Abstract
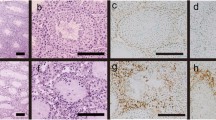
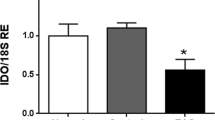
Male haploid cells, spermatids and spermatozoa, that appear after the establishment of immune tolerance express novel cell surface and intracellular proteins that can be recognized as foreign antigens by the self-immune system. However, these germ cells do not normally evoke a pathological immune response. The immune-privileged micro-circumstance in testis involving the blood–testis–barrier formed by Sertoli cells protects these germ cells from autoimmune attack. We recently found that immunization with heat shock protein family A member 4-like (HSPA4L), one of the new differentiation antigens of haploid cells, induced experimental autoimmune orchitis (EAO) in A/J male mice. In this study, we focused on G protein-coupled receptor kinase interacting protein-1 (GIT1), another haploid cell-specific differentiation antigen, to investigate whether GIT1 is a target autoantigen for EAO induction. GIT1 emulsified with complete Freund’s adjuvant was injected subcutaneously into the mice inguinal region once on day 0 and again on day 14, and the optimum condition of EAO induction was determined. Mice immunized with 200 μg GIT1 showed significantly higher incidence of EAO than that of immunization with other concentrations. In particular, significant lymphocytic inflammation and extensive aspermatogenesis were observed in these mice at 120 days after the first immunization. These findings indicate that GIT1 is also a target antigen that induces EAO, like HSPA4L.






Similar content being viewed by others
Availability of data and materials
All data are available within the article or supplementary information or from the corresponding author upon reasonable request.
Abbreviations
- BP:
-
Bordetella pertussis
- BTB:
-
Blood–testis barrier
- CFA:
-
Complete Freund’s adjuvant
- DAB:
-
3,3′-Diaminobenzidine
- EAO:
-
Experimental autoimmune orchitis
- GIT1:
-
G protein-coupled receptor kinase interacting protein-1
- HEK293T:
-
Human embryonic kidney 293 T
- HRP:
-
Horseradish peroxidase
- HSPA4L:
-
Heat shock protein family A member 4-like
- TGC:
-
Testicular germ cell
References
Albertinazzi C, Za L, Paris S, de Curtis I (2003) ADP-ribosylation factor 6 and a functional PIX/p95–APP1 complex are required for Rac1B-mediated neurite outgrowth. Mol Biol Cell 14:1295–1307. https://doi.org/10.1091/mbc.e02-07-0406
Claing A, Perry SJ, Achiriloaie M, Walker JK, Albanesi JP, Lefkowitz RJ, Premont RT (2000) Multiple endocytic pathways of G protein-coupled receptors delineated by GIT1 sensitivity. Proc Natl Acad Sci USA 97:1119–1124. https://doi.org/10.1073/pnas.97.3.1119
Doncel GF, Di Paola JA, Lustig L (1989) Sequential study of the histopathology and cellular and humoral immune response during the development of an autoimmune orchitis in Wistar rats. Am J Reprod Immunol 20:44–51. https://doi.org/10.1111/j.1600-0897.1989.tb00638.x
Dym M (1973) The fine structure of the monkey (Macaca) sertoli cell and its role in maintaining the blood-testis barrier. Anat Rec 175:639–656. https://doi.org/10.1002/ar.1091750402
Dym M, Fawcett DW (1970) The blood-testis barrier in the rat and the physiological compartmentation of the seminiferous epithelium. Biol Reprod 3:308–326. https://doi.org/10.1093/biolreprod/3.3.308
Fijak M, Meinhardt A (2006) The testis in immune privilege. Immunol Rev 213:66–81. https://doi.org/10.1111/j.1600-065x.2006.00438.x
Fijak M, Iosub R, Schneider E, Linder M, Respondek K, Klug J, Meinhardt A (2005) Identification of immunodominant autoantigens in rat autoimmune orchitis. J Pathol 207:127–138. https://doi.org/10.1002/path.1828
Guazzone VA, Jacobo P, Theas MS, Lustig L (2009) Cytokines and chemokines in testicular inflammation: a brief review. Microsc Res Tech 72:620–628. https://doi.org/10.1002/jemt.20704
Hassanin AM, Ayad E (2016) The impact of chronic testicular inflammatory infiltration on spermatogenesis in azoospermic men, evidence-based pilot study. Middle East Fertil Soc J 21:31–35
Hasselmann JPC, Karim H, Khalaj AJ, Ghosh S, Tiwari-Woodruff SK (2017) Consistent induction of chronic experimental autoimmune encephalomyelitis in C57BL/6 mice for the longitudinal study of pathology and repair. J Neurosci Methods 284:71–84. https://doi.org/10.1016/j.jneumeth.2017.04.003
