Abstract
Purpose
To evaluate the short-term efficacy of aflibercept monotherapy for patients with treatment-naïve polypoidal choroidal vasculopathy (PCV).
Design
Prospective, consecutive case series.
Methods
Thirty-three consecutive eyes of 33 symptomatic PCV patients (17 men, 16 women, mean age 75 ± 8.7 years), not treated previously, received an intravitreal injection of 2.0 mg of aflibercept monthly for 3 months. Changes in best-corrected visual acuity (BCVA), optical coherence tomography (OCT) findings, and indocyanine green angiography (ICGA) findings 3 months after initial injection were evaluated.
Results
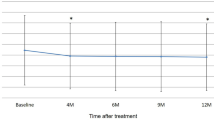
Compared with baseline, mean BCVA at 3-month visit significantly improved (0.40 ± 0.34 vs 0.22 ± 0.20 log minimum angle of resolution [logMAR] unit, P < 12 0.001). Eight eyes (24 %) showed improvement in BCVA ≥ 0.3 logMAR unit, and no eyes (0 %) showed a decrease in BCVA of ≥ 0.3 logMAR unit. Mean foveal thickness improved significantly (348 ± 184 μm at baseline vs 194 ± 32 μm at 3-month visit, P < 15 0.001). At 3-month visit, 31 eyes (97 %) achieved dry macula evaluated on OCT. Polypoidal lesions disappeared completely on ICGA in 16 eyes (48 %), and the number and/or the size of polypoidal lesions decreased in nine eyes (27 %). The remaining eight eyes (24 %) had unchanged polypoidal lesions. A branching vascular network remained and was unchanged in diameter in all 27 eyes in which it was detected at baseline.
Conclusion
Intravitreal aflibercept was well-tolerated in patients with treatment-naïve PCV over the short-term.



Similar content being viewed by others
References
Yannuzzi LA, Sorenson J, Spaide RF, Lipson B (1990) Idiopathic polypoidal choroidal vasculopathy (IPCV). Retina 10:1–8
Spaide RF, Yannuzzi LA, Slakter JS, Sorenson J, Orlach DA (1995) Indocyanine green videoangiography of idiopathic polypoidal choroidal vasculopathy. Retina 15:100–110
Yannuzzi LA, Ciardella A, Spaide RF, Rabb M, Freund KB, Orlock DA (1997) The expanding clinical spectrum of idiopathic polypoidal choroidal vasculopathy. Arch Ophthalmol 115:478–485
Yannuzzi LA, Wong DW, Sforzolini BS, Goldbaum M, Tang KC, Spaide RF, Freund KB, Slakter JS, Guyer DR, Sorenson JA, Fisher Y, Maberley D, Orlock DA (1999) Polypoidal choroidal vasculopathy and neovascularized age-related macular degeneration. Arch Ophthalmol 117:1503–1510
Ciardella AP, Donsoff IM, Huang SJ, Costa DL, Yannuzzi LA (2004) Polypoidal choroidal vasculopathy. Surv Ophthalmol 49:25–37
Laude A, Tan LE, Wilson CG, Lascaratos G, Elashry M, Aslam T, Patton N, Dhillon B (2010) Intravitreal therapy for neovascular age-related macular degeneration and inter-individual variations in vitreous pharmacokinetics. Prog Retin Eye Res 29:466–475
Gomi F, Ohji M, Sayanagi K, Sawa M, Sakaguchi H, Oshima Y, Ikuno Y, Tano Y (2008) One-year outcomes of photodynamic therapy in age-related macular degeneration and polypoidal choroidal vasculopathy in Japanese patients. Ophthalmology 115:141–146
Hikichi T, Ohtsuka H, Higuchi M, Matsushita T, Ariga H, Kosaka S, Matsushita R, Takami K (2010) Improvement of angiographic findings of polypoidal choroidal vasculopathy after intravitreal injection of ranibizumab monthly for 3 months. Am J Ophthalmol 150:674–682
