Abstract
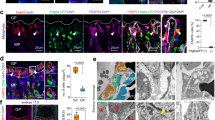
Septoclasts are mononuclear spindle-shaped phagocytes with their long processes in uncalcified cartilage matrices and locate adjacent to the capillary endothelium at the chondro-osseous junction of the growth plate. We have previously revealed a selective expression of epidermal-type fatty acid-binding protein (E-FABP/FABP5) in septoclasts. Although, pericytes are known to distribute along capillaries and directly surround their endothelial cells in a situation similar to septoclasts, no clear evidence is available on the relationship between septoclasts and pericytes. We investigated the chronological localization and morphological change of septoclasts during development of the tibia of mice to clarify the development of septoclasts and the immune-localization of pericyte markers in septoclasts to clarify the origin of septoclasts. E-FABP-immunoreactive septoclasts emerged at the perichondrium in the middle of the cartilaginous templates of the tibia in prenatal development. Septoclasts migrated to the surface of the cartilage adjacent to invading blood vessels. Processes of septoclasts became longer and their apexes attached to Von Kossa-negative uncalcified matrices during the formation process of the primary ossification center. Not only platelet-derived growth factor receptor beta, but also neuron-glial antigen 2 was localized in septoclasts of mice from E15 (embryonic day 15) to P6w (postnatal 6 week). Our results suggest that septoclasts are originated from pericytes and involved in the blood vessel invasion during formation of the primary ossification center.





Similar content being viewed by others
References
Albelda SM, Muller WA, Buck CA, Newman PJ (1991) Molecular and cellular properties of PECAM-1 (endoCAM/CD31): a novel vascular cell–cell adhesion molecule. J Cell Biol 114(5):1059–1068
Allsopp G, Gamble HJ (1979) An electron microscopic study of the pericytes of the developing capillaries in human fetal brain and muscle. J Anat 128(Pt 1):155–168
Amano O, Yamane A, Shimada M, Koshimizu U, Nakamura T, Iseki S (2002) Hepatocyte growth factor is essential for migration of myogenic cells and promotes their proliferation during the early periods of tongue morphogenesis in mouse embryos. Dev Dyn 223(2):169–179. https://doi.org/10.1002/dvdy.1228
Andrae J, Gallini R, Betsholtz C (2008) Role of platelet-derived growth factors in physiology and medicine. Genes Dev 22(10):1276–1312. https://doi.org/10.1101/gad.1653708
Armulik A, Genove G, Betsholtz C (2011) Pericytes: developmental, physiological, and pathological perspectives, problems, and promises. Dev Cell 21(2):193–215. https://doi.org/10.1016/j.devcel.2011.07.001
Arora PD, Manolson MF, Downey GP, Sodek J, McCulloch CA (2000) A novel model system for characterization of phagosomal maturation, acidification, and intracellular collagen degradation in fibroblasts. J Biol Chem 275(45):35432–35441. https://doi.org/10.1074/jbc.M003221200
Asahina K, Zhou B, Pu WT, Tsukamoto H (2011) Septum transversum-derived mesothelium gives rise to hepatic stellate cells and perivascular mesenchymal cells in developing mouse liver. Hepatology 53(3):983–995. https://doi.org/10.1002/hep.24119
Bando Y, Yamamoto M, Sakiyama K, Inoue K, Takizawa S, Owada Y, Iseki S, Kondo H, Amano O (2014) Expression of epidermal fatty acid binding protein (E-FABP) in septoclasts in the growth plate cartilage of mice. J Mol Histol 45(5):507–518. https://doi.org/10.1007/s10735-014-9576-1
Bando Y, Yamamoto M, Sakiyama K, Sakashita H, Taira F, Miyake G, Iseki S, Owada Y, Amano O (2017) Retinoic acid regulates cell-shape and -death of E-FABP (FABP5)-immunoreactive septoclasts in the growth plate cartilage of mice. Histochem Cell Biol 148(3):229–238. https://doi.org/10.1007/s00418-017-1578-0
Bruns RR, Palade GE (1968) Studies on blood capillaries. I. General organization of blood capillaries in muscle. J Cell Biol 37(2):244–276
Crisan M, Yap S, Casteilla L, Chen CW, Corselli M, Park TS, Andriolo G, Sun B, Zheng B, Zhang L, Norotte C, Teng PN, Traas J, Schugar R, Deasy BM, Badylak S, Buhring HJ, Giacobino JP, Lazzari L, Huard J, Peault B (2008) A perivascular origin for mesenchymal stem cells in multiple human organs. Cell Stem Cell 3(3):301–313. https://doi.org/10.1016/j.stem.2008.07.003