Hatakeyama S (1965) A study on the interstitial cells of the human testis, especially on their fine-structural pathology. Acta Pathol Jpn 15:155–196. https://doi.org/10.1111/j.1440-1827.1965.tb00003.x
Hess RA, Renato de Franca L (2008) Spermatogenesis and cycle of the seminiferous epithelium. Adv Exp Med Biol 636:1–15. https://doi.org/10.1007/978-0-387-09597-4_1
Itoh M (2017) Microcircumstance for induction and prevention of testicular autoimmunity. Testicular Autoimmity. Springer, Japan, pp 17–64
Itoh M, Hiramine C, Hojo K (1991) A new murine model of autoimmune orchitis induced by immunization with viable syngeneic testicular germ cells alone. I. Immunological and histological studies. Clin Exp Immunol 83:137–142. https://doi.org/10.1111/j.1365-2249.1991.tb05603.x
Itoh M, Mukasa A, Tokunaga Y, Hiramine C, Hojo K (1992) Suppression of efferent limb of testicular autoimmune response by a regulatory CD4+ T cell line in mice. Clin Exp Immunol 87:455–460. https://doi.org/10.1111/j.1365-2249.1992.tb03019.x
Itoh M, De-Rooij D, Takeuchi Y (1995) Mode of inflammatory cell infiltration in testes of mice injected with syngeneic testicular germ cells without adjuvant. J Anat 187:671–679
Itoh M, Ueno M, Li XQ, Satriotomo I, Takeuchi Y (1998) Topographical uptake of blood-borne horseradish peroxidase (HRP) in the murine testis at the light microscopic level. Int J Androl 21:74–80. https://doi.org/10.1046/j.1365-2605.1998.00100.x
Jacobo P (2018) The role of regulatory T Cells in autoimmune orchitis. Andrologia 50(11):e13092. https://doi.org/10.1111/and.13092
Kim S, Ko J, Shin H, Lee JR, Lim C, Han JH, Altrock WD et al (2003) The GIT family of proteins forms multimers and associates with the presynaptic cytomatrix protein Piccolo. J Biol Chem 278:6291–6300. https://doi.org/10.1074/jbc.m212287200
Ko J, Kim S, Valtschanoff JG, Shin H, Lee JR, Sheng M, Premont RT et al (2003) Interaction between liprin-alpha and GIT1 is required for AMPA receptor targeting. J Neurosci 23:1667–1677. https://doi.org/10.1523/jneurosci.23-05-01667.2003
Kohno S, Munoz JA, Williams TM, Teuscher C, Bernard CC, Tung KS (1983) Immunopathology of murine experimental allergic orchitis. J Immunol 130:2675–2682
Lin YH, Lin YM, Kuo YC, Wang YY, Kuo PL (2011) Identification and characterization of a novel Rab GTPase-activating protein in spermatids. Int J Androl 34:e358-367. https://doi.org/10.1111/j.1365-2605.2010.01126.x
Lucca LE, Dominguez-Villar M (2020) Modulation of regulatory T cell function and stability by co-inhibitory receptors. Nat Rev Immunol 20:680–693. https://doi.org/10.1038/s41577-020-0296-3
Menon P, Deane R, Sagare A, Lane SM, Zarcone TJ, O’Dell MR, Yan C et al (2010) Impaired spine formation and learning in GPCR kinase 2 interacting protein-1 (GIT1) knockout mice. Brain Res 1317:218–226. https://doi.org/10.1016/j.brainres.2009.11.084
Mukasa A, Hiramine C, Hojo K (1994) Generation and characterization of a continuous line of CD8+ suppressively regulatory T lymphocytes which down-regulates experimental autoimmune orchitis (EAO) in mice. Clin Exp Immunol 96:138–145. https://doi.org/10.1111/j.1365-2249.1994.tb06243.x
Nagahori K, Hirai S, Hatayama N, Kuramasu M, Omotehara T, Kawata S, Li Z et al (2021) Heat shock protein A4L is a potent autoantigen for testicular autoimmunity in mice. J Reprod Immunol 145:103318. https://doi.org/10.1016/j.jri.2021.103318
Premont RT, Claing A, Vitale N, Freeman JLR, Pitcher JA, Patton WA, Moss J et al (1998) β2-Adrenergic receptor regulation by GIT1, a G protein-coupled receptor kinase-associated ADP ribosylation factor GTPase-activating protein. Proc Natl Acad Sci USA 95:14082–14087. https://doi.org/10.1073/pnas.95.24.14082
Qu N, Naito M, Terayama H, Hirai S, Li J, Ogawa Y, Kitaoka M et al (2010) Developmental ontogeny of autoantigens associated with localized autoimmunity in murine testis and epididymis. J Reprod Immunol 87:45–51. https://doi.org/10.1016/j.jri.2010.05.005
Qu N, Ogawa Y, Kuramasu M, Nagahori K, Sakabe K, Itoh M (2019) Immunological microenvironment in the testis. Reprod Med Biol 19:24–31. https://doi.org/10.1002/rmb2.12293
Sakagami H, Hara Y, Fukaya M (2016) Interaction of serologically defined colon cancer antigen-3 with Arf6 and its predominant expression in the mouse testis. Biochem Biophys Res Commun 477:868–873. https://doi.org/10.1016/j.bbrc.2016.06.150
