Abstract
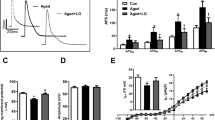
l-Carnitine (β-hydroxy-γ-trimethylaminobutyric acid, LC) is a crucial molecule for the mitochondrial oxidation of fatty acids. It facilitates the transport of long-chain fatty acids into the mitochondrial matrix. The reduction in LC levels during the aging process has been linked to numerous cardiovascular disorders, including contractility dysfunction, and disrupted intracellular Ca2+ homeostasis. The aim of this study was to examine the effects of long-term (7 months) LC administration on cardiomyocyte contraction and intracellular Ca2+ transients ([Ca2+]i) in aging rats. Male albino Wistar rats were randomly assigned to either the control or LC-treated groups. LC (50 mg/kg body weight/day) was dissolved in distilled water and orally administered for a period of 7 months. The control group received distilled water alone. Subsequently, ventricular single cardiomyocytes were isolated, and the contractility and Ca2+ transients were recorded in aging (18 months) rats. This study demonstrates, for the first time, a novel inotropic effect of long-term LC treatment on rat ventricular cardiomyocyte contraction. LC increased cardiomyocyte cell shortening and resting sarcomere length. Furthermore, LC supplementation led to a reduction in resting [Ca2+]i level and an increase in the amplitude of [Ca2+]i transients, indicative of enhanced contraction. Consistent with these results, decay time of Ca2+ transients also decreased significantly in the LC-treated group. The long-term administration of LC may help restore the Ca2+ homeostasis altered during aging and could be used as a cardioprotective medication in cases where myocyte contractility is diminished.



Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author by reasonable request.
References
Bers DM (2002) Cardiac excitation–contraction coupling. Nature 415:198–205. https://doi.org/10.1038/415198a
Ceylan-Isik AF, Dong M, Zhang Y et al (2013) RETRACTED ARTICLE: cardiomyocyte-specific deletion of endothelin receptor A rescues aging-associated cardiac hypertrophy and contractile dysfunction: role of autophagy. Basic Res Cardiol 108:335. https://doi.org/10.1007/s00395-013-0335-3
Cheng H, Lederer WJ (2008) Calcium sparks. Physiol Rev 88:1491–1545. https://doi.org/10.1152/physrev.00030.2007
Chiao YA, Zhang H, Sweetwyne M et al (2020) Late-life restoration of mitochondrial function reverses cardiac dysfunction in old mice. Elife 9:1–26. https://doi.org/10.7554/eLife.55513
Domeier TL, Roberts CJ, Gibson AK et al (2014) Dantrolene suppresses spontaneous Ca2+ release without altering excitation-contraction coupling in cardiomyocytes of aged mice. Am J Physiol Circ Physiol 307:H818–H829. https://doi.org/10.1152/ajpheart.00287.2014
Dong M, Yang Z, Fang H et al (2020) Aging attenuates cardiac contractility and affects therapeutic consequences for myocardial infarction. Aging Dis 11:365. https://doi.org/10.14336/AD.2019.0522
Emran T, Chowdhury NI, Sarker M et al (2021) L-carnitine protects cardiac damage by reducing oxidative stress and inflammatory response via inhibition of tumor necrosis factor-alpha and interleukin-1beta against isoproterenol-induced myocardial infarction. Biomed Pharmacother 143:112139. https://doi.org/10.1016/j.biopha.2021.112139
Fan Z, Han Y, Ye Y et al (2017) L-carnitine preserves cardiac function by activating p38 MAPK/Nrf2 signalling in hearts exposed to irradiation. Eur J Pharmacol 804:7–12. https://doi.org/10.1016/j.ejphar.2017.04.003
Farrell SR, Howlett SE (2007) The effects of isoproterenol on abnormal electrical and contractile activity and diastolic calcium are attenuated in myocytes from aged Fischer 344 rats. Mech Ageing Dev 128:566–573. https://doi.org/10.1016/j.mad.2007.08.003
Gnoni A, Longo S, Gnoni GV, Giudetti AM (2020) Carnitine in human muscle bioenergetics: can carnitine supplementation improve physical exercise? Molecules 25:182. https://doi.org/10.3390/molecules25010182
Grandy SA, Howlett SE (2006) Cardiac excitation-contraction coupling is altered in myocytes from aged male mice but not in cells from aged female mice. Am J Physiol Circ Physiol 291:H2362–H2370. https://doi.org/10.1152/ajpheart.00070.2006
Hamilton S, Terentyev D (2019) Altered intracellular calcium homeostasis and arrhythmogenesis in the aged heart. Int J Mol Sci 20:2386. https://doi.org/10.3390/ijms20102386
Herraiz-Martínez A, Álvarez-García J, Llach A et al (2015) Ageing is associated with deterioration of calcium homeostasis in isolated human right atrial myocytes. Cardiovasc Res 106:76–86. https://doi.org/10.1093/cvr/cvv046
Howlett SE (2010) Age-associated changes in excitation-contraction coupling are more prominent in ventricular myocytes from male rats than in myocytes from female rats. Am J Physiol Circ Physiol 298:H659–H670. https://doi.org/10.1152/ajpheart.00214.2009
