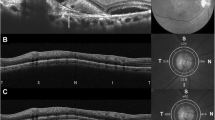
Abstract
Purpose
We aimed to compare changes in subfoveal and peripapillary choroidal thickness (CT) after intravitreal aflibercept or ranibizumab injections for neovascular age-related macular degeneration (AMD).
Methods
Medical records of 54 treatment-naïve, consecutive patients (54 eyes) who were diagnosed with neovascular AMD and received three monthly injections of aflibercept (21 eyes) or ranibizumab (33 eyes) were reviewed. Subfoveal and peripapillary CT were measured with images obtained using spectral domain optical coherence tomography at baseline and at three months.
Results
Subfoveal CT decreased from 232.2 ± 94.4 μm at baseline to 207.1 ± 89.3 μm at three months in the aflibercept group (p < 0.001) and from 231.5 ± 102.9 μm to 220.0 ± 98.0 μm in the ranibizumab group (p = 0.006). The reduction was greater in the aflibercept group than in the ranibizumab group (p = 0.024). Peripapillary CT decreased from 157.2 ± 62.2 μm at baseline to 147.4 ± 62.2 μm at three months in the aflibercept group (p < 0.001). However, the change in peripapillary CT from 154.9 ± 46.5 μm at baseline to 152.3 ± 50.0 μm at three months was not significant in the ranibizumab group (p = 0.123).
Conclusions
Intravitreally injected aflibercept significantly decreased subfoveal CT more than ranibizumab. Choroidal thinning after aflibercept injection was not limited to the subfoveal area, but extended beyond the macula as well.





Similar content being viewed by others

References
Lim LS, Mitchell P, Seddon JM, Holz FG, Wong TY (2012) Age-related macular degeneration. Lancet 379:1728–1738. doi:10.1016/S0140-6736(12)60282-7
Campbell JP, Bressler SB, Bressler NM (2012) Impact of availability of anti-vascular endothelial growth factor therapy on visual impairment and blindness due to neovascular age-related macular degeneration. Arch Ophthalmol 130:794–795. doi:10.1001/archophthalmol.2011.2480
Ferrara N, Damico L, Shams N, Lowman H, Kim R (2006) Development of ranibizumab, an anti-vascular endothelial growth factor antigen binding fragment, as therapy for neovascular age-related macular degeneration. Retina 26:859–870. doi:10.1097/01.iae.0000242842.14624.e7
Heier JS, Brown DM, Chong V, Korobelnik JF, Kaiser PK, Nguyen QD, Kirchhof B, Ho A, Ogura Y, Yancopoulos GD, Stahl N, Vitti R, Berliner AJ, Soo Y, Anderesi M, Groetzbach G, Sommerauer B, Sandbrink R, Simader C, Schmidt-Erfurth U, View GVS (2012) Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 119:2537–2548. doi:10.1016/j.ophtha.2012.09.006
Mitchell P, Korobelnik JF, Lanzetta P, Holz FG, Prunte C, Schmidt-Erfurth U, Tano Y, Wolf S (2010) Ranibizumab (Lucentis) in neovascular age-related macular degeneration: evidence from clinical trials. Br J Ophthalmol 94:2–13. doi:10.1136/bjo.2009.159160
Stewart MW (2012) Clinical and differential utility of VEGF inhibitors in wet age-related macular degeneration: focus on aflibercept. Clin Ophthalmol 6:1175–1186. doi:10.2147/OPTH.S33372
Stewart MW, Rosenfeld PJ (2008) Predicted biological activity of intravitreal VEGF Trap. Br J Ophthalmol 92:667–668. doi:10.1136/bjo.2007.134874
Kvanta A, Algvere PV, Berglin L, Seregard S (1996) Subfoveal fibrovascular membranes in age-related macular degeneration express vascular endothelial growth factor. Invest Ophthalmol Vis Sci 37:1929–1934
