Abstract
Purpose
The goal of this project was to demonstrate the feasibility of coupling the indirect ophthalmoscope laser delivery system with the 690 nm wavelength diode laser used to perform photodynamic therapy (PDT) in the treatment of retinoblastoma.
Methods
For phase 1, a total of six pigmented rabbits were treated with the indirect laser delivery system. The laser source was provided by the Lumenis Opal 690 nm laser unit, delivered through a 810 nm Indirect ophthalmoscope headpiece and a hand-held 28-diopter indirect lens (1.0 mm spot size). Four rabbits received intravenous verteporfin at doses of 0.43 or 0.86 mg/kg, and two rabbits did not receive verteporfin (controls). A second phase of the study involved eight rabbits using a retinoblastoma xenograft to determine the effect of indirect PDT on subretinal tumors.
Results
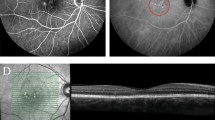
For phase 1, a total of 20 laser treatments were performed in the right eyes of six rabbits. Laser power levels ranged between 40 and 150 mW/cm2 and treatment duration ranged between 1 and 3 min. In the four rabbits that received verteporfin, focal retinal scars were noted at 40 mW/cm2 and higher power levels. In the two control rabbits that did not receive verteporfin, thermal burns were confirmed at 75 mW/cm2 and higher power levels. Histopathology showed focal retino-choroidal scars at the site of PDT treatment, without evidence of generalized ocular damage. Using the retinoblastoma xenograft, the indirect PDT system was shown to cause areas of tumor necrosis on histopathology.
Conclusions
The results of this pre-clinical study suggest verteporfin may be activated in the rabbit retina with the indirect delivery system and the 690 nm laser unit (i.e., Indirect PDT). Using verteporfin, treatment effects were observed at 40–50 mW/cm2 in the rabbit retina, while photocoagulation was achieved at 75 mW/cm2 and higher power levels. Fundoscopic and histopathologic examination of treated areas showed circumscribed areas of retinal damage and a lack of generalized ocular toxicity, suggesting that this modality may represent a safe and localized method for treating intraocular retinoblastoma.







Similar content being viewed by others
References
Fisher AM, Murphree AL, Gomer CJ (1995) Clinical and preclinical photodynamic therapy. Lasers Surg Med 17:2–31
Gomer CJ, Ferrario A, Hayashi N, Rucker N, Szirth BC, Murphree AL (1988) Molecular, cellular, and tissue responses following photodynamic therapy. Lasers Surg Med 8:450–463
Gomer CJ, Ferrario A, Murphree AL (1987) The effect of localized porphyrin photodynamic therapy on the induction of tumour metastasis. Br J Cancer 56:27–32
Gomer CJ, Hayashi N, Murphree AL (1987) The influence of sodium pentobarbital anesthesia on in vivo photodynamic therapy. Photochem Photobiol 46:843–846
Gomer CJ, Rucker N, Razum NJ, Murphree AL (1985) In vitro and in vivo light dose rate effects related to hematoporphyrin derivative photodynamic therapy. Cancer Res 45:1973–1977
Gomer CJ, Rucker N, Ferrario A, Murphree AL (1986) Expression of potentially lethal damage in Chinese hamster cells exposed to hematoporphyrin derivative photodynamic therapy. Cancer Res 46:3348–3352
Gomer CJ, Rucker N, Murphree AL (1988) Transformation and mutagenic potential of porphyrin photodynamic therapy in mammalian cells. Int J Radiat Biol Relat Stud Phys Chem Med 53:651–659
Murphree AL, Cote M, Gomer CJ (1987) The evolution of photodynamic therapy techniques in the treatment of intraocular tumors. Photochem Photobiol 46:919–923
Liu LH, Ni C (1983) Hematoporphyrin phototherapy for experimental intraocular malignant melanoma. Arch Ophthalmol 101:901–903
Sery TW, Dougherty TJ (1984) Photoradiation of rabbit ocular malignant melanoma sensitized with hematoporphyrin derivative. Curr Eye Res 3:519–528
Ohnishi Y, Yamana Y, Minei M (1986) Photoradiation therapy using argon laser and a hematoporphyrin derivative for retinoblastoma--a preliminary report. Jpn J Ophthalmol 30:409–419
Iridex (2014) Laser Indirect Ophthalmoscope (LIO) by Iridex. http://www.iridex.com/Portals/0/pdf/LT0011_LIO.pdf. Accessed 15 Jun 2015
Gomer CJ, Jester JV, Razum NJ, Szirth BC, Murphree AL (1985) Photodynamic therapy of intraocular tumors: examination of hematoporphyrin derivative distribution and long-term damage in rabbit ocular tissue. Cancer Res 45:3718–3725