Dellavalle A, Sampaolesi M, Tonlorenzi R, Tagliafico E, Sacchetti B, Perani L, Innocenzi A, Galvez BG, Messina G, Morosetti R, Li S, Belicchi M, Peretti G, Chamberlain JS, Wright WE, Torrente Y, Ferrari S, Bianco P, Cossu G (2007) Pericytes of human skeletal muscle are myogenic precursors distinct from satellite cells. Nat Cell Biol 9(3):255–267. https://doi.org/10.1038/ncb1542
Diaz-Flores L, Gutierrez R, Madrid JF, Varela H, Valladares F, Acosta E, Martin-Vasallo P, Diaz-Flores L Jr (2009) Pericytes. Morphofunction, interactions and pathology in a quiescent and activated mesenchymal cell niche. Histol Histopathol 24(7):909–969. https://doi.org/10.14670/HH-24.909
Dore-Duffy P, Katychev A, Wang X, Van Buren E (2006) CNS microvascular pericytes exhibit multipotential stem cell activity. J Cereb Blood Flow Metab 26(5):613–624. https://doi.org/10.1038/sj.jcbfm.9600272
Etchevers HC, Vincent C, Le Douarin NM, Couly GF (2001) The cephalic neural crest provides pericytes and smooth muscle cells to all blood vessels of the face and forebrain. Development 128(7):1059–1068
Farrington-Rock C, Crofts NJ, Doherty MJ, Ashton BA, Griffin-Jones C, Canfield AE (2004) Chondrogenic and adipogenic potential of microvascular pericytes. Circulation 110(15):2226–2232. https://doi.org/10.1161/01.CIR.0000144457.55518.E5
Fosang AJ, Neame PJ, Last K, Hardingham TE, Murphy G, Hamilton JA (1992) The interglobular domain of cartilage aggrecan is cleaved by PUMP, gelatinases, and cathepsin B. J Biol Chem 267(27):19470–19474
Fosang AJ, Last K, Knauper V, Murphy G, Neame PJ (1996) Degradation of cartilage aggrecan by collagenase-3 (MMP-13). FEBS Lett 380(1–2):17–20
Gartland A, Mason-Savas A, Yang M, MacKay CA, Birnbaum MJ, Odgren PR (2009) Septoclast deficiency accompanies postnatal growth plate chondrodysplasia in the toothless (tl) osteopetrotic, colony-stimulating factor-1 (CSF-1)-deficient rat and is partially responsive to CSF-1 injections. Am J Pathol 175(6):2668–2675. https://doi.org/10.2353/ajpath.2009.090185
Geevarghese A, Herman IM (2014) Pericyte-endothelial crosstalk: implications and opportunities for advanced cellular therapies. Transl Res 163(4):296–306. https://doi.org/10.1016/j.trsl.2014.01.011
Gerhardt H, Betsholtz C (2003) Endothelial-pericyte interactions in angiogenesis. Cell Tissue Res 314(1):15–23. https://doi.org/10.1007/s00441-003-0745-x
Heldin CH, Westermark B (1999) Mechanism of action and in vivo role of platelet-derived growth factor. Physiol Rev 79(4):1283–1316
Hellstrom M, Kalen M, Lindahl P, Abramsson A, Betsholtz C (1999) Role of PDGF-B and PDGFR-beta in recruitment of vascular smooth muscle cells and pericytes during embryonic blood vessel formation in the mouse. Development 126(14):3047–3055
Kronenberg HM (2003) Developmental regulation of the growth plate. Nature 423(6937):332–336. https://doi.org/10.1038/nature01657
Lee ER, Lamplugh L, Shepard NL, Mort JS (1995) The septoclast, a cathepsin B-rich cell involved in the resorption of growth plate cartilage. J Histochem Cytochem 43(5):525–536
Lindahl P, Johansson BR, Leveen P, Betsholtz C (1997) Pericyte loss and microaneurysm formation in PDGF-B-deficient mice. Science 277(5323):242–245
Lindblom P, Gerhardt H, Liebner S, Abramsson A, Enge M, Hellstrom M, Backstrom G, Fredriksson S, Landegren U, Nystrom HC, Bergstrom G, Dejana E, Ostman A, Lindahl P, Betsholtz C (2003) Endothelial PDGF-B retention is required for proper investment of pericytes in the microvessel wall. Genes Dev 17(15):1835–1840. https://doi.org/10.1101/gad.266803
Long F, Ornitz DM (2013) Development of the endochondral skeleton. Cold Spring Harb Perspect Biol 5(1):a008334. https://doi.org/10.1101/cshperspect.a008334
Maes C (2013) Role and regulation of vascularization processes in endochondral bones. Calcif Tissue Int 92(4):307–323. https://doi.org/10.1007/s00223-012-9689-z
Mitchell PG, Magna HA, Reeves LM, Lopresti-Morrow LL, Yocum SA, Rosner PJ, Geoghegan KF, Hambor JE (1996) Cloning, expression, and type II collagenolytic activity of matrix metalloproteinase-13 from human osteoarthritic cartilage. J Clin Investig 97(3):761–768. https://doi.org/10.1172/JCI118475
Mort JS, Magny MC, Lee ER (1998) Cathepsin B: an alternative protease for the generation of an aggrecan ‘metalloproteinase’ cleavage neoepitope. Biochem J 335(Pt 3):491–494
