Abstract
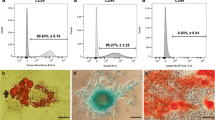
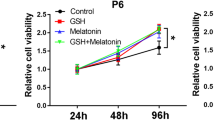
Mesenchymal stem cells (MSCs) are multipotent cells that have the ability to self-renew and regulate paracrine signalling and immune system processes. MSCs have extensive clinical applications in regeneration, functional reconstruction and cellular therapies. However, studies are needed to discover ways to improve the properties of MSCs, such as differentiation, and prevent senescence in culture, which are both very important for cell therapies. Royal jelly (RJ) is a nutritional substance produced by worker bees that contains a substantial amounts of proteins that are beneficial for cell growth and proliferation. RJ is widely used in traditional medicine today, and due to the specific components in its content, it has been reported to have antioxidant, antiproliferative, antimicrobial, neuroprotective, anti-inflammatory, immunomodulatory and anti-ageing properties. In our study, human Wharton’s jelly mesenchymal stem cells (WJ-MSCs) derived from umbilical cord matrix were grown in culture medium supplemented with RJ. The control group comprised minimum essential medium (MEM) and 10% foetal bovine serum (FBS); RJ groups were formed using MEM, 10% FBS and 0.075 mg/ml or 0.150 mg/ml RJ. In our study, we evaluated the effect of RJ on WJ-MSC growth by MTT assay, proliferating cell nuclear antigen ELISA, β-galactosidase activity assay, MitoTracker Green staining and differentiation tests in adipogenic, osteogenic and chondrogenic cell lines. It was observed that the number of mitochondria increased, senescence decreased and osteogenic differentiation increased after differentiation induction after the addition of RJ to MSC culture. In general, the results of this study indicate that WJ-MSCs enhance mitochondrial numbers and important cellular activities, such as antisenescence and osteogenic differentiation, and with increasing evidence from further studies, RJ supplementation may be found beneficial for the use of MSCs in bone engineering regenerative medicine or cell therapy.






Similar content being viewed by others
Data availability
All data generated or analysed during this study are included in this published article.
References
Al-Azab M, Safi M, Idiiatullina E, Al-Shaebi F et al (2022) Aging of mesenchymal stem cell: machinery, markers, and strategies of fighting. Cell Mol Biol Lett 27(1):69. https://doi.org/10.1186/s11658-022-00366-0
Alves-Paiva RM, Nascimento Sd, Oliveira Dd, Larissa C et al (2022) Senescence state in mesenchymal stem cells at low passages: implications in clinical use. Front Cell Dev Biol 10:858996. https://doi.org/10.3389/fcell.2022.858996
Bielak-Zmijewska A, Wnuk M, Przybylska D, Grabowska W et al (2014) A comparison of replicative senescence and doxorubicin-induced premature senescence of vascular smooth muscle cells isolated from human aorta. Biogerontology 15(1):47–64. https://doi.org/10.1007/s10522-013-9477-9
Chazotte B (2011) Labeling mitochondria with MitoTracker dyes. Cold Spring Harb Protoc 8:990–992. https://doi.org/10.1101/pdb.prot5648
Chen D, Xin XX, Qian HC, Yu ZY, Shen LR (2016) Evaluation of the major royal jelly proteins as an alternative to fetal bovine serum in culturing human cell lines. J Zhejiang Univ Sci B 17(6):476–483. https://doi.org/10.1631/jzus.B1500295
Cheraghi O, Abdollahpourasl M, Rezabakhsh A, Rahbarghazi R (2018) Distinct effects of royal jelly on human endothelial cells under high glucose condition. Iran J Pharm Res 17(4):1361–1370
Dominici M, Le Blanc K, Mueller I et al (2006) Minimal criteria for defining multipotent mesenchymal stromal cells The International society for cellular therapy position statement. Cytotherapy 8(4):315–317. https://doi.org/10.1080/14653240600855905
Fehrer C, Lepperdinger G (2005) Mesenchymal stem cell aging. Exp Gerontol 40(12):926–930. https://doi.org/10.1016/j.exger.2005.07.006
Ghasemi M, Turnbull T, Sebastian S, Kempson I (2021) The MTT Assay: utility, limitations, pitfalls, and interpretation in bulk and single-cell analysis. Int J Mol Sci. https://doi.org/10.3390/ijms222312827
Han Y, Li X, Zhang Y et al (2019) Mesenchymal stem cells for regenerative medicine. Cells 8(8):886. https://doi.org/10.3390/cells8080886
Hashem KS, Elkelawy A, Abd-Allah S, Helmy NA (2020) Involvement of Mfn2, Bcl2/Bax signaling and mitochondrial viability in the potential protective effect of Royal jelly against mitochondria-mediated ovarian apoptosis by cisplatin in rats. Iran J Basic Med Sci 23(4):515–526. https://doi.org/10.22038/ijbms.2020.40401.9563
Hayashi T, Takamatsu N, Nakashima T, Arita T (2011) Immunological characterization of honey proteins and identification of MRJP 1 as an IgE-binding protein. Biosci Biotechnol Biochem 75(3):556–560. https://doi.org/10.1271/bbb.100778
Izuta H, Chikaraishi Y, Shimazawa M, Mishima S et al (2009) 10-Hydroxy-2-decenoic acid, a major fatty acid from royal jelly, inhibits VEGF-induced angiogenesis in human umbilical vein endothelial cells. Evid Based Complement Alternat Med 6(4):489–494. https://doi.org/10.1093/ecam/nem152