Blaauwgeers HG, Holtkamp GM, Rutten H, Witmer AN, Koolwijk P, Partanen TA, Alitalo K, Kroon ME, Kijlstra A, van Hinsbergh VW, Schlingemann RO (1999) Polarized vascular endothelial growth factor secretion by human retinal pigment epithelium and localization of vascular endothelial growth factor receptors on the inner choriocapillaris. Evidence for a trophic paracrine relation. Am J Pathol 155:421–428. doi:10.1016/S0002-9440(10)65138-3
Pournaras CJ, Rungger-Brandle E, Riva CE, Hardarson SH, Stefansson E (2008) Regulation of retinal blood flow in health and disease. Prog Retin Eye Res 27:284–330. doi:10.1016/j.preteyeres.2008.02.002
Saint-Geniez M, Kurihara T, Sekiyama E, Maldonado AE, D'Amore PA (2009) An essential role for RPE-derived soluble VEGF in the maintenance of the choriocapillaris. Proc Natl Acad Sci U S A 106:18751–18756. doi:10.1073/pnas.0905010106
Julien S, Biesemeier A, Taubitz T, Schraermeyer U (2014) Different effects of intravitreally injected ranibizumab and aflibercept on retinal and choroidal tissues of monkey eyes. Br J Ophthalmol 98:813–825. doi:10.1136/bjophthalmol-2013-304019
Peters S, Heiduschka P, Julien S, Ziemssen F, Fietz H, Bartz-Schmidt KU, Tubingen Bevacizumab Study G, Schraermeyer U (2007) Ultrastructural findings in the primate eye after intravitreal injection of bevacizumab. Am J Ophthalmol 143:995–1002. doi:10.1016/j.ajo.2007.03.007
Branchini L, Regatieri C, Adhi M, Flores-Moreno I, Manjunath V, Fujimoto JG, Duker JS (2013) Effect of intravitreous anti-vascular endothelial growth factor therapy on choroidal thickness in neovascular age-related macular degeneration using spectral-domain optical coherence tomography. JAMA Ophthalmol 131:693–694. doi:10.1001/jamaophthalmol.2013.692
Gharbiya M, Cruciani F, Mariotti C, Grandinetti F, Marenco M, Cacace V (2015) Choroidal thickness changes after Intravitreal antivascular endothelial growth factor therapy for age-related macular degeneration: ranibizumab versus aflibercept. J Ocul Pharmacol Ther 31:357–362. doi:10.1089/jop.2014.0160
Hata M, Oishi A, Tsujikawa A, Yamashiro K, Miyake M, Ooto S, Tamura H, Nakanishi H, Takahashi A, Yoshikawa M, Yoshimura N (2014) Efficacy of intravitreal injection of aflibercept in neovascular age-related macular degeneration with or without choroidal vascular hyperpermeability. Invest Ophthalmol Vis Sci 55:7874–7880. doi:10.1167/iovs.14-14610
Koizumi H, Kano M, Yamamoto A, Saito M, Maruko I, Kawasaki R, Sekiryu T, Okada AA, Iida T (2015) Short-term changes in choroidal thickness after aflibercept therapy for neovascular age-related macular degeneration. Am J Ophthalmol 159:627–633. doi:10.1016/j.ajo.2014.12.025
Mazaraki K, Fassnacht-Riederle H, Blum R, Becker M, Michels S (2015) Change in choroidal thickness after intravitreal aflibercept in pretreated and treatment-naive eyes for neovascular age-related macular degeneration. Br J Ophthalmol. doi:10.1136/bjophthalmol-2015-306636
Yamazaki T, Koizumi H, Yamagishi T, Kinoshita S (2012) Subfoveal choroidal thickness after ranibizumab therapy for neovascular age-related macular degeneration: 12-month results. Ophthalmology 119:1621–1627. doi:10.1016/j.ophtha.2012.02.022
Hayreh SS (2004) Posterior ciliary artery circulation in health and disease: the Weisenfeld lecture. Invest Ophthalmol Vis Sci 45:749–757, 748
Vujosevic S, Martini F, Cavarzeran F, Pilotto E, Midena E (2012) Macular and peripapillary choroidal thickness in diabetic patients. Retina 32:1781–1790. doi:10.1097/IAE.0b013e31825db73d
Yun C, Oh J, Ahn SE, Hwang SY, Kim SW, Huh K (2015) Peripapillary choroidal thickness in patients with early age-related macular degeneration and reticular pseudodrusen. Graefes Arch Clin Exp Ophthalmol. doi:10.1007/s00417-015-3054-7