Nakamura H, Ozawa H (1996) Ultrastructural, enzyme-, lectin, and immunohistochemical studies of the erosion zone in rat tibiae. J Bone Miner Res 11(8):1158–1164. https://doi.org/10.1002/jbmr.5650110815
Nakamura H, Sato G, Hirata A, Yamamoto T (2004) Immunolocalization of matrix metalloproteinase-13 on bone surface under osteoclasts in rat tibia. Bone 34(1):48–56
Nishiyama A, Boshans L, Goncalves CM, Wegrzyn J, Patel KD (2016) Lineage, fate, and fate potential of NG2-glia. Brain Res 1638(Pt B):116–128. https://doi.org/10.1016/j.brainres.2015.08.013
Odgren PR, Witwicka H, Reyes-Gutierrez P (2016) The cast of clasts: catabolism and vascular invasion during bone growth, repair, and disease by osteoclasts, chondroclasts, and septoclasts. Connect Tissue Res 57(3):161–174. https://doi.org/10.3109/03008207.2016.1140752
Owada Y, Abdelwahab SA, Suzuki R, Iwasa H, Sakagami H, Spener F, Kondo H (2001) Localization of epidermal-type fatty acid binding protein in alveolar macrophages and some alveolar type II epithelial cells in mouse lung. Histochem J 33(8):453–457
Ozerdem U, Grako KA, Dahlin-Huppe K, Monosov E, Stallcup WB (2001) NG2 proteoglycan is expressed exclusively by mural cells during vascular morphogenesis. Dev Dyn 222(2):218–227. https://doi.org/10.1002/dvdy.1200
Que J, Wilm B, Hasegawa H, Wang F, Bader D, Hogan BL (2008) Mesothelium contributes to vascular smooth muscle and mesenchyme during lung development. Proc Natl Acad Sci USA 105(43):16626–16630. https://doi.org/10.1073/pnas.0808649105
Rugh R (1990) The mouse. Its reproduction and development. Oxford University Press, Oxford
Schenk RK, Spiro D, Wiener J (1967) Cartilage resorption in the tibial epiphyseal plate of growing rats. J Cell Biol 34(1):275–291
Stallcup WB, You WK, Kucharova K, Cejudo-Martin P, Yotsumoto F (2016) NG2 proteoglycan-dependent contributions of pericytes and macrophages to brain tumor vascularization and progression. Microcirculation 23(2):122–133. https://doi.org/10.1111/micc.12251
Stapor PC, Sweat RS, Dashti DC, Betancourt AM, Murfee WL (2014) Pericyte dynamics during angiogenesis: new insights from new identities. J Vasc Res 51(3):163–174. https://doi.org/10.1159/000362276
Theiler K (2013) The house mouse: atlas of embryonic development. Springer Science and Business Media, New York
Trost A, Schroedl F, Lange S, Rivera FJ, Tempfer H, Korntner S, Stolt CC, Wegner M, Bogner B, Kaser-Eichberger A, Krefft K, Runge C, Aigner L, Reitsamer HA (2013) Neural crest origin of retinal and choroidal pericytes. Investig Ophthalmol Vis Sci 54(13):7910–7921. https://doi.org/10.1167/iovs.13-12946
Tussiwand R, Gautier EL (2015) Transcriptional regulation of mononuclear phagocyte development. Front Immunol 6:533. https://doi.org/10.3389/fimmu.2015.00533
Wilm B, Ipenberg A, Hastie ND, Burch JB, Bader DM (2005) The serosal mesothelium is a major source of smooth muscle cells of the gut vasculature. Development 132(23):5317–5328. https://doi.org/10.1242/dev.02141
Winkler EA, Bell RD, Zlokovic BV (2010) Pericyte-specific expression of PDGF beta receptor in mouse models with normal and deficient PDGF beta receptor signaling. Mol Neurodegener 5:32. https://doi.org/10.1186/1750-1326-5-32
Acknowledgements
The authors thank to Dr. Hisatake Kondo (Professor emeritus, Tohoku University) and Dr. Shoichi Iseki (Professor emeritus, Kanazawa University) for their critical advice in manuscript preparation.
Funding
This work was supported by JSPS KAKENHI Grant Numbers JP16K11457 to Y. B. and JP17K11626 to O. A. and Meikai University Miyata Research Grant (2016 A to Y. B.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Statement on the welfare of animals
All animal procedures were in accordance with the Guidelines for care and use of Laboratory Animals of Meikai University School of Dentistry, and these experiments were approved by Meikai University Animal Ethics Committee (A1708).
Rights and permissions
About this article
Cite this article
Bando, Y., Sakashita, H., Taira, F. et al. Origin and development of septoclasts in endochondral ossification of mice. Histochem Cell Biol 149, 645–654 (2018). https://doi.org/10.1007/s00418-018-1653-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-018-1653-1