Abstract
Background
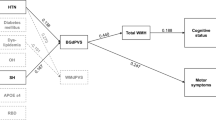
Orthostatic hypotension (OH) is a potential modifiable risk factor for cognitive impairment in patients with Parkinson’s disease (PD). Although other risk factors for dementia, hyposmia and REM sleep behavior disorder (RBD), are closely associated with autonomic dysfunction in PD, little is known about how these risk factors influence cognitive function and cerebral pathology.
Objective
We investigated how these three factors contribute to gray matter atrophy by considering the interaction of OH with hyposmia and RBD.
Methods
We analyzed cortical thickness, subcortical gray matter volume, and cognitive measures from 78 patients with de novo PD who underwent the head-up tilt test for the diagnosis of OH.
Results
Whole-brain analyses with Monte Carlo corrections revealed that hyposmia was associated with decreased cortical thickness in a marginal branch of the cingulate sulcus among patients with OH, and cortical thickness in this area correlated with cognitive functioning only in patients with OH. Subcortical gray matter volume analysis indicated that severe RBD was associated with decreased volume in the left hippocampus and bilateral amygdala among patients with OH.
Conclusion
Even in early PD, OH exerts effects on gray matter atrophy and cognitive dysfunction by interacting with RBD and hyposmia. OH might exacerbate cerebral pathology induced by hyposmia or RBD.



Similar content being viewed by others
Data availability
The data sets of the current study are available from the corresponding author upon reasonable request.
References
Longardner K, Bayram E, Litvan I (2020) Orthostatic hypotension is associated with cognitive decline in Parkinson disease. Front Neurol 11:897. https://doi.org/10.3389/fneur.2020.00897
Barrio IR, Miki Y, Jaunmuktane ZT et al (2022) Association between orthostatic hypotension and dementia in patients with Parkinson disease and multiple system atrophy. Neurology 100:e998–e1008. https://doi.org/10.1212/wnl.0000000000201659
Baba T, Kikuchi A, Hirayama K et al (2012) Severe olfactory dysfunction is a prodromal symptom of dementia associated with Parkinson’s disease: a 3 year longitudinal study. Brain 135:161–169. https://doi.org/10.1093/brain/awr321
Pagano G, De Micco R, Yousaf T et al (2018) REM behavior disorder predicts motor progression and cognitive decline in Parkinson disease. Neurology 91:e894–e905. https://doi.org/10.1212/WNL.0000000000006134
Palma JA, Kaufmann H (2017) Epidemiology, diagnosis, and management of neurogenic orthostatic hypotension. Mov Disord Clin Pract 4:298–308. https://doi.org/10.1002/mdc3.12478
McDonald C, Newton JL, Burn DJ (2016) Orthostatic hypotension and cognitive impairment in Parkinson’s disease: causation or association? Mov Disord 31:937–946. https://doi.org/10.1002/mds.26632
Ten Harmsen BL, van Rumund A, Aerts MB et al (2018) Clinical correlates of cerebral white matter abnormalities in patients with Parkinson’s disease. Parkinsonism Relat Disord 49:28–33. https://doi.org/10.1016/j.parkreldis.2017.12.029
Pilotto A, Romagnolo A, Scalvini A et al (2021) Association of orthostatic hypotension with cerebral atrophy in patients with Lewy body disorders. Neurology 97:e814–e824. https://doi.org/10.1212/WNL.0000000000012342
Riboldi GM, Russo MJ, Pan L et al (2022) Dysautonomia and REM sleep behavior disorder contributions to progression of Parkinson’s disease phenotypes. NPJ Parkinsons Dis 8:110. https://doi.org/10.1038/s41531-022-00373-0
Mizutani Y, Nakamura T, Okada A et al (2014) Hyposmia and cardiovascular dysautonomia correlatively appear in early-stage Parkinson’s disease. Parkinsonism Relat Disord 20:520–524. https://doi.org/10.1016/j.parkreldis.2014.02.010
Oka H, Toyoda C, Yogo M, Mochio S (2010) Olfactory dysfunction and cardiovascular dysautonomia in Parkinson’s disease. J Neurol 257:969–976. https://doi.org/10.1007/s00415-009-5447-1
Goldstein DS, Sewell L, Holmes C (2010) Association of anosmia with autonomic failure in Parkinson disease. Neurology 74:245–251
Oltra J, Uribe C, Segura B et al (2022) Brain atrophy pattern in de novo Parkinson’s disease with probable RBD associated with cognitive impairment. NPJ Parkinsons Dis 8:60. https://doi.org/10.1038/s41531-022-00326-7
Lee EY, Eslinger PJ, Du G et al (2014) Olfactory-related cortical atrophy is associated with olfactory dysfunction in Parkinson’s disease. Mov Disord 29:1205–1208. https://doi.org/10.1002/mds.25829
Postuma RB, Berg D, Stern M et al (2015) MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord 30:1591–1601. https://doi.org/10.1002/mds.26424
