Abstract
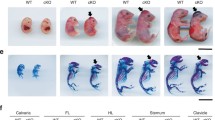
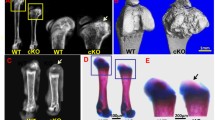
Long bones of the appendicular skeleton are formed through endochondral ossification. Endochondral bone formation initiates with mesenchymal condensation, followed by the formation of a cartilage template which is replaced by bone. Fibroblast growth factor 9 (FGF9) regulates bone development. Fgf9−/− mice exhibit disproportionate shortening of proximal skeletal elements. Fgf9 missense mutations in mice and humans induce joint synostosis. Thus, FGF9 is critical for regulating bone length and joint formation. Conversely, mechanisms regulating bone width remain unclear. Here, we showed that the homozygous elbow knee synostosis (Eks) mutant mice harboring N143T mutation in Fgf9 have wide long bones at birth. We investigated the cellular and molecular mechanisms underlying the widened prospective humerus in Fgf9Eks/Eks embryos. Increased and expanded FGF signaling in concert with wider expression domain of Fgf receptor 3 (Fgfr3) during chondrogenic condensation of the humerus led to widened cartilage, which resulted in the formation of wider prospective humeri in neonatal Fgf9Eks/Eks mice. Increased and expanded FGF signaling during chondrogenic condensation led to increased density of chondrocytes of the humeri accompanied by increased proliferation of chondrocytes which express inappropriately higher levels of cyclin D1 in Fgf9Eks/Eks embryos. The results suggest that FGF9 regulates the width of prospective long bones by controlling the width of chondrogenic condensation.




Similar content being viewed by others
Abbreviations
- COL1:
-
Type I collagen
- COL2:
-
Type II collagen
- E:
-
Embryonic day
- EdU:
-
5-Ethynyl- 2′-deoxyuridine
- Eks:
-
Elbow knee synostosis
- FGF:
-
Fibroblast growth factor
- FGFR:
-
Fibroblast growth factor receptor
- HE:
-
Hematoxylin and eosin
- P:
-
Postnatal
- PBS:
-
Phosphate-buffered saline
- pERK1/2:
-
ERK1/2 phosphorylation
- RUNX2:
-
Runt-related transcription factor 2
- SOX9:
-
SRY-box9
References
Akiyama H, Kim JE, Nakashima K, Balmes G, Iwai N, Deng JM, Zhang Z, Martin JF, Behringer RR, Nakamura T, de Crombrugghe B (2005) Osteo-chondroprogenitor cells are derived from Sox9 expressing precursors. Proc Natl Acad Sci USA 102:14665–14670. https://doi.org/10.1073/pnas.0504750102
Behr B, Leucht P, Longaker MT, Quarto N (2010) Fgf-9 is required for angiogenesis and osteogenesis in long bone repair. Proc Natl Acad Sci USA 107:11853–11858. https://doi.org/10.1073/pnas.1003317107
Beier F, Ali Z, Mok D, Taylor AC, Leask T, Albanese C, Pestell RG, LuValle P (2001) TGFbeta and PTHrP control chondrocyte proliferation by activating cyclin D1 expression. Mol Biol Cell 12:3852–3863. https://doi.org/10.1091/mbc.12.12.3852
Brewer JR, Mazot P, Soriano P (2016) Genetic insights into the mechanisms of Fgf signaling. Genes Dev 30:751–771. 10.1101/gad.277137.115
Eswarakumar VP, Monsonego-Ornan E, Pines M, Antonopoulou I, Morriss-Kay GM, Lonai P (2002) The IIIc alternative of Fgfr2 is a positive regulator of bone formation. Development 129:3783–3793
Eswarakumar VP, Horowitz MC, Locklin R, Morriss-Kay GM, Lonai P (2004) A gain-of-function mutation of Fgfr2c demonstrates the roles of this receptor variant in osteogenesis. Proc Natl Acad Sci USA 101:12555–12560. https://doi.org/10.1073/pnas.0405031101
Harada M, Murakami H, Okawa A, Okimoto N, Hiraoka S, Nakahara T, Akasaka R, Shiraishi Y, Futatsugi N, Mizutani-Koseki Y, Kuroiwa A, Shirouzu M, Yokoyama S, Taiji M, Iseki S, Ornitz DM, Koseki H (2009) FGF9 monomer-dimer equilibrium regulates extracellular matrix affinity and tissue diffusion. Nature Genet 41:289–298. https://doi.org/10.1038/ng.316
Inada M, Yasui T, Nomura S, Miyake S, Deguchi K, Himeno M, Sato M, Yamagiwa H, Kimura T, Yasui N, Ochi T, Endo N, Kitamura Y, Kishimoto T, Komori T (1999) Maturational disturbance of chondrocytes in Cbfa1-deficient mice. Dev Dyn 214:279–290. https://doi.org/10.1002/(SICI)1097-0177(199904)214:4%3c279:AID-AJA1%3e3.0.CO;2-W
Iwata T, Chen L, Li C, Ovchinnikov DA, Behringer RR, Francomano CA, Deng CX (2000) A neonatal lethal mutation in FGFR3 uncouples proliferation and differentiation of growth plate chondrocytes in embryos. Hum Mol Genet 9:1603–1613. https://doi.org/10.1093/hmg/9.11.1603
Kim IS, Otto F, Zabel B, Mundlos S (1999) Regulation of chondrocyte differentiation by Cbfa1. Mech Dev 80:159–170. https://doi.org/10.1016/S0925-4773(98)00210-X