Jiang C, Liu MX, Li CX, Qian HC et al (2018) Anti-senescence effect and molecular mechanism of the major royal jelly proteins on human embryonic lung fibroblast (HFL-I) cell line. J Zhejiang Univ Sci B 19(12):960–972. https://doi.org/10.1631/jzus.B1800257
Ksiazek K (2009) A comprehensive review on mesenchymal stem cell growth and senescence. Rejuvenation Res 12(2):105–116. https://doi.org/10.1089/rej.2009.0830
Kunugi H, Mohammed Ali A (2019) Royal jelly and its components promote healthy aging and longevity: From animal models to humans. Int J Mol Sci 20(19):4662. https://doi.org/10.3390/ijms20194662
Liao L, Chen ZZC, Wang SS, Liu WB et al (2021) NLRP3 inflammasome activation contributes to the pathogenesis of cardiocytes aging. Aging 13(16):20534–20551. https://doi.org/10.18632/aging.203435
Mehta KJ (2022) Iron oxide nanoparticles in mesenchymal stem cell detection and therapy. Stem Cell Rev Rep 18(7):2234–2261. https://doi.org/10.1007/s12015-022-10343-x
Mollentze J, Durandt C, Pepper MS (2021) An in vitro and in vivo comparison of osteogenic differentiation of human mesenchymal stromal/stem cells. Stem Cells Int 2021:9919361. https://doi.org/10.1155/2021/9919361
Münstedt K, Bargello M, Hauenschild A (2009) Royal jelly reduces the serum glucose levels in healthy subjects. J Med Food 12(5):1170–1172. https://doi.org/10.1089/jmf.2008.0289
Musa M, Nasir NF, Thirumulu KP (2014) Evaluation of royal jelly as an alternative to fetal bovine serum in cell culture using cell proliferation assays and live cell imaging. Afr J Tradit Complement Altern Med. 11(1):148–155. https://doi.org/10.4314/ajtcam.v11i1.23
Özan F, Çörekçi B, Toptaş O, Halicioğlu K et al (2015) Effect of Royal Jelly on new bone formation in rapid maxillary expansion in rats. Med Oral Patol Oral Cir Bucal. 20(6):e651-656. https://doi.org/10.4317/medoral.20581
Perminaite K, Marksa M, Stančiauskaitė M, Juknius T et al (2021) Formulation of ocular in situ gels with lithuanian royal jelly and their biopharmaceutical evaluation in vitro. Molecules. 26(12):3552. https://doi.org/10.3390/molecules26123552
Presley AD, Fuller KM, Arriaga EA (2003) MITOTRACKER green labeling of mitochondrial proteins and their subsequent analysis by capillary electrophoresis with laser-induced fluorescence detection. J Chromatogr B Analyt Technol Biomed Life Sci. 793(1):141–150. https://doi.org/10.1016/s1570-0232(03)00371-4
Song E, Gao Y, Wu C, Shi T et al (2017) Targeted proteomic assays for quantitation of proteins identified by proteogenomic analysis of ovarian cancer. Sci Data 4:170091. https://doi.org/10.1038/sdata.2017.91
Viuda-Martos M, Ruiz-Navajas Y, Fernández-López J, Pérez-Alvarez JA (2008) Functional properties of honey, propolis, and royal jelly. J Food Sci. 73(9):R117-124. https://doi.org/10.1111/j.1750-3841.2008.00966.x
Wan DC, Morgan SL, Spencley AL, Mariano N et al (2018) Honey bee royalactin unlocks conserved pluripotency pathway in mammals. Nat Commun 9(1):5078. https://doi.org/10.1038/s41467-018-06256-4
Xiao B, Deng X, Zhou W, Tan EK (2016) Flow cytometry-based assessment of mitophagy using mitotracker.”. Front Cell Neurosci 10:76. https://doi.org/10.3389/fncel.2016.00076
Xu Y, Tian Y, Tong D, Zhang H et al (2020) Wnt signaling inhibits high-density cell sheet culture induced mesenchymal stromal cell aging by targeting cell cycle inhibitor p27. Front Bioeng Biotechnol 8:946. https://doi.org/10.3389/fbioe.2020.00946
Yan W, Diao S, Fan Z (2021) The role and mechanism of mitochondrial functions and energy metabolism in the function regulation of the mesenchymal stem cells. Stem Cell Res Ther. 12(1):140. https://doi.org/10.1186/s13287-021-02194-z
Acknowledgements
This research was supported by the Necmettin Erbakan University Scientific Research Unit (project number: 221218010).
Author information
Authors and Affiliations
Contributions
GÇ: conception or design of the work, the acquisition of data, drafted the work, reviewed it critically for important intellectual content, the version to be published. FÖB: acquisition of data, analysis of data, critical revision for important intellectual content and interpretation of data.
Corresponding author
Ethics declarations
Conflict of interest
We declare that there is no conflict of interest between authors and that all authors have read and submitted the manuscript.
Ethical approval
The study was approved by the Local Ethical Review Committee (protocol number: 2022/3900-10733) in accordance with the World Medical Association Declaration of Helsinki Ethical Principles for Medical Research Involving Human Subjects and Samples.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Çiçek, G., Öz Bağcı, F. Effects of royal jelly on the antisenescence, mitochondrial viability and osteogenic differentiation capacity of umbilical cord-derived mesenchymal stem cells. Histochem Cell Biol 161, 183–193 (2024). https://doi.org/10.1007/s00418-023-02243-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-023-02243-z