Abstract
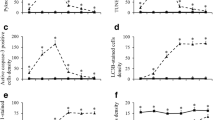
An important step in bromodeoxyuridine (BrdU) immunohistochemistry is the production of single-stranded DNA to make the incorporated BrdU accessible to the antibodies. This paper examines the effect of distinct DNA denaturation pretreatments (DNase I, sodium citrate buffer, endonuclease Eco RI and exonuclease III, and HCl hydrolysis) on detection of BrdU. We found that all the methods used in the partial denaturation of DNA combined good nuclear immunostaining with acceptable tissue integrity. We also observed that these immunohistochemical protocols revealed a spatial pattern in the distribution of DNA-synthesizing cells within the cerebellar external granular layer (EGL) of 10-day-old rats, allowing us to estimate the fraction of S-phase cells. Our results indicate that detection of BrdU-stained cells is affected by the distinct histological procedures used in such detection. Additionally, as the duration and phases of the cell cycle in EGL neuroblasts are estimated in accordance with BrdU detection, an effect on this detection can render the measurement of cell cycle inaccurate. The present work shows that DNase I and citrate buffer, at appropriate conditions, may be good alternatives for acid denaturation. However, they are less sensitive than autoradiographic techniques that use 3H-thymidine administration. Finally, current data reveal that short survival times after a single BrdU exposure do not seem to affect the cell cycle progression of the EGL neuroblasts.












Similar content being viewed by others
References
Altman J, Bayer SA (1997) Development of the cerebellar system: in relation to its evolution, structure and functions. CRC Press, Inc., Boca Raton
Anda S, Boye E, Grallert B (2014) Cell-cycle analyses using thymidine analogues in fission yeast. PLoS One 9(2):e88629. doi:10.1371/journal.pone.0088629
Borges S, Lewis PD (1983) Effects of ethanol on postnatal cell acquisition in the rat cerebellum. Brain Res 271:388–391
Böswald M, Harasim S, Maurer-Schultze B (1990) Tracer dose and availability time of thymidine and bromodeoxyuridine: application of bromodeoxyuridine in cell kinetic studies. Cell Tissue Kinet 23:169–181
Carletti B, Rossi F (2008) Neurogenesis in the cerebellum. Neurocientist 14:91–100. doi:10.1177/1073858407304629
Cawood AH, Savage JRK (1983) A comparison of the use of bromodeoxyuridine and 3H-thymidine in studies of the cell cycle. Cell Tissue Kinet 16:51–57
Chedotal A (2010) Should I stay or should I go? Becoming a granule cell. Trends Neurosci 33:163–172. doi:10.1016/j.tins.2010.01.004
Dinjens WN, ten Kate J, Lenders MH, van der Linden EP, Bosman FT (1992) Bromodeoxyuridine (BrdU) immunocytochemistry by exonuclease III (Exo III) digestion. Histochemistry 98:199–205
Dolbeare F (1995) Bromodeoxyuridine: a diagnostic tool in biology and medicine, Part I. Historical perspectives, histochemical methods and cell kinetics. Histochem J 27:339–369
Dolbeare F, Gray JW (1988) Use of restriction endonucleases and exonuclease III to expore halogenated pyrimidines for immunochemical staining. Cytometry 9:631–635
Duque A, Rakic P (2011) Different effects of BrdU and 3H-Thymidine incorporation into DNA on cell proliferation, position and fate. J Neurosci 31:15205–15217
Duque A, Rakic P (2015) Immunocytochemistry and related techniques. In: Merighi A, Lossi L (eds.) Identification of proliferating and migration cell by BrdU and other thymidine analogs: benefits and limitations. Chapter 7, vol. 101. Humana Press, New York, 123–139 doi:10.1007/978-1-4939-2313-7
Ffrench M, Morel F, Souchier C, Benchaib M, Catallo R, Bryon PA (1994) Choice of fixation and denaturation for the triple labelling of intra-cytoplasmic antigen, bromodeoxyuridine and DNA. Application to bone marrow plasma cells. Histochemistry 101:385–390
Gratzner HG (1982) Monoclonal antibody to 5-bromo- and 5-iododeoxyuridine. A new reagent for detection of DNA replication. Science 218:474–475
Hervás JP, Martí-Clúa J, Muñoz-García A, Santa-Cruz MC (2002) Proliferative activity in the cerebellar external granular layer evaluated by bromodeoxyuridine labeling. Biotech Histochem 77:27–35
Huard JMT, Forster CC, Carter ML, Sicinski P, Ross ME (1999) Cerebellar histogenesis is disturbed in mice lacking cyclin D2. Development 126:1927–1935
Lauder JM (1977) The effects of early hypo- and hyperthyroidism on the development of rat cerebellar cortex. III. Kinetics of cell proliferation in the external granular layer. Brain Res 126:31–51