Yun C, Oh J, Han JY, Hwang SY, Moon SW, Huh K (2015) Peripapillary choroidal thickness in central serous chorioretinopathy: is choroid outside the macula also thick? Retina. doi:10.1097/IAE.0000000000000539
Lengyel I, Csutak A, Florea D, Leung I, Bird AC, Jonasson F, Peto T (2015) A population-based ultra-Widefield digital image grading study for age-related macular degeneration-like lesions at the peripheral retina. Ophthalmology 122:1340–1347. doi:10.1016/j.ophtha.2015.03.005
Koh AH, Expert PCVP, Chen LJ, Chen SJ, Chen Y, Giridhar A, Iida T, Kim H, Yuk Yau Lai T, Lee WK, Li X, Han Lim T, Ruamviboonsuk P, Sharma T, Tang S, Yuzawa M (2013) Polypoidal choroidal vasculopathy: evidence-based guidelines for clinical diagnosis and treatment. Retina 33:686–716. doi:10.1097/IAE.0b013e3182852446
Ahuja RM, Stanga PE, Vingerling JR, Reck AC, Bird AC (2000) Polypoidal choroidal vasculopathy in exudative and haemorrhagic pigment epithelial detachments. Br J Ophthalmol 84:479–484
Perkovich BT, Zakov ZN, Berlin LA, Weidenthal D, Avins LR (1990) An update on multiple recurrent serosanguineous retinal pigment epithelial detachments in black women. Retina 10:18–26
De Salvo G, Vaz-Pereira S, Keane PA, Tufail A, Liew G (2015) Sensitivity and specificity of spectral-domain optical coherence tomography in detecting idiopathic polypoidal choroidal vasculopathy. Am J Ophthalmol 158:1228–1238.e1. doi:10.1016/j.ajo.2014.08.025
Freund KB, Zweifel SA, Engelbert M (2010) Do we need a new classification for choroidal neovascularization in age-related macular degeneration? Retina 30:1333–1349. doi:10.1097/IAE.0b013e3181e7976b
Macular Photocoagulation Study Group (1991) Subfoveal neovascular lesions in age-related macular degeneration. Guidelines for evaluation and treatment in the macular photocoagulation study. Arch Ophthalmol 109:1242–1257
Freund KB, Ho IV, Barbazetto IA, Koizumi H, Laud K, Ferrara D, Matsumoto Y, Sorenson JA, Yannuzzi L (2008) Type 3 neovascularization: the expanded spectrum of retinal angiomatous proliferation. Retina 28:201–211. doi:10.1097/IAE.0b013e3181669504
Griffith JF, Goldberg JL (2015) Prevalence of comorbid retinal disease in patients with glaucoma at an academic medical center. Clin Ophthalmol 9:1275–1284. doi:10.2147/OPTH.S85851. eCollection 2015
Spaide RF, Koizumi H, Pozzoni MC (2008) Enhanced depth imaging spectral-domain optical coherence tomography. Am J Ophthalmol 146:496–500. doi:10.1016/j.ajo.2008.05.032
Yiu G, Pecen P, Sarin N, Chiu SJ, Farsiu S, Mruthyunjaya P, Toth CA (2014) Characterization of the choroid-scleral junction and suprachoroidal layer in healthy individuals on enhanced-depth imaging optical coherence tomography. JAMA Ophthalmol 132:174–181. doi:10.1001/jamaophthalmol.2013.7288
Oh J, Yoo C, Yun CM, Yang KS, Kim SW, Huh K (2013) Simplified method to measure the peripapillary choroidal thickness using three-dimensional optical coherence tomography. Korean J Ophthalmol 27:172–177. doi:10.3341/kjo.2013.27.3.172
Papadopoulos N, Martin J, Ruan Q, Rafique A, Rosconi MP, Shi E, Pyles EA, Yancopoulos GD, Stahl N, Wiegand SJ (2012) Binding and neutralization of vascular endothelial growth factor (VEGF) and related ligands by VEGF Trap, ranibizumab and bevacizumab. Angiogenesis 15:171–185. doi:10.1007/s10456-011-9249-6