Matsumoto Y, Matsumoto K, Irie F, Fukushi J, Stallcup WB, Yamaguchi Y (2010) Conditional ablation of the heparan sulfate-synthesising enzyme Ext1 leads to dysregulation of bone morphogenic protein signaling and severe skeletal defects. J Biol Chem 285:19227–19234 10.1074/jbc.M110.105338
Matsuo I, Kimura-Yoshida C (2014) Extracellular distribution of diffusible growth factors controlled by heparan sulfate proteoglycans during mammalian embryogenesis. Philos Trans R Soc Lond B Biol Sci 369:1657. https://doi.org/10.1098/rstb.2013.0545
Murakami S, Kan M, McKeehan WL, de Crombrugghe B (2000) Up-regulation of the chondrogenic Sox9 gene by fibroblast growth factors is mediated by the mitogen-activated protein kinase pathway. Proc Natl Acad Sci USA 97:1113–1118. https://doi.org/10.1073/pnas.97.3.1113
Murakami H, Okawa A, Yoshida H, Nishikawa S, Moriya H, Koseki H (2002) Elbow knee synostosis (Eks): a new mutation on mouse Chromosome 14. Mamm Genome 13:341–344. https://doi.org/10.1007/s00335-001-2143-6
Ono N, Kronenberg HM (2015) Mesenchymal progenitor cells for the osteogenic lineage. Curr Mol Biol Rep 1:95–100. https://doi.org/10.1007/s40610-015-0017-z
Ono N, Ono W, Nagasawa T, Kronenberg HM (2014) A subset of chondrogenic cells provides early mesenchymal progenitors in growing bones. Nat Cell Biol 16:1157–1167. https://doi.org/10.1038/ncb3067
Ornitz DM, Marie PJ (2015) Fibroblast growth factor signaling in skeletal development and disease. Genes Dev 29:1463–1486. https://doi.org/10.1101/gad.266551.115
Shung CY, Ota S, Zhou ZQ, Keene DR, Hurlin PJ (2012) Disruption of a Sox9–β-catenin circuit by mutant Fgfr3 in thanatophoric dysplasia type II. Hum Mol Genet 21:4628–4644. https://doi.org/10.1093/hmg/dds305
Su N, Jin M, Chen L (2014) Role of FGF/FGFR signaling in skeletal development and homeostasis: learning from mouse models. Bone Res 2:14003. https://doi.org/10.1038/boneres.2014.3
Suzuki K, Yamanishi K, Mori O, Kamikawa M, Andersen B, Kato S, Toyoda T, Yamada G (2000) Defective terminal differentiation and hypoplasia of the epidermis in mice lacking the Fgf10 gene. FEBS Lett 481:53–56. https://doi.org/10.1016/S0014-5793(00)01968-2
Tsang M, Dawid IB (2004) Promotion and attenuation of FGF signaling through the Ras-MAPK pathway. Sci STKE. https://doi.org/10.1126/stke.2282004pe17
Wallner C, Schira J, Wagner JM, Schulte M, Fischer S, Hirsch T, Richter W, Abraham S, Kneser U, Lehnhardt M, Behr B (2015) Application of VEGFA and FGF-9 enhances angiogenesis, osteogenesis and bone remodelling in type 2 diabetic long bone regeneration. PLoS ONE 10:e0118823. https://doi.org/10.1371/journal.pone.0118823
Wang Q, Green RP, Zhao G, Ornitz DM (2001) Differential regulation of endochondral bone growth and joint development by FGFR1 and FGFR3 tyrosine kinase domains. Development 128:3867–3876
Wang L, Roth T, Abbott M, Ho L, Wattanachanya L, Nissenson RA (2017) Osteoblast-derived FGF9 regulates skeletal homeostasis. Bone 98:18–25. https://doi.org/10.1016/j.bone.2016.12.005
Zhang X, Ibrahimi OA, Olsen SK, Umemori H, Mohammadi M, Ornitz DM (2006) Receptor specificity of the fibroblast growth factor family. The complete mammalian FGF family. J Biol Chem 281:15694–15700. https://doi.org/10.1074/jbc.M601252200
Zhao Q, Eberspaecher H, Lefebvre V, de Crombrugghe B (1997) Parallel expression of Sox9 and Col2a1 in cells undergoing chondrogenesis. Dev Dyn 209:377–386. https://doi.org/10.1002/(SICI)1097-0177(199708)209:4%3c377:AID-AJA5%3e3.0.CO;2-F
Acknowledgements
We thank H. Koseki for sharing the Eks mouse line. We also thank Enago (www.enago.jp) for English language editing. This work was supported in part by JSPS KAKENHI Grant Number JP19K07243 to M. H
Author information
Authors and Affiliations
Contributions
Masayo Harada designed the study and performed the experiments. Masayo Harada and Keiichi Akita wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted (Institutional Animal Care and Use Committee of Tokyo Medical and Dental University, permit number A2019-097C). This article does not contain any studies with human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Harada, M., Akita, K. Mouse fibroblast growth factor 9 N143T mutation leads to wide chondrogenic condensation of long bones. Histochem Cell Biol 153, 215–223 (2020). https://doi.org/10.1007/s00418-020-01844-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-020-01844-2