Abstract
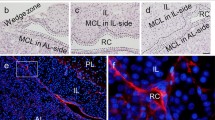
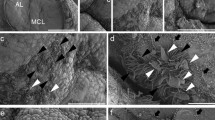
The adenohypophysis is composed of the anterior and intermediate lobes (AL and IL), and secretes important hormones for growth, sexual development, metabolism, and reproduction. In the marginal cell layer (MCL) facing Rathke’s cleft between the IL and AL, cluster of differentiation (CD) 9-, CD81-, S100β-, and SOX2-quadruple positive (CD9/CD81/S100β/SOX2-positive) cells in the adult IL are settled as tissue-resident stem/progenitor cells supplying hormone-producing cells to the AL. However, it is unclear how CD9/CD81/S100β/SOX2-positive cells in the IL-side MCL migrate into the AL across Rathke’s cleft. In the present study, we performed chimeric pituitary tissue culture using S100β/GFP-transgenic rats and Wistar rats, and traced the footprint of S100β/GFP-expressing cells. We detected IL-side S100β/GFP-expressing cells in the AL tissue, demonstrating that these cells migrate from the IL to the AL. However, the cells failed to migrate in the opposite direction. Consistently, scanning electron microscopic analysis revealed well-developed cytoplasmic protrusions in the IL-side MCL, but not in the AL-side MCL, suggesting that IL-side CD9/CD81/S100β/SOX2-positive cells had higher migratory activity. We also searched for a specific marker for IL-side CD9/CD81/S100β/SOX2-positive cells and identified tetraspanin 1 (TSPAN1) from microarray analysis. Downregulation of Tspan1 by specific siRNA impaired cell migration and significantly reduced expression of snail family transcriptional repressor 2 (Slug), a marker of epithelial-mesenchymal transition (EMT). Therefore, CD9/CD81/S100β/SOX2-positive cells in the IL-side MCL can be stem/progenitor cells that provide stem/progenitor cells to the AL-side MCL via SLUG-mediated EMT and cell migration.






Similar content being viewed by others
References
Chen J, Hersmus N, Van Duppen V, Caesens P, Denef C, Vankelecom H (2005) The adult pituitary contains a cell population displaying stem/progenitor cell and early embryonic characteristics. Endocrinol 146(9):3985–3998. https://doi.org/10.1210/en.2005-0185
Chen M, Kato T, Higuchi M, Yoshida S, Yako H, Kanno N, Kato Y (2013) Coxsackievirus and adenovirus receptor-positive cells compose the putative stem/progenitor cell niches in the marginal cell layer and parenchyma of the rat anterior pituitary. Cell Tissue Res 354(3):823–836. https://doi.org/10.1007/s00441-013-1713-8
Detchokul S, Williams ED, Parker MW, Frauman AG (2014) Tetraspanins as regulators of the tumour microenvironment: implications for metastasis and therapeutic strategies. Br J Pharmacol 171(24):5462–5490. https://doi.org/10.1111/bph.12260
Gahmberg CG, Fagerholm SC, Nurmi SM, Chavakis T, Marchesan S, Gronholm M (2009) Regulation of integrin activity and signalling. Biochim Biophys Acta 1790(6):431–444. https://doi.org/10.1016/j.bbagen.2009.03.007
Gremeaux L, Fu Q, Chen J, Vankelecom H (2012) Activated phenotype of the pituitary stem/progenitor cell compartment during the early-postnatal maturation phase of the gland. Stem Cells Dev 21(5):801–813. https://doi.org/10.1089/scd.2011.0496
Horiguchi K, Fujiwara K, Kouki T, Kikuchi M, Yashiro T (2008) Immunohistochemistry of connexin 43 throughout anterior pituitary gland in a transgenic rat with green fluorescent protein-expressing folliculo-stellate cells. Anat Sci Int 83(4):256–260. https://doi.org/10.1111/j.1447-073X.2008.00239.x
Horiguchi K, Kikuchi M, Kusumoto K, Fujiwara K, Kouki T, Kawanishi K, Yashiro T (2010) Living-cell imaging of transgenic rat anterior pituitary cells in primary culture reveals novel characteristics of folliculo-stellate cells. J Endocrinol 204(2):115–123. https://doi.org/10.1677/joe-09-0333
Horiguchi K, Kouki T, Fujiwara K, Kikuchi M, Yashiro T (2011) The extracellular matrix component laminin promotes gap junction formation in the rat anterior pituitary gland. J Endocrinol 208(3):225–232. https://doi.org/10.1677/joe-10-0297
Horiguchi K, Fujiwara K, Yoshida S, Higuchi M, Tsukada T, Kanno N, Yashiro T, Tateno K, Osako S, Kato T, Kato Y (2014) Isolation of dendritic-cell-like S100beta-positive cells in rat anterior pituitary gland. Cell Tissue Res 357(1):301–308. https://doi.org/10.1007/s00441-014-1817-9
