Abstract
Purpose
To identify common optical coherence tomography (OCT) characteristics of taxane-related CME (T-CME) to differentiate it from CME associated with other causes (O-CME) and to present multimodal imaging findings of T-CME.
Methods
To differentiate T-CME from O-CME, pooled SD-OCT images from 14 previous publications and images obtained from our multicenter case series of 3 patients with multimodal imaging of T-CME were compared with 16 consecutive cases of O-CME. Images were graded by 2 masked retinal specialists based on the presence of pre-specified OCT characteristics such as CME centered around fovea, outer retinal cysts more prominent compared with inner retinal cysts, continuous outer plexiform layer (OPL) and inner plexiform layer (IPL), intact outer retina layer, attenuation of outer retina layers by overlying retinal layers, and the presence of subretinal fluid.
Results
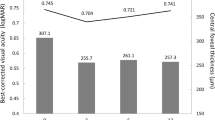
Comparing 19 and 16 SD-OCT images of T-CME and O-CME, respectively, T-CME showed a significantly higher rate of the continuous OPL and IPL layer and a higher composite score of the various pre-specified OCT features. All other individual features showed no significant difference between T-CME and O-CME. All our patients had T-CME that had vague petalloid patterns on the late-stage FFA, with late leakage on ICGA. OCT angiography in one case showed an intact foveal avascular zone.
Conclusions
T-CME is a rare but important complication of taxane chemotherapy. Specific OCT features such as an intact continuous OPL and IPL layer combined with other OCT features can help distinguish T-CME from O-CME, and early diagnosis is clinically important as cessation of taxanes before the retinal layers are disrupted may prevent permanent vision loss.




Similar content being viewed by others
References
Yokoe T, Fukada I, Kobayashi K et al (2017) Cystoid macular edema during treatment with paclitaxel and bevacizumab in a patient with metastatic breast cancer: a case report and literature review. Case Rep Oncol 10(2):605–612
Eisner A, Luoh S (2011) Breast cancer medications and vision: effects of treatments for early-stage disease. Curr Eye Res 36(10):867–885
Hofstra L, de Vries E, Willemse P (1997) Ophthalmic toxicity following paclitaxel infusion. Ann Oncol 8(10):1053–1053
Li J, Tripathi R, Tripathi B (2008) Drug-induced ocular disorders. Drug Saf 31(2):127–141
Teitelbaum B, Tresley D (2003) Cystic maculopathy with normal capillary permeability secondary to docetaxel. Optom Vis Sci 80(4):277–279
Joshi M (2007) Paclitaxel maculopathy. Arch Ophthalmol 125(5):709
Koo N, Kim Y (2012) A case of paclitaxel-induced maculopathy treated with methazolamide. Korean J Ophthalmol 26(5):394
Ehlers J, Rayess H, Steinle N (2012) Topical dorzolamide therapy for taxane-related macular oedema. Eye. 27(1):102–104
Ham D, Lee J, Kim H, Yun I (2012) A case of cystoid macular edema associated with paclitaxel chemotherapy. Korean J Ophthalmol 26(5):388
Murphy C, Walsh J, Hudis C, Lake D, Theodoulou M (2010) Cystoid macular edema secondary to nab-paclitaxel therapy. J Clin Oncol 28(33):e684–e687
Kuznetcova TI, Cech P, Herbort CP (2012) The mystery of angiographically silent macular oedema due to taxanes. Int Ophthalmol 32(3):299–304
Matsuoka N, Hasebe H, Mayama T, Fukuchi T (2015) Sub-tenon injections of triamcinolone acetonide had limited effect on cystoid macular edema secondary to nanoparticle albumin-bound paclitaxel (abraxane). Case Rep Ophthalmol Med 2015:1–4
Freitas-da-Costa P, Brandão E, Bragança T, Falcão-Reis F, Carneiro A (2015) Multimodal imaging in paclitaxel-induced macular edema: the microtubule disfunction. Cutan Ocul Toxicol 34(4):347–349
Hassall MM, Andrew NH (2016) Single-eye trial of a topical carbonic anhydrase inhibitor versus intravitreal bevacizumab for the treatment of taxane drug-induced cystoid macula oedema. BMJ Case Rep. https://doi.org/10.1136/bcr2015212733
Shih C-H, Lee Y-C (2018) Impaired retinal pigment epithelium in paclitaxel-induced macular edema. Medicine 97(26):e11229
Telander DG, Sarraf D (2007) Cystoid macular edema with docetaxel chemotherapy and the fluid retention syndrome. Semin Ophthalmol 22(3):151–153
Semb K, Aamdal S, Oian P (1998) Capillary protein leak syndrome appears to explain fluid retention in cancer patients who receive docetaxel treatment. J Clin Oncol 16(10):3426–3432
Spirn M, Warren F, Guyer D, Klancnik J, Spaide R (2003) Optical coherence tomography findings in nicotinic acid maculopathy. Am J Ophthalmol 135(6):913–914
Coscas G, Loewenstein A, Cunha-Vaz J, Soubrane G (2017) Macular edema, 2nd edn. S. Karger Ag, Basel, pp 1–10
Makri OE, Georgalas I, Georgakopoulos CD (2013) Drug-induced macular edema. Drugs. 73(8):789–802
Bassi E, Loizzi V, Furino C et al (2017) Cystoid macular edema secondary to paclitaxel therapy for ovarian cancer: a case report. Mol Clin Oncol 7(2):285–287
Nomi N, Ota M, Fukumura M et al (2017) Indocyanine green angiography findings of cystoid macular edema secondary to paclitaxel therapy. Jpn J Ophthalmol 62(2):163–167
Chelala E, Arej N, Antoun J et al (2017) Central macular thickness monitoring after a taxane-based therapy in visually asymptomatic patients. Chemotherapy. 62(3):199–204
Crabtree DV, Ojima I, Geng X, Adler AJ (2001) Tubulins in the primate retina: evidence that xanthophylls may be endogenous ligands for the paclitaxel-binding site. Bioorg Med Chem 9(8):1967–1976
Meyer K, Klink T, Ugurel S, Bröcker E (2011) Regression of paclitaxel-induced maculopathy with oral acetazolamide. Graefes Arch Clin Exp Ophthalmol 250(3):463–464
Author information
Authors and Affiliations
Contributions
AT and JP developed the study design, conducted the analysis, and prepared the manuscript and figures; KT and RO performed the analysis; MM and KBF were the senior authors who provided the cases, edited and reviewed the manuscript, and provided the scientific expert advice on the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
AT speaks and receives sponsorship for Zeis, Nidek, Bayer, and Novartis and travel grants from Allergan and Mandarin Optics. KBF is a consultant to Genentech, Optovue, Zeiss, Heidelberg Engineering, Allergan, and Novartis.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Health Insurance Portability and Accountability Act of 1996 and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
ESM 1
(DOCX 19.5 kb)
Rights and permissions
About this article
Cite this article
Perez, J.M., Teo, K., Ong, R. et al. Optical coherence tomography characteristics of taxane-induced macular edema and other multimodal imaging findings. Graefes Arch Clin Exp Ophthalmol 258, 1607–1615 (2020). https://doi.org/10.1007/s00417-020-04755-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-020-04755-z