Koizumi H, Yamagishi T, Yamazaki T, Kinoshita S (2011) Predictive factors of resolved retinal fluid after intravitreal ranibizumab for polypoidal choroidal vasculopathy. Br J Ophthalmol 95:1555–1559
Koh A, Lee WK, Chen LJ, Chen SJ, Hashad Y, Kim H, Lai TY, Pilz S, Ruamviboonsuk P, Tokaji E, Weisberger A, Lim TH (2012) Everest study: efficacy and safety of verteporfin photodynamic therapy in combination with ranibizumab or alone versus ranibizumab monotherapy in patients with symptomatic macular polypoidal choroidal vasculopathy. Retina 32:1453–1464
Gomi F, Sawa M, Sakaguchi H, Tsujikawa M, Oshima Y, Kamei M, Tano Y (2008) Efficacy of intravitreal bevacizumab for polypoidal choroidal vasculopathy. Br J Ophthalmol 92:70–73
Lai TY, Chan WM, Liu DT, Luk FO, Lam DS (2008) Intravitreal bevacizumab (Avastin) with or without photodynamic therapy for the treatment of polypoidal choroidal vasculopathy. Br J Ophthalmol 92:661–666
Akaza E, Yuzawa M, Mori R (2011) Three-year follow-up results of photodynamic therapy for polypoidal choroidal vasculopathy. Jpn J Ophthalmol 55:39–44
Cho M, Barbazetto IA, Freund KB (2009) Refractory neovascular age-related macular degeneration secondary to polypoidal choroidal vasculopathy. Am J Ophthalmol 148:70–78
Stangos AN, Gandhi JS, Nair-Sahni J, Heimann H, Pournaras CJ, Harding SP (2010) Polypoidal choroidal vasculopathy masquerading as neovascular age-related macular degeneration refractory to ranibizumab. Am J Ophthalmol 150:666–673
Stewart MW, Rosenfeld PJ (2008) Predicted biological activity of intravitreal VEGF Trap. Br J Ophthalmol 92:667–668
Papadopoulos N, Martin J, Ruan Q, Rafique A, Rosconi MP, Shi E, Pyles EA, Yancopoulos GD, Stahl N, Wiegand SJ (2012) Binding and neutralization of vascular endothelial growth factor (VEGF) and related ligands by VEGF Trap, ranibizumab and bevacizumab. Angiogenesis 15:171–185
Heier JS, Brown DM, Chong V, Korobelnik JF, Kaiser PK, Nguyen QD, Kirchhof B, Ho A, Ogura Y, Yancopoulos GD, Stahl N, Vitti R, Berliner AJ, Soo Y, Anderesi M, Groetzbach G, Sommerauer B, Sandbrink R, Simader C, Schmidt-Erfurth U; VIEW 1 and VIEW 2 Study Groups (2012) Intravitreal aflibercept (VEGF trap-eye) in wet age-related macular degeneration. Ophthalmology 119:2537–2548
Kumar N, Marsiglia M, Mrejen S, Fung AT, Slakter J, Sorenson J, Freund KB (2013) Visual and anatomical outcomes of intravitreal aflibercept in eyes with persistent subfoveal fluid despite previous treatments with ranibizumab in patients with neovascular age-related macular degeneration. Retina 33:1605–1612
Yonekawa Y, Andreoli C, Miller JB, Loewenstein JI, Sobrin L, Eliott D, Vavvas DG, Miller JW, Kim IK (2013) Conversion to aflibercept for chronic refractory or recurrent neovascular age-related macular degeneration. Am J Ophthalmol 156:29–35
Bakall B, Folk JC, Boldt HC, Sohn EH, Stone EM, Russell SR, Mahajan VB (2013) Aflibercept therapy for exudative age-related macular degeneration resistant to bevacizumab and ranibizumab. Am J Ophthalmol 156:15–22
Ho VY, Yeh S, Olsen TW, Bergstrom CS, Yan J, Cribbs BE, Hubbard GB 3rd (2013) Short-term outcomes of aflibercept for neovascular age-related macular degeneration in eyes previously treated with other vascular endothelial growth factor inhibitors. Am J Ophthalmol 156:23–28