Høydal MA, Kirkeby-Garstad I, Karevold A et al (2018) Human cardiomyocyte calcium handling and transverse tubules in mid-stage of post-myocardial-infarction heart failure. ESC Hear Fail 5:332–342. https://doi.org/10.1002/ehf2.12271
Janczewski AM, Lakatta EG (2010) Modulation of sarcoplasmic reticulum Ca(2+) cycling in systolic and diastolic heart failure associated with aging. Heart Fail Rev 15:431–445. https://doi.org/10.1007/s10741-010-9167-5
Khedr NF, El-Feky OA, Werida RH (2022) l-Carnitine mitigates trazadone induced rat cardiotoxicity mediated via modulation of autophagy and oxidative stress. Cardiovasc Toxicol 22:831–841. https://doi.org/10.1007/s12012-022-09759-1
Kiyuna LA, Albuquerque RP et al (2018) Targeting mitochondrial dysfunction and oxidative stress in heart failure: challenges and opportunities. Free Radic Biol Med 129:155–168
Lakatta EG (2003) Arterial and cardiac aging: major shareholders in cardiovascular disease enterprises. Circulation 107:490–497. https://doi.org/10.1161/01.CIR.0000048894.99865.02
Li M, Xu S, Geng Y et al (2019) The protective effects of L-carnitine on myocardial ischaemia–reperfusion injury in patients with rheumatic valvular heart disease undergoing CPB surgery are associated with the suppression of NF-κB pathway and the activation of Nrf. Clin Exp Pharmacol Physiol 46:1001–1012. https://doi.org/10.1111/1440-1681.13155
Lim CC, Apstein CS, Colucci WS, Liao R (2000) Impaired cell shortening and relengthening with increased pacing frequency are intrinsic to the senescent mouse cardiomyocyte. J Mol Cell Cardiol 32:2075–2082. https://doi.org/10.1006/jmcc.2000.1239
Mellor KM, Curl CL, Chandramouli C et al (2014) Ageing-related cardiomyocyte functional decline is sex and angiotensin II dependent. Age (Dordr) 36:9630. https://doi.org/10.1007/s11357-014-9630-7
Oyanagi E, Yano H, Uchida M et al (2011) Protective action of l-carnitine on cardiac mitochondrial function and structure against fatty acid stress. Biochem Biophys Res Commun 412:61–67. https://doi.org/10.1016/j.bbrc.2011.07.039
Qin F, Siwik DA, Lancel S et al (2013) Hydrogen peroxide-mediated SERCA cysteine 674 oxidation contributes to impaired cardiac myocyte relaxation in senescent mouse heart. J Am Heart Assoc 2:e000184. https://doi.org/10.1161/JAHA.113.000184
Rueckschloss U, Villmow M, Klöckner U (2010) NADPH oxidase-derived superoxide impairs calcium transients and contraction in aged murine ventricular myocytes. Exp Gerontol 45:788–796. https://doi.org/10.1016/j.exger.2010.05.002
Sawicka A, Hartmane D, Lipinska P et al (2018) l-Carnitine supplementation in older women a pilot study on aging skeletal muscle mass and function. Nutrients 10:255. https://doi.org/10.3390/nu10020255
Steenman M, Lande G (2017) Cardiac aging and heart disease in humans. Biophys Rev 9:131–137. https://doi.org/10.1007/s12551-017-0255-9
Stowe DF, Camara AKS (2009) Mitochondrial reactive oxygen species production in excitable cells: modulators of mitochondrial and cell function. Antioxid Redox Signal 11:1373–1414. https://doi.org/10.1089/ars.2008.2331
Swift LM, Burke M, Guerrelli D et al (2020) Age-dependent changes in electrophysiology and calcium handling: implications for pediatric cardiac research. Am J Physiol Circ Physiol 318:H354–H365. https://doi.org/10.1152/ajpheart.00521.2019
Turdi S, Fan X, Li J et al (2010) AMP-activated protein kinase deficiency exacerbates aging-induced myocardial contractile dysfunction. Aging Cell 9:592–606. https://doi.org/10.1111/j.1474-9726.2010.00586.x
Weisser-Thomas J, Nguyen Q, Schuettel M et al (2007) Age and hypertrophy related changes in contractile post-rest behavior and action potential properties in isolated rat myocytes. Age (Dordr) 29:205–217. https://doi.org/10.1007/s11357-007-9040-1
Xu H, Ahn B, Van Remmen H (2022) Impact of aging and oxidative stress on specific components of excitation contraction coupling in regulating force generation. Sci Adv 8:7377. https://doi.org/10.1126/sciadv.add7377
Zhu X, Altschafl BA, Hajjar RJ et al (2005) Altered Ca2+ sparks and gating properties of ryanodine receptors in aging cardiomyocytes. Cell Cal 37:583–591. https://doi.org/10.1016/j.ceca.2005.03.002
Funding
This study was supported by Akdeniz University Research Coordination Unit. (1347/2021.09.002).
Author information
Authors and Affiliations
Contributions
Research concept and design: NY, ND, YG; Supervision: ND, NY; Capturing and housing the animals: BD and GA; Experimental process and analyses: YG, BD, GA; Literature: BD, GA, YG; Writing the article: YG, ND, and NY.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Gökçe, Y., Danisman, B., Akcay, G. et al. l-Carnitine improves mechanical responses of cardiomyocytes and restores Ca2+ homeostasis during aging. Histochem Cell Biol 160, 341–347 (2023). https://doi.org/10.1007/s00418-023-02215-3
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-023-02215-3