Goetz CG, Tilley BC, Shaftman SR et al (2008) Movement Disorder Society-Sponsored Revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord 23:2129–2170. https://doi.org/10.1002/mds.22340
Dubois B, Slachevsky A, Litvan I, Pillon B (2000) The FAB A frontal assessment battery at bedside. Neurology 55:1621–1626
Stiasny-Kolster K, Mayer G, Schäfer S et al (2007) The REM sleep behavior disorder screening questionnaire—a new diagnostic instrument. Mov Disord 22:2386–2393. https://doi.org/10.1002/mds.21740
Saito S, Ayabe-Kanamura S, Takashima Y et al (2006) Development of a smell identification test using a novel stick-type odor presentation kit. Chem Senses 31:379–391. https://doi.org/10.1093/chemse/bjj042
Baba T, Takeda A, Kikuchi A et al (2011) Association of olfactory dysfunction and brain. Metabolism in Parkinson’s disease. Mov Disord 26:621–628. https://doi.org/10.1002/mds.23602
Iijima M, Kobayakawa T, Saito S et al (2008) Smell identification in Japanese Parkinson’s disease patients: using the odor stick identification test for Japanese subjects. Intern Med 47:1887–1892. https://doi.org/10.2169/internalmedicine.47.1345
Ogihara H, Kobayashi M, Nishida K et al (2011) Applicability of the cross-culturally modified University of Pennsylvania Smell Identification Test in a Japanese population. Am J Rhinol Allergy 25:404–410. https://doi.org/10.2500/ajra.2011.25.3658
Freeman R, Wieling W, Axelrod FB et al (2011) Consensus statement on the definition of orthostatic hypotension, neurally mediated syncope and the postural tachycardia syndrome. Clin Auton Res 21:69–72. https://doi.org/10.1007/s10286-011-0119-5
Dale AM, Fischl B, Sereno MI (1999) Cortical surface-based analysis I. Segmentation and surface reconstruction. Neuroimage 9:179–194
Fischl B, Van Der Kouwe A, Destrieux C et al (2004) Automatically parcellating the human cerebral cortex. Cereb Cortex 14:11–22. https://doi.org/10.1093/cercor/bhg087
Fischi B, Salat DH, Brusa E (2002) Neurotechnique whole brain segmentation: automated labeling of neuroanatomical structures in the human brain. Neuron 33:341–355
Rahayel S, Gaubert M, Postuma RB et al (2019) Brain atrophy in Parkinson’s disease with polysomnographyconfirmed REM sleep behavior disorder. Sleep 42:1–12. https://doi.org/10.1093/sleep/zsz062
Yoon EJ, Monchi O (2021) Probable REM sleep behavior disorder is associated with longitudinal cortical thinning in Parkinson’s disease. NPJ Parkinsons Dis 7:19. https://doi.org/10.1038/s41531-021-00164-z
Benjamini Y, Hochberg Y (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B 57:289–300
Wei K, Tran T, Chu K et al (2019) White matter hypointensities and hyperintensities have equivalent correlations with age and CSF β-amyloid in the nondemented elderly. Brain Behav 9:e01457. https://doi.org/10.1002/brb3.1457
Lee H, Mackenzie IRA, Beg MF et al (2022) White-matter abnormalities in presymptomatic GRN and C9orf72 mutation carriers. Brain Commun 5:fcac333. https://doi.org/10.1093/braincomms/fcac333
Shin NY, Park YW, Yoo SW et al (2021) Adverse effects of hypertension, supine hypertension, and perivascular space on cognition and motor function in PD. NPJ Parkinsons Dis 7:69. https://doi.org/10.1038/s41531-021-00214-6
Palma JA, Redel-Traub G, Porciuncula A et al (2020) The impact of supine hypertension on target organ damage and survival in patients with synucleinopathies and neurogenic orthostatic hypotension. Parkinsonism Relat Disord 75:97–104. https://doi.org/10.1016/j.parkreldis.2020.04.011
Campabadal A, Segura B, Junque C et al (2019) Comparing the accuracy and neuroanatomical correlates of the UPSIT-40 and the Sniffin’ Sticks test in REM sleep behavior disorder. Parkinsonism Relat Disord 65:197–202. https://doi.org/10.1016/j.parkreldis.2019.06.013
Acknowledgements
We are grateful to our hospital's ward doctors. This work was supported by the Jikei University Research Fund and a Grant-in-Aid for JSPS Research Fellows (202212840) to T. Shiraishi.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
All authors declare no financial or non-financial competing interests.
Ethical approval
Approval was received from the Regional Ethics and Hospital Management Committee of Jikei University School of Medicine. Written informed consent was obtained from all subjects prior to study participation according to the Declaration of Helsinki.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shiraishi, T., Yoshimaru, D., Umehara, T. et al. Interactive effect of orthostatic hypotension on gray matter atrophy associated with hyposmia and RBD in de novo Parkinson’s disease. J Neurol 270, 5924–5934 (2023). https://doi.org/10.1007/s00415-023-11934-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-11934-5