes in neuromuscular function. Scand J Med Sci Sports 27:35–44. https://doi.org/10.1111/sms.12626
Tillin NA, Bishop D (2009) Factors modulating post-activation potentiation and its effect on performance of subsequent explosive activities. Sports Med 39:147–166. https://doi.org/10.2165/00007256-200939020-00004
Vandenboom R (2016) Modulation of skeletal muscle contraction by myosin phosphorylation. Compr Physiol 7:171–212. https://doi.org/10.1002/cphy.c150044
Vandenboom R, Gittings W, Smith IC, Grange RW, Stull JT (2013) Myosin phosphorylation and force potentiation in skeletal muscle: evidence from animal models. J Muscle Res Cell Motil 34:317–332. https://doi.org/10.1007/s10974-013-9363-8
Verbickas V, Baranauskiene N, Eimantas N, Kamandulis S, Rutkauskas S, Satkunskiene D et al (2017) Effect of sprint cycling and stretch-shortening cycle exercises on the neuromuscular, immune and stress indicators in young men. J Physiol Pharmacol 68:125–132
Verbickas V, Kamandulis S, Snieckus A, Venckunas T, Baranauskiene N, Brazaitis M et al (2018) Serum brain-derived neurotrophic factor and interleukin-6 response to high-volume mechanically demanding exercise. Muscle Nerve 57:E46–E51. https://doi.org/10.1002/mus.25687
Westerblad H, Lännergren J (1986) Force and membrane potential during and after fatiguing, intermittent tetanic stimulation of single Xenopus muscle fibres. Acta Physiol Scand 128:369–378. https://doi.org/10.1111/j.1748-1716.1986.tb07990.x
Wilson JM, Duncan NM, Marin PJ, Brown LE, Loenneke JP, Wilson SM et al (2013) Meta-analysis of postactivation potentiation and power: effects of conditioning activity, volume, gender, rest periods, and training status. J Strength Cond Res 27:854–859. https://doi.org/10.1519/JSC.0b013e31825c2bdb
Xenofondos A, Bassa E, Vrabas IS, Kotzamanidis C, Patikas DA (2018) Muscle twitch torque during two different in volume isometric exercise protocols: fatigue effects on post-activation potentiation. J Strength Cond Res 32:578–586. https://doi.org/10.1519/JSC.0000000000002311
Author information
Authors and Affiliations
Contributions
AS and DM: conception and design of the study, AS, GJ, SK, DM, MB, DV, DK, MM and GM: data collection and analyses, AS: wrote the manuscript. All authors edited and approved the manuscript final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Additional information
Communicated by Philip D. Chilibeck.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Skurvydas, A., Jurgelaitiene, G., Kamandulis, S. et al. What are the best isometric exercises of muscle potentiation?. Eur J Appl Physiol 119, 1029–1039 (2019). https://doi.org/10.1007/s00421-019-04092-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-019-04092-y