Chang AA, Li H, Broadhead GK, Hong T, Schlub TE, Wijeyakumar W, Zhu M (2014) Intravitreal aflibercept for treatment-resistant neovascular age-related macular degeneration. Ophthalmology 121(1):188–192. doi:10.1016/j.ophtha.2013.08.035
Miura M, Iwasaki T, Goto H (2013) Intravitreal aflibercept for polypoidal choroidal vasculopathy after developing ranibizumab tachyphylaxis. Clin Ophthalmol 7:1591–1595. doi:10.2147/OPTH.S50634
Japanese study group of polypoidal choroidal vasculopathy (2005) Criteria for diagnosis of polypoidal choroidal vasculopathy [in japanese]. Nippon Ganka Gakkai Zasshi 109:417–427
Tano Y, Ohji M, Ishibashi T, Shiraga F, Tokoro T, Yuzawa M, Yoshimura N (2009) Re-treatment guideline of ranibizumab (genetical recombination) in the maintenance phase. Nihon Ganka Gakkai Zasshi 113:1098–1103
Yuzawa M, Mori R, Haruyama M (2003) A study of laser photocoagulation for polypoidal choroidal vasculopathy. Jpn J Ophthalmol 47:379–384
Wakabayashi T, Gomi F, Sawa M, Tsujikawa M, Tano Y (2008) Marked vascular changes of polypoidal choroidal vasculopathy after photodynamic therapy. Br J Ophthalmol 92:936–940
Rakic JM, Lambert V, Devy L, Luttun A, Carmeliet P, Claes C, Nguyen L, Foidart JM, Noël A, Munaut C (2003) Placental growth factor, a member of the VEGF family, contributes to the development of choroidal neovascularization. Invest Ophthalmol Vis Sci 44:3186–3193
Zhang F, Tang Z, Hou X, Lennartsson J, Li Y, Koch AW, Scotney P, Lee C, Arjunan P, Dong L, Kumar A, Rissanen TT, Wang B, Nagai N, Fons P, Fariss R, Zhang Y, Wawrousek E, Tansey G, Raber J, Fong GH, Ding H, Greenberg DA, Becker KG, Herbert JM, Nash A, Yla-Herttuala S, Cao Y, Watts RJ, Li X (2009) VEGF-B is dispensable for blood vessel growth but critical for their survival, and VEGF-B targeting inhibits pathological angiogenesis. Proc Natl Acad Sci U S A 106:6152–6157
Uyama M, Matsubara T, Fukushima I, Matsunaga H, Iwashita K, Nagai Y, Takahashi K (1999) Idiopathic polypoidal choroidal vasculopathy in Japanese patients. Arch Ophthalmol 117:1035–1042
Uyama M, Wada M, Nagai Y, Matsubara T, Matsunaga H, Fukushima I, Takahashi K, Matsumura M (2002) Polypoidal choroidal vasculopathy: natural history. Am J Ophthalmol 133:639–648
Lai TY, Chan WM, Liu DT, Lam DS (2009) Intravitreal ranibizumab with or without photodynamic therapy for the treatment of symptomatic polypoidal choroidal vasculopathy. Retina 29:750–756
Acknowledgments
The authors have no proprietary interest in any aspect of this report, and they report no financial support or financial conflict of interest. Involvement in tasks as follows: conception and design of study (S.I.), analysis and interpretation (S.I., K.S.), writing of the article (S.I.), critical revision of the article (S.I., K.S.), final approval of the article (S.I., K.S.), and data collection (S.I.). The current research followed the tenets of the Declaration of Helsinki, and informed consent was obtained from all subjects after explanation of the study protocol. The institutional review board at the Kanazawa University Hospital approved the study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ijiri, S., Sugiyama, K. Short-term efficacy of intravitreal aflibercept for patients with treatment-naïve polypoidal choroidal vasculopathy. Graefes Arch Clin Exp Ophthalmol 253, 351–357 (2015). https://doi.org/10.1007/s00417-014-2707-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00417-014-2707-2