Horiguchi K, Fujiwara K, Tsukada T, Yako H, Tateno K, Hasegawa R, Takigami S, Ohsako S, Yashiro T, Kato T, Kato Y (2016a) Expression of Slug in S100beta-protein-positive cells of postnatal developing rat anterior pituitary gland. Cell Tissue Res 363(2):513–524. https://doi.org/10.1007/s00441-015-2256-y
Horiguchi K, Nakakura T, Yoshida S, Tsukada T, Kanno N, Hasegawa R, Takigami S, Ohsako S, Kato T, Kato Y (2016b) Identification of THY1 as a novel thyrotrope marker and THY1 antibody-mediated thyrotrope isolation in the rat anterior pituitary gland. Biochem Biophys Res Commun 480(2):273–279. https://doi.org/10.1016/j.bbrc.2016.10.049
Horiguchi K, Fujiwara K, Yoshida S, Nakakura T, Arae K, Tsukada T, Hasegawa R, Takigami S, Ohsako S, Yashiro T, Kato T, Kato Y (2018) Isolation and characterisation of CD9-positive pituitary adult stem/progenitor cells in rats. Sci Rep 8(1):5533. https://doi.org/10.1038/s41598-018-23923-0
Horiguchi K, Fujiwara K, Yoshida S, Tsukada T, Hasegawa R, Takigami S, Ohsako S, Yashiro T, Kato T, Kato Y (2020a) CX3CL1/CX3CR1-signalling in the CD9/S100beta/SOX2-positive adult pituitary stem/progenitor cells modulates differentiation into endothelial cells. Histochem Cell Biol 153(6):385–396. https://doi.org/10.1007/s00418-020-01862-0
Horiguchi K, Yoshida S, Tsukada T, Fujiwara K, Nakakura T, Hasegawa R, Takigami S, Ohsako S (2020b) Cluster of differentiation (CD) 9-positive mouse pituitary cells are adult stem/progenitor cells. Histochem Cell Biol 155(3):391–404. https://doi.org/10.1007/s00418-020-01943-0
Horiguchi K, Yoshida S, Tsukada T, Nakakura T, Fujiwara K, Hasegawa R, Takigami S, Ohsako S (2020c) Expression and functions of cluster of differentiation 9 and 81 in rat mammary epithelial cells. J Reprod Dev 66(6):515–522. https://doi.org/10.1262/jrd.2020-082
Horiguchi K, Fujiwara K, Takeda Y, Nakakura T, Tsukada T, Yoshida S, Hasegawa R, Takigami S, Ohsako S (2021) CD9-positive cells in the intermediate lobe of the pituitary gland are important supplier for prolactin-producing cells in the anterior lobe. Cell Tissue Res. https://doi.org/10.1007/s00441-021-03460-5
Jin Y, Takeda Y, Kondo Y, Tripathi LP, Kang S, Takeshita H, Kuhara H, Maeda Y, Higashiguchi M, Miyake K, Morimura O, Koba T, Hayama Y, Koyama S, Nakanishi K, Iwasaki T, Tetsumoto S, Tsujino K, Kuroyama M, Iwahori K, Hirata H, Takimoto T, Suzuki M, Nagatomo I, Sugimoto K, Fujii Y, Kida H, Mizuguchi K, Ito M, Kijima T, Rakugi H, Mekada E, Tachibana I, Kumanogoh A (2018) Double deletion of tetraspanins CD9 and CD81 in mice leads to a syndrome resembling accelerated aging. Sci Rep 8(1):5145. https://doi.org/10.1038/s41598-018-23338-x
Jorda M, Olmeda D, Vinyals A, Valero E, Cubillo E, Llorens A, Cano A, Fabra A (2005) Upregulation of MMP-9 in MDCK epithelial cell line in response to expression of the Snail transcription factor. J Cell Sci 118(Pt 15):3371–3385. https://doi.org/10.1242/jcs.02465
Joseph MJ, Dangi-Garimella S, Shields MA, Diamond ME, Sun L, Koblinski JE, Munshi HG (2009) Slug is a downstream mediator of transforming growth factor-beta1-induced matrix metalloproteinase-9 expression and invasion of oral cancer cells. J Cell Biochem 108(3):726–736. https://doi.org/10.1002/jcb.22309
Liu G, Wang Y, Yang L, Zou B, Gao S, Song Z, Lin Z (2019) Tetraspanin 1 as a mediator of fibrosis inhibits EMT process and Smad2/3 and beta-catenin pathway in human pulmonary fibrosis. J Cell Mol Med 23(5):3583–3596. https://doi.org/10.1111/jcmm.14258
Nieto MA (2002) The snail superfamily of zinc-finger transcription factors. Nat Rev Mol Cell Biol 3(3):155–166. https://doi.org/10.1038/nrm757
Shields MA, Krantz SB, Bentrem DJ, Dangi-Garimella S, Munshi HG (2012) Interplay between beta1-integrin and Rho signaling regulates differential scattering and motility of pancreatic cancer cells by snail and Slug proteins. J Biol Chem 287(9):6218–6229. https://doi.org/10.1074/jbc.M111.308940
Vankelecom H, Chen J (2014) Pituitary stem cells: where do we stand? Mol Cell Endocrinol 385(1–2):2–17. https://doi.org/10.1016/j.mce.2013.08.018
Wang Y, Liang Y, Yang G, Lan Y, Han J, Wang J, Yin D, Song R, Zheng T, Zhang S, Pan S, Liu X, Zhu M, Liu Y, Cui Y, Meng F, Zhang B, Liang S, Guo H, Liu Y, Hassan MK, Liu L (2018) Tetraspanin 1 promotes epithelial-to-mesenchymal transition and metastasis of cholangiocarcinoma via PI3K/AKT signaling. J Exp Clin Cancer Res 37(1):300. https://doi.org/10.1186/s13046-018-0969-y