Zehetner C, Kralinger MT, Modi YS, Waltl I, Ulmer H, Kirchmair R, Bechrakis NE, Kieselbach GF (2015) Systemic levels of vascular endothelial growth factor before and after intravitreal injection of aflibercept or ranibizumab in patients with age-related macular degeneration: a randomised, prospective trial. Acta Ophthalmol 93:e154–e159. doi:10.1111/aos.12604
Lowe J, Araujo J, Yang J, Reich M, Oldendorp A, Shiu V, Quarmby V, Lowman H, Lien S, Gaudreault J, Maia M (2007) Ranibizumab inhibits multiple forms of biologically active vascular endothelial growth factor in vitro and in vivo. Exp Eye Res 85:425–430. doi:10.1016/j.exer.2007.05.008
dell’Omo R, Cassetta M, dell’Omo E, di Salvatore A, Hughes JM, Aceto F, Porcellini A, Costagliola C (2012) Aqueous humor levels of vascular endothelial growth factor before and after intravitreal bevacizumab in type 3 versus type 1 and 2 neovascularization. A prospective, case-control study. Am J Ophthalmol 153:155–161.e152. doi:10.1016/j.ajo.2011.06.001
Witmer AN, Vrensen GF, Van Noorden CJ, Schlingemann RO (2003) Vascular endothelial growth factors and angiogenesis in eye disease. Prog Retin Eye Res 22:1–29
Papadopoulou DN, Mendrinos E, Mangioris G, Donati G, Pournaras CJ (2009) Intravitreal ranibizumab may induce retinal arteriolar vasoconstriction in patients with neovascular age-related macular degeneration. Ophthalmology 116:1755–1761. doi:10.1016/j.ophtha.2009.03.017
Sacu S, Pemp B, Weigert G, Matt G, Garhofer G, Pruente C, Schmetterer L, Schmidt-Erfurth U (2011) Response of retinal vessels and retrobulbar hemodynamics to intravitreal anti-VEGF treatment in eyes with branch retinal vein occlusion. Invest Ophthalmol Vis Sci 52:3046–3050. doi:10.1167/iovs.10-5842
Kawashima Y, Oishi A, Tsujikawa A, Yamashiro K, Miyake M, Ueda-Arakawa N, Yoshikawa M, Takahashi A, Yoshimura N (2014) Effects of aflibercept for ranibizumab-resistant neovascular age-related macular degeneration and polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol. doi:10.1007/s00417-014-2838-5
Pinheiro-Costa J, Costa JM, Beato JN, Freitas-da-Costa P, Brandao E, Falcao MS, Falcao-Reis F, Carneiro AM (2015) Switch to aflibercept in the treatment of neovascular AMD: one-year results in clinical practice. Ophthalmologica 233:155–161. doi:10.1159/000381221
Razavi S, Souied EH, Darvizeh F, Querques G (2015) Assessment of choroidal topographic changes by swept-source optical coherence tomography after Intravitreal Ranibizumab for exudative age-related macular degeneration. Am J Ophthalmol 160:1006–1013. doi:10.1016/j.ajo.2015.08.009
Grunwald JE, Daniel E, Huang J, Ying GS, Maguire MG, Toth CA, Jaffe GJ, Fine SL, Blodi B, Klein ML, Martin AA, Hagstrom SA, Martin DF, CATT Research Group (2014) Risk of geographic atrophy in the comparison of age-related macular degeneration treatments trials. Ophthalmology 121:150–161. doi:10.1016/j.ophtha.2013.08.015
Grunwald JE, Pistilli M, Ying GS, Maguire MG, Daniel E, Martin DF, Comparison of Age-related Macular Degeneration Treatments Trials Research Group (2015) Growth of geographic atrophy in the comparison of age-related macular degeneration treatments trials. Ophthalmology 122:809–816. doi:10.1016/j.ophtha.2014.11.007
Young M, Chui L, Fallah N, Or C, Merkur AB, Kirker AW, Albiani DA, Forooghian F (2014) Exacerbation of choroidal and retinal pigment epithelial atrophy after anti-vascular endothelial growth factor treatment in neovascular age-related macular degeneration. Retina 34:1308–1315. doi:10.1097/IAE.0000000000000081
Xu L, Mrejen S, Jung JJ, Gallego-Pinazo R, Thompson D, Marsiglia M, Freund KB (2015) Geographic atrophy in patients receiving anti-vascular endothelial growth factor for neovascular age-related macular degeneration. Retina 35:176–186. doi:10.1097/IAE.0000000000000374