Leuner B, Glasper ER, Gould E (2009) Thymidine analog methods for studies of adult neurogenesis are not equally sensitive. J Comp Neurol 517(2):123–133
Lewis PD, Balázs R, Patel AJ, Johson AL (1975) The effect of undernutrition in early life on cell generation in the rat brain. Brain Res 83:235–247
Lewis PD, Patel AJ, Johnson AL, Balázs R (1976) Effect of thyroid deficiency on cell acquistion in the postnatal rat brain: a quantitative histological study. Brain Res 104:49–62
Lewis PD, Patel AJ, Béndek G, Balázs R (1977) Effect of reserppine on cell proliferation in the developing rat brain: a quantitative histological study. Brain Res 129:299–308
Marti J, Santa-Cruz MC, Serra R, Hervas JP (2016) Hydroxyurea treatment and development of the rat cerebellum: effects on the neurogenetic profiles and settled patterns of Purkinje cells and deep cerebellar nuclei neurons. Neurotox Res 30:563–580
Martí J, Santa-Cruz MC, Serra R, Hervás JP (2015) Systematic differences in time of cerebellar-neuron origin derived from bromodeoxyuridine immunoperoxidase staining protocols and tritiated thymidine autoradiographic: a comparative study. Int J Dev Neurosci 47:216–228
Martínez S, Andreu A, Mecklenburg N, Echevarria D (2013) Cellular and molecular basis of cerebellar development. Front Neuroanat 7:18. doi:10.3389/fnana.2013.00018
Marzban H, Del Bigio MR, Alizadeh J, Ghavami S, Zachariah RM, Rastegar M (2015) Cellular commitment in the developing cerebellum. Front Cell Neurosci 12(8):450. doi:10.3389/fncel.2014.00450
Miller MW, Nowakowski RS (1988) Use of bromodeoxyuridine-immunohistochemistry to examine the proliferation, migration and time of origin of cells in the central nervous system. Brain Res 457:44–52
Nowakowski RS, Lewin SB, Miller MW (1989) Bromodeoxyuridine immunohistochemical determination of the lengths of the cell cycle and the DNA-synthetic phase for an anatomically defined population. J Neurocytol 18:311–318
Packard DS, Menzies RA, Skalko RG (1973) Incorporation of thymidine and its analog, bromodeoxyuridine, into embryos and maternal tissues of the mouse. Differentiation 1:397–405
Paxinos G, Watson C (1998) The rat brain in stereotaxic coordinates, 4th edn. Academic Press, San Diego
Sekerkova G, Ilijic E, Mugnaini E (2004a) Bromodeoxyuridine administered during neurogenesis of the projection neurons causes cerebellar defects in rat. J Comp Neurol. 470:221–239
Sekerkova G, Ilijic E, Mugnaini E (2004b) Time of origin of unipolar brush cells in the rat cerebellum as observed by prenatal bromodeoxyuridine labeling. Neuroscience 127:845–858
Shimada A, Shibata T, Komatsu K, Nifuji A (2008) Improved methods for immunohistochemical detection of BrdU in hard tissue. J Immunol Methods 339:11–16
Takahashi T, Nowakowski RS, Caviness VS Jr (1992) BUdR as an S-phase marker for quantitative studies of cytokinetic behaviour in the murine cerebral ventricular zone. J Neurocytol 21:185–197
Takahashi T, Goto T, Miyama S, Nowakowski RS, Caviness VS Jr (1999) Sequence of neuron origin and neocortical laminar fate: relation to cell cycle in the developing murine cerebral wall. J Neurocytol 19:10357–10371
Tang X, Fall DL, Li X, Lane T, Luskin MB (2007) Antigen-retrieval procedure for bromodeoxyuridine immunolabeling with concurrent labeling of nuclear DNA and antigens damaged by HCl pretreatment. J Neurosci 27:5837–5844
Taupin P (2007) BrdU immunohistochemistry for studying adult neurogenesis: paradigms, pitfalls, limitations, and validation. Brain Res Rev 53:198–214
Vanderlaan M, Thomas CB (1985) Characterization of monoclonal antibodies to bromodeoxyuridine. Cytometry 6:501–505
Wakayama T, Nakata H, Kumchantuek T, Gewaily MS, Iseki S (2015) Identification of 5-bromo-2′-deoxyuridine-labeled cells during mouse spermatogenesis by heat-induced antigen retrieval in lectin staining and immunohistochemistry. J Histochem Cytochem 63(3):190–205
Ye W, Mairet-Coello G, DiCicco-Bloom E (2007) DNase I pre-treatment markedly enhances detection of nuclear cycli-dependent kinase inhibitor p57Kip2 and BrdU double immunostaining in embryonic rat brain. Histochem Cell Biol 127:195–203
Acknowledgements
The authors are grateful to Dr María del Carmen Santa-Cruz and Dr José Pablo Hervás for providing the rats used in the experiments described in this discussion.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Molina, V., Rodríguez-Vázquez, L., Owen, D. et al. Cell cycle analysis in the rat external granular layer evaluated by several bromodeoxyuridine immunoperoxidase staining protocols. Histochem Cell Biol 148, 477–488 (2017). https://doi.org/10.1007/s00418-017-1593-1
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00418-017-1593-1