Sasamoto Y, Kotake S, Yoshikawa K, Wiggert B, Gery I, Matsuda H (1994) Interphotoreceptor retinoid-binding protein derived peptide can induce experimental autoimmune uveoretinitis in various rat strains. Curr Eye Res 13:845–849. https://doi.org/10.3109/02713689409025141
Schmalzigaug R, Phee H, Davidson CE, Weiss A, Premont RT (2007) Differential expression of the ARF GAP genes GIT1 and GIT2 in mouse tissues. J Histochem Cytochem 55:1039–1048. https://doi.org/10.1369/jhc.7A7207.2007
Segura I, Essmann CL, Weinges S, Acker-Palmer A (2007) Grb4 and GIT1 transduce ephrinB reverse signals modulating spine morphogenesis and synapse formation. Nat Neurosci 10:301–310. https://doi.org/10.1038/nn1858
Tainosho S, Naito M, Hirai S, Terayama H, Qu N, Itoh M (2011) Multilayered structure of the basal lamina of the tubuli recti in normal mice. Med Mol Morphol 44:34–38. https://doi.org/10.1007/s00795-010-0501-8
Terayama H, Naito M, Qu N, Hirai S, Kitaoka M, Ogawa Y, Itoh M (2011) Intratesticular expression of mRNAs of both interferon γ and tumor necrosis factor α is significantly increased in experimental autoimmune orchitis in mice. J Reprod Dev 57:296–302. https://doi.org/10.1262/jrd.10-121n
Terayama H, Hirai S, Naito M, Qu N, Katagiri C, Nagahori K, Hayashi S et al (2016) Specific autoantigens identified by sera obtained from mice that are immunized with testicular germ cells alone. Sci Rep 6:35599. https://doi.org/10.1038/srep35599
Tokuda N, Kasahara M, Levy RB (1990) Differential regulation and expression of major histocompatibility complex (MHC) and Ly-6 gene products on mouse testicular Leydig and Sertoli cell lines. J Autoimmun 3:457–471. https://doi.org/10.1016/s0896-8411(05)80013-7
Tung KS, Unanue ER, Dixon FJ (1971) Pathogenesis of experimental allergic orchitis. II. The role of antibody. J Immunol 106:1463
Tung KS, Primakoff P, Woolman-Gamer L, Myles DG (1997) Mechanism of infertility in male guinea pigs immunized with sperm PH-20. Biol Reprod 56:1133–1141. https://doi.org/10.1095/biolreprod56.5.1133
Tung KS, Harakal J, Qiao H, Rival C, Li JC, Paul AG, Wheeler K et al (2017) Egress of sperm autoantigen from seminiferous tubules maintains systemic tolerance. J Clin Invest 127:1046–1060. https://doi.org/10.1172/JCI89927
Vergouwen RP, Huiskamp R, Bas RJ, Roepers-Gajadien HL, Davids JA, de Rooij DG (1995) Radiosensitivity of testicular cells in the fetal mouse. Radiat Res 141:66–73. https://doi.org/10.2307/3579091
Won H, Mah W, Kim E, Kim JW, Hahm EK, Kim MH, Cho S et al (2011) GIT1 is associated with ADHD in humans and ADHD-like behaviors in mice. Nat Med 17:566–572. https://doi.org/10.1038/nm.2330
Zhang H, Webb DJ, Asmussen H, Horwitz AF (2003) Synapse formation is regulated by the signaling adaptor GIT1. J Cell Biol 161:131–142. https://doi.org/10.1083/jcb.200211002
Zhang H, Webb DJ, Asmussen H, Niu S, Horwitz AFA (2005) GIT1/PIX/Rac/PAK signaling module regulates spine morphogenesis and synapse formation through MLC. J Neurosci 25:3379–3388. https://doi.org/10.1523/jneurosci.3553-04.2005
Zhao SJ, Liu H, Chen J, Qian DF, Kong FQ, Jie J, Yin GY et al (2020) Macrophage GIT1 contributes to bone regeneration by regulating inflammatory responses in an ERK/NRF2-dependent way. J Bone Mineral Res 35:2015–2031. https://doi.org/10.1002/jbmr.4099
Funding
This study was supported by Grants-in-Aid for Scientific Research (C: 15K08159, 19K18572) from the Japan Society for the Promotion of Science (JSPS), KAKENHI. The funders had no role in the study design, data collection and analysis, decision to publish, or manuscript preparation.
Author information
Authors and Affiliations
Contributions
K.N., S.H., and M.I. conceived and designed the study. K.N., M.K., S.K., Z.-L.L., and N.Q. performed the experiments. K.N., M.K., and M.I. provided critical reagents and analytical tools. K.N., S.K., and T.Y. analyzed the data. K.N. and M.I. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Consent to participate
All authors approved participation in the manuscript.
Consent for publication
All authors approved the final version of the manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Nagahori, K., Kuramasu, M., Kawata, S. et al. GIT1 is an untolerized autoantigen involved in immunologic disturbance of spermatogenesis. Histochem Cell Biol 157, 309–319 (2022). https://doi.org/10.1007/s00418-021-02061-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-021-02061-1