Cho HJ, Lee TG, Han SY, Kim HS, Kim JH, Han JI, Lew YJ, Kim JW (2015) Long-term visual outcome and prognostic factors of Intravitreal anti-vascular endothelial growth factor treatment for retinal angiomatous proliferation. Graefes Arch Clin Exp Ophthalmol. doi:10.1007/s00417-015-2993-3
Fujiwara A, Shiragami C, Shirakata Y, Manabe S, Izumibata S, Shiraga F (2012) Enhanced depth imaging spectral-domain optical coherence tomography of subfoveal choroidal thickness in normal Japanese eyes. Jpn J Ophthalmol 56:230–235. doi:10.1007/s10384-012-0128-5
Wei WB, Xu L, Jonas JB, Shao L, Du KF, Wang S, Chen CX, Xu J, Wang YX, Zhou JQ, You QS (2013) Subfoveal choroidal thickness: the Beijing Eye Study. Ophthalmology 120:175–180. doi:10.1016/j.ophtha.2012.07.048
Kim SW, Oh J, Kwon SS, Yoo J, Huh K (2011) Comparison of choroidal thickness among patients with healthy eyes, early age-related maculopathy, neovascular age-related macular degeneration, central serous chorioretinopathy, and polypoidal choroidal vasculopathy. Retina 31:1904–1911. doi:10.1097/IAE.0b013e31821801c5
Gupta P, Cheung CY, Baskaran M, Tian J, Marziliano P, Lamoureux EL, Cheung CM, Aung T, Wong TY, Cheng CY (2015) Relationship between peripapillary choroid and retinal nerve fiber layer thickness in a population-based sample of nonglaucomatous eyes. Am J Ophthalmol. doi:10.1016/j.ajo.2015.09.018
Huang W, Wang W, Zhou M, Chen S, Gao X, Fan Q, Ding X, Zhang X (2013) Peripapillary choroidal thickness in healthy Chinese subjects. BMC Ophthalmol 13:23. doi:10.1186/1471-2415-13-23
Laviers H, Zambarakji H (2014) Enhanced depth imaging-OCT of the choroid: a review of the current literature. Graefes Arch Clin Exp Ophthalmol 252:1871–1883. doi:10.1007/s00417-014-2840-y
Adhi M, Duker JS (2013) Optical coherence tomography—current and future applications. Curr Opin Ophthalmol 24:213–221. doi:10.1097/ICU.0b013e32835f8bf8
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
Korea University provided financial support in the form of research grant (grant number K1421461). The sponsor had no role in the design or conduct of this research.
Conflict of interest
All authors certify that they have no affiliations with or involvement in any organization or entity with any financial interest (such as honoraria; educational grants; participation in speakers’ bureaus; membership, employment, consultancies, stock ownership, or other equity interest; and expert testimony or patent-licensing arrangements), or non-financial interest (such as personal or professional relationships, affiliations, knowledge or beliefs) in the subject matter or materials discussed in this manuscript.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. For this type of study formal consent is not required.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Electronic Supplementary Material 1
(PDF 10 kb)
Electronic Supplementary Material 2
(PDF 14 kb)
Electronic Supplementary Material 3
(PDF 69 kb)
Rights and permissions
About this article
Cite this article
Yun, C., Oh, J., Ahn, J. et al. Comparison of intravitreal aflibercept and ranibizumab injections on subfoveal and peripapillary choroidal thickness in eyes with neovascular age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol 254, 1693–1702 (2016). https://doi.org/10.1007/s00417-015-3260-3
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-015-3260-3