Yoshida S, Kato T, Yako H, Susa T, Cai LY, Osuna M, Inoue K, Kato Y (2011) Significant quantitative and qualitative transition in pituitary stem / progenitor cells occurs during the postnatal development of the rat anterior pituitary. J Neuroendocrinol 23(10):933–943. https://doi.org/10.1111/j.1365-2826.2011.02198.x
Yoshida S, Kato T, Kato Y (2016a) Regulatory System for Stem/Progenitor Cell Niches in the Adult Rodent Pituitary. Int J Mol Sci 17(1):75. https://doi.org/10.3390/ijms17010075
Yoshida S, Nishimura N, Ueharu H, Kanno N, Higuchi M, Horiguchi K, Kato T, Kato Y (2016b) Isolation of adult pituitary stem/progenitor cell clusters located in the parenchyma of the rat anterior lobe. Stem Cell Res 17(2):318–329. https://doi.org/10.1016/j.scr.2016.08.016
Acknowledgements
We are grateful to Dr. Y. Kato and T. Kato (Institute for Reproduction and Endocrinology, Meiji University) for helpful discussions. We would like to thank Editage (www.editage.jp) for English language editing.
Funding
This work was supported by JSPS KAKENHI Grants (nos. 19K07255 to K.H.).
Conflicts of interest
None.
Availability of data and material
Not applicable.
Code availability
Not applicable.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosure statement
The authors have nothing to declare.
Ethical approval
The current study was approved by the Committee on Animal Experiments of the Kyorin University based on the NIH Guidelines for the Care and Use of Laboratory Animals. This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
418_2021_2009_MOESM1_ESM.tif
Supplementary file 1 (TIF 16728 KB) Expression of CD9- and CD81-positive cells in the adult pituitary gland. (a) Upper panels: HE staining of the pituitary gland. Lower panels: high magnification of boxed area of upper panels. Dotted lines indicate boundary between the IL and the PL. (b) Immunohistochemistry for CD9 (red) and CD81 (green). (c) Double immunohistochemistry staining for CD9 and either S100β, or SOX2, with DAPI staining (blue). Scale bars, 500 μm (a, upper panel), 200 (b, right panel), 100 μm (a, lower panel), 50 μm (b, lower panel, and c)
418_2021_2009_MOESM2_ESM.tif
Supplementary file 2 (TIF 11193 KB) Double immunohistochemistry staining for CD9 (red) and CD81 (green) in developing pituitary gland at E12.5, E15.5, and E20.5. Left panels are HE staining. Right panels are merged image of immunohistochemistry with DAPI staining. AL, anterior lobe. IL, intermediate lobe. PL, posterior lobe. PP, pituitary placode. Dotted lines, the border of PP (E12.5) and the adenohypophysis (E15.5 and E20.5). Scale bars, 100 μm
418_2021_2009_MOESM3_ESM.tif
Supplementary file 3 (TIF 6408 KB) Migration assay using chimeric pituitary culture. (a) Left panel shows bright field and GFP of chimeric pituitary tissue of adult pituitary (9W). Fluorescent and non-fluorescent tissues indicate the IL/PL tissue from S100β/GFP-TG rats and the AL tissue from Wistar rats, respectively. Right panel shows the cryosection of chimeric pituitary tissue (2-day cultivation). Arrowheads indicate GFP-expressing cells in AL-side MCL. (b) and (c) bright field and GFP of chimeric pituitary tissue of P5 and 9W pituitary, respectively. Fluorescent and non-fluorescent tissues indicate the AL tissue from S100β/GFP-TG rats and the IL/PL tissue from Wistar rats, respectively. Right panel shows the cryosection of chimeric pituitary tissue (2-day cultivation). Arrowheads indicate GFP-expressing cells in AL-side MCL. AL, anterior lobe, IL, intermediate lobe, PL, posterior lobe. Scale bars, 1 mm (a), 500 μm (b, left panel), 50 μm (a and c, right panels)
418_2021_2009_MOESM4_ESM.tif
Supplementary file 4 (TIF 8879 KB) Immunohistochemistry for TSPAN1 (green) in the developing pituitary gland at E12.5, E14.5, E15.5, E20.5, P5, and P20. Right panels are merged images of immunohistochemistry and DAPI staining. Scale bars, 100 μm
Rights and permissions
About this article
Cite this article
Horiguchi, K., Fujiwara, K., Tsukada, T. et al. CD9-positive cells in the intermediate lobe migrate into the anterior lobe to supply endocrine cells. Histochem Cell Biol 156, 301–313 (2021). https://doi.org/10.1007/s00418-021-02009-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-021-02009-5