Abstract
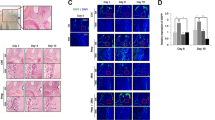
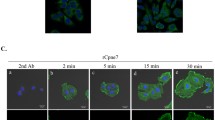
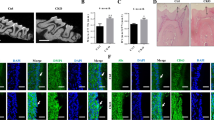
We hypothesized that odontoblasts release exosomes as well as dental pulp cells and focused on the exosome membrane marker CD63. Odontoblasts are well-differentiated mesenchymal cells that produce dentin. Dental pulp, a tissue complex formed with odontoblasts, releases exosomes to epithelial cells and stimulates their differentiation to ameloblasts. However, the localization of CD63 in differentiated odontoblasts is poorly understood. Therefore, herein, we aimed to reveal the expression of CD63 in odontoblasts during tooth development. We first investigated the localization of CD63 in mouse incisors and molars using immunofluorescence. In adult mouse incisors, the anti-CD63 antibody was positive in mature odontoblasts and dental pulp cells but not in pre-odontoblasts along the ameloblasts in the apical bud. Additionally, the anti-CD63 antibody was observed as a vesicular shape in the apical area of odontoblast cytosol and inside Tomes’ fibers. The anti-CD63 antibody-positive vesicles were also observed using immunoelectron microscopy. Moreover, during mouse mandibular molar tooth morphogenesis (E16 to postnatal 6 weeks), labeling of anti-CD63 antibody was positive in the odontoblasts at E18. In contrast, the anti-CD63 antibody was positive in the dental pulp after postnatal day 10. Furthermore, anti-CD63 antibody was merged with the multivesicular body marker Rab7 in dental pulp tissues but not with the lysosome marker Lamp1. Finally, we determined the effect of a ceramide-generation inhibitor GW4869 on the mouse organ culture of tooth germ in vitro. After 28 days of GW4869 treatment, both CD63 and Rab7 were negative in Tomes’ fibers, but were positive in control odontoblasts. These results suggest that CD63-positive vesicular organelles are important for mouse tooth morphogenesis.






Similar content being viewed by others
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Catalano M, O’Driscoll L (2020) Inhibiting extracellular vesicles formation and release: a review of EV inhibitors. J Extracell Vesicles 9:1703244. https://doi.org/10.1080/20013078.2019.1703244
Chaudhary SC, Kuzynski M, Bottini M et al (2016) Phosphate induces formation of matrix vesicles during odontoblast-initiated mineralization in vitro. Matrix Biol 52:284–300. https://doi.org/10.1016/j.matbio.2016.02.003
Chen L, Feng Z, Yue H et al (2018) Exosomes derived from HIV-1-infected cells promote growth and progression of cancer via HIV TAR RNA. Nat Commun 9:4585. https://doi.org/10.1038/s41467-018-07006-2
Gaete M, Lobos N, Torres-Quintana MA (2004) Mouse tooth development time sequence determination for the ICR/Jcl strain. J Oral Sci 46:135–141. https://doi.org/10.2334/josnusd.46.135
Garant PR, Szabo G, Nalbandian J (1968) The fine structure of the mouse odontoblast. Arch Oral Biol 13:857–876. https://doi.org/10.1016/0003-9969(68)90002-2
Goldberg M, Kulkarni AB, Young M, Boskey A (2011) Dentin: structure, composition and mineralization: the role of dentin ECM in dentin formation and mineralization. Front Biosci (Elit Ed) 3:711–735. https://doi.org/10.2741/e281
Gori A, Romanato A, Greta B et al (2020) Membrane-binding peptides for extracellular vesicles on-chip analysis. J Extracell Vesicles 9:1751428. https://doi.org/10.1080/20013078.2020.1751428
Huotari J, Helenius A (2011) Endosome maturation. EMBO J 30:3481–3500. https://doi.org/10.1080/20013078.2020.1751428
Joyner CJ, Bennett A, Triffitt JT (1997) Identification and enrichment of human osteoprogenitor cells by using differentiation stage-specific monoclonal antibodies. Bone 21:1–6. https://doi.org/10.1016/S8756-3282(97)00074-4
Mahdee A, Eastham J, Whitworth JM, Gillespie JI (2018) Evidence for programmed odontoblast process retraction after dentine exposure in the rat incisor. Arch Oral Biol 85:130–141. https://doi.org/10.1016/j.archoralbio.2017.10.001
Massa LF, Ramachandran A, George A, Arana-Chavez VE (2005) Developmental appearance of dentin matrix protein 1 during the early dentinogenesis in rat molars as identified by high-resolution immunocytochemistry. Histochem Cell Biol 124:197–205. https://doi.org/10.1007/s00418-005-0009-9
Menck K, Sönmezer C, Worst TS et al (2017) Neutral sphingomyelinases control extracellular vesicles budding from the plasma membrane. J Extracell Vesicles 6:1378056. https://doi.org/10.1080/20013078.2017.1378056