Schefler AC, Cicciarelli N, Feuer W, Toledano S, Murray TG (2007) Macular retinoblastoma: evaluation of tumor control, local complications, and visual outcomes for eyes treated with chemotherapy and repetitive foveal laser ablation. Ophthalmology 114:162–169
Shields CL, Shields JA, Kiratli H, De Potter PV (1995) Treatment of retinoblastoma with indirect ophthalmoscope laser photocoagulation. J Pediatr Ophthalmol Strabismus 32:317–322
Masuyama Y, Fukuzaki M, Kodama Y, Baba Y, Sawada A (1984) Treatment of retinoblastoma with argon laser photocoagulation. J Pediatr Ophthalmol Strabismus 21:169–171
Augsburger JJ, Faulkner CB (1992) Indirect ophthalmoscope argon laser treatment of retinoblastoma. Ophthalmic Surg 23:591–593
Abramson DH, Schefler AC (2004) Transpupillary thermotherapy as initial treatment for small intraocular retinoblastoma: technique and predictors of success. Ophthalmology 111:984–991
Zhu D, Berry JL, Ediriwickrema L, Wong K, Lee TC, Murphree AL, Kim JW, Jubran R (2015) Long-term outcomes of group B eyes in patients with retinoblastoma treated with short-course chemoreduction: experience from Children’s hospital Los Angeles/University of Southern California. Ocul Oncol Pathol 2(2):105–111
Seshadri M, Belinier DA, Vaughan LA, Spernyak JA, Mazurchuk R, Foster TH, Henderson BW (2008) Light delivery over extended time periods enhances the effectiveness of photodynamic therapy. Clin Cancer Res 14:2796–2805
Henderson BW et al (2000) Photofrin photodynamic therapy can significantly deplete or preserve oxygenation in human basal cell carcinomas during treatment, depending on fluence rate. Cancer Res 60:525–529
Henderson BW, Gollnick SO, Snyder JW, Busch TM, Kousis PC, Cheney RT, Morgan J (2004) Choice of oxygen-conserving treatment regimen determines the inflammatory response and outcome of photodynamic therapy of tumors. Cancer Res 64:2120–2126
Richter AM, Cerruti-Sola S, Sternberg ED, Dolphin D, Levy JG (1990) Biodistribution of tritiated benzoporphyrin derivative (3H-BPD-MA), a new potent photosensitizer, in normal and tumor-bearing mice. J Photochem Photobiol B 5(2):231–244
Gomer CJ, Rucker N, Banerjee A, Benedict WF (1983) Comparison of mutagenicity and induction of sister chromatid exchange in Chinese hamster cells exposed to hematoporphyrin derivative photoradiation, ionizing radiation, or ultraviolet radiation. Cancer Res 43:2622–2627
Cerman E, Cekic O (2015) Clinical use of photodynamic therapy in ocular tumors. Surv Ophthalmol 60:557–574
Kaliki S, Shields CL, Al-Dahmash SA, Mashayekhi A, Shields JA (2012) Photodynamic therapy for choroidal metastasis in 8 cases. Ophthalmology 119:1218–1222
Hussain RN, Jmor F, Damato B, Heimann H (2015) Verteporfin photodynamic therapy for the treatment of retinal vasoproliferative tumors. Ophthalmology 122:2361–2363
Giansanti F, Virgili G, Varano M et al (2005) Photodynamic therapy for choroidal neovascularization in pediatric patients. Retina 25:590–596
Varano M, Iacono P, Giorno P, Chiaravalloti A, Parravano M (2014) Photodynamic therapy in subfoveal and juxtafoveal myopic choroidal neovascularization: a 10-year retrospective analysis. Ophthalmologica 231:204–210
Yildirim C, Cetin EN, Yayla K, Avunduk AM, Yaylali V (2011) Photodynamic therapy for unilateral idiopathic peripapillary choroidal neovascularization in a child. Int Ophthalmol 31:333–335
Chuang L, Hwang Y, Wang N et al (2014) The chorioretinal damage caused by different half parameters of photodynamic therapy in rabbits. J Ocul Pharmacol Ther 30(8):642–649
Framme C, Flucke B, Birngruber R (2004) Comparison of reduced and standard light application in photodynamic therapy of the eye in two rabbit models. Graefes Arch Clin Exp Ophthalmol 244(7):773–781
Kang SJ, Grossniklaus HE (2011) Rabbit model of retinoblastoma. J Biomed Biotechnol 2011:1–5
Funding
Research to Prevent Blindness and the Las Madrinas Endowment in Experimental Therapeutics.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge, or beliefs) in the subject matter or materials discussed in this manuscript.
Animal experiments
Ethical approval: “All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.”
University of California, Los Angeles: “All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.”
Rights and permissions
About this article
Cite this article
Kim, J.W., Jacobsen, B., Zolfaghari, E. et al. Rabbit model of ocular indirect photodynamic therapy using a retinoblastoma xenograft. Graefes Arch Clin Exp Ophthalmol 255, 2363–2373 (2017). https://doi.org/10.1007/s00417-017-3805-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-017-3805-8