Nanci A, Fortin M, Ghitescu L (1996) Endocytotic functions of ameloblasts and odontoblasts: immunocytochemical and tracer studies on the uptake of plasma proteins. Anat Rec 245:219–234. https://doi.org/10.1002/(SICI)1097-0185(199606)245:2%3c219::AID-AR9%3e3.0.CO;2-R
Nieuwenhuis HK, Van Oosterhout JJG, Rozemuller E et al (1987) Studies with a monoclonal antibody against activated platelets: evidence that a secreted 53,000-molecular weight lysosome-like granule protein is exposed on the surface of activated platelets in the circulation. Blood 70:838–845. https://doi.org/10.1182/blood.v70.3.838.838
Nydegger S, Khurana S, Krementsov DN et al (2006) Mapping of tetraspanin-enriched microdomains that can function as gateways for HIV-1. J Cell Biol 173:795–807. https://doi.org/10.1083/jcb.200508165
Ohki R, Matsuki-Fukushima M, Fujikawa K et al (2021) In the absence of a basal lamina, ameloblasts absorb enamel in a serumless and chemically defined organ culture system. J Oral Biosci 63:66–73. https://doi.org/10.1016/j.job.2020.12.004
Orsini G, Ruggeri A, Mazzoni A et al (2007) Immunohistochemical identification of decorin and biglycan in human dentin: a correlative field emission scanning electron microscopy/transmission electron microscopy study. Calcif Tissue Int 81:39–45. https://doi.org/10.1007/s00223-007-9027-z
Perez-Hernandez D, Gutiérrez-Vázquez C, Jorge I et al (2013) The intracellular interactome of tetraspanin-enriched microdomains reveals their function as sorting machineries toward exosomes. J Biol Chem 288:11649–11661. https://doi.org/10.1074/jbc.M112.445304
Sasaki T, Garant PR (1996) Structure and organization of odontoblasts. Anat Rec 245:235–249. https://doi.org/10.1002/(SICI)1097-0185(199606)245:2%3c235::AID-AR10%3e3.0.CO;2-Q
Sasaki R, Aoki S, Yamato M et al (2008) Neurosphere generation from dental pulp of adult rat incisor. Eur J Neurosci 27:538–548. https://doi.org/10.1111/j.1460-9568.2008.06026.x
Schröder J, Lüllmann-Rauch R, Himmerkus N et al (2009) Deficiency of the tetraspanin CD63 associated with kidney pathology but normal lysosomal function. Mol Cell Biol 29:1083–1094. https://doi.org/10.1128/mcb.01163-08
Schulze U, Brast S, Grabner A et al (2017) Tetraspanin CD63 controls basolateral sorting of organic cation transporter 2 in renal proximal tubules. FASEB J 31:1421–1433. https://doi.org/10.1096/fj.201600901R
Shapiro JL, Wen X, Okamoto CT et al (2007) Cellular uptake of amelogenin, and its localization to CD63, and lamp1-positive vesicles. Cell Mol Life Sci 64:244–256. https://doi.org/10.1007/s00018-006-6429-4
Stratmann U, Wen X, Schaarschmidt K et al (1997) The mineralization of mantle dentine and of circumpulpal dentine in the rat: an ultrastructural and element-analytical study. Anat Embryol 195:289–297. https://doi.org/10.1007/s004290050048
Swanson WB, Gong T, Zhang Z et al (2020) Controlled release of odontogenic exosomes from a biodegradable vehicle mediates dentinogenesis as a novel biomimetic pulp capping therapy. J Control Release 324:679–694. https://doi.org/10.1016/j.jconrel.2020.06.006
Tominaga N, Hagiwara K, Kosaka N et al (2014) RPN2-mediated glycosylation of tetraspanin CD63 regulates breast cancer cell malignancy. Mol Cancer 13:1–11. https://doi.org/10.1186/1476-4598-13-134
Veziroglu EM, Mias GI (2020) Characterizing extracellular vesicles and their diverse RNA contents. Front Genet 11:1–30. https://doi.org/10.3389/fgene.2020.00700
Wang HS, Yang FH, Wang YJ et al (2019) Odontoblastic exosomes attenuate apoptosis in neighboring cells. J Dent Res 98:1271–1278. https://doi.org/10.1177/0022034519869580
Yamakoshi Y, Hu JCC, Fukae M et al (2005) Dentin glycoprotein: the protein in the middle of the dentin sialophosphoprotein chimera. J Biol Chem 280:17472–17479. https://doi.org/10.1074/jbc.M413220200
Funding
This work was supported by an intramural grant of Showa University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflicts of interest.
Ethical approval
Approval for this study was obtained from the ethics committee of the Showa University School of Dentistry (approval number 19006 and 12018 for ICR mice, 13034 for C57BL/6 mice).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Matsuki-Fukushima, M., Fujikawa, K., Inoue, S. et al. Expression and localization of CD63 in the intracellular vesicles of odontoblasts. Histochem Cell Biol 157, 611–622 (2022). https://doi.org/10.1007/s00418-022-02072-6
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-022-02072-6