Abstract
Background
Monoaminergic network dysfunction may have a role in multiple sclerosis (MS) fatigue pathogenesis.
Objective
To investigate modifications of fatigue severity and resting state (RS) functional connectivity (FC) in monoaminergic networks of 45 fatigued MS patients after different symptomatic treatments.
Methods
Patients were randomly, blindly assigned to fampridine (n = 15), amantadine (n = 15) or placebo (n = 15) treatment and underwent clinical and 3T-MRI evaluations at baseline (t0) and week 4 (w4), i.e. after four weeks of treatment. Fifteen healthy controls (HC) were enrolled. Dopamine-, noradrenaline- and serotonin-related RS FC was assessed by PET-guided constrained independent component analysis.
Results
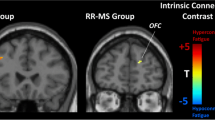
At t0, MS patients showed widespread monoamine-related RS FC abnormalities. At w4, fatigue scores decreased in all groups (p = range < 0.001–0.002). Concomitantly, fampridine and amantadine patients showed increased insular RS FC in dopamine-related and noradrenaline-related networks (p < 0.001, uncorrected). Amantadine patients also showed increased RS FC of anterior cingulate cortex in dopamine-related and noradrenaline-related networks (p < 0.001, uncorrected). Placebo patients showed increased precuneus/middle cingulate RS FC in the noradrenaline-related network (p < 0.001, uncorrected). In fampridine and placebo patients, just tendencies towards correlations between RS FC and fatigue modifications were found.
Conclusions
In MS patients, specific RS FC modifications in PET-guided monoaminergic networks were observed, concomitantly with fatigue improvements following treatment.
Trial registration number
EudraCT 2010-023678-38.


Similar content being viewed by others
Data availability
The dataset used and analysed during the current study is available from the corresponding author on reasonable request.
References
Filippi M, Bar-Or A, Piehl F, Preziosa P, Solari A, Vukusic S, Rocca MA (2018) Multiple sclerosis. Nat Rev Dis Primers 4:43
Krupp LB, Alvarez LA, LaRocca NG, Scheinberg LC (1988) Fatigue in multiple sclerosis. Arch Neurol 45:435–437
DeLuca J, Genova HM, Capili EJ, Wylie GR (2009) Functional neuroimaging of fatigue. Phys Med Rehabil Clin N Am 20:325–337
Bertoli M, Tecchio F (2020) Fatigue in multiple sclerosis: Does the functional or structural damage prevail? Mult Scler 26:1809–1815
Arm J, Ribbons K, Lechner-Scott J, Ramadan S (2019) Evaluation of MS related central fatigue using MR neuroimaging methods: scoping review. J Neurol Sci 400:52–71
Nourbakhsh B, Revirajan N, Morris B, Cordano C, Creasman J, Manguinao M, Krysko K, Rutatangwa A, Auvray C, Aljarallah S, Jin C, Mowry E, McCulloch C, Waubant E (2021) Safety and efficacy of amantadine, modafinil, and methylphenidate for fatigue in multiple sclerosis: a randomised, placebo-controlled, crossover, double-blind trial. Lancet Neurol 20:38–48
Generali JA, Cada DJ (2014) Amantadine: multiple sclerosis-related fatigue. Hosp Pharm 49:710–712
Smith KJ, Felts PA, John GR (2000) Effects of 4-aminopyridine on demyelinated axons, synapses and muscle tension. Brain 123(Pt 1):171–184
Yang TT, Wang L, Deng XY, Yu G (2017) Pharmacological treatments for fatigue in patients with multiple sclerosis: a systematic review and meta-analysis. J Neurol Sci 380:256–261
Rocca MA, Valsasina P, Colombo B, Martinelli V, Filippi M (2021) Cortico-subcortical functional connectivity modifications in fatigued multiple sclerosis patients treated with fampridine and amantadine. Eur J Neurol 28:2249–2258
Lin QH, Liu J, Zheng YR, Liang H, Calhoun VD (2010) Semiblind spatial ICA of fMRI using spatial constraints. Hum Brain Mapp 31:1076–1088
Carotenuto A, Valsasina P, Preziosa P, Mistri D, Filippi M, Rocca MA (2022) Monoaminergic network abnormalities: a marker for multiple sclerosis-related fatigue and depression. J Neurol Neurosurg Psychiatry 94(2):94–101
Carotenuto A, Wilson H, Giordano B, Caminiti SP, Chappell Z, Williams SCR, Hammers A, Silber E, Brex P, Politis M (2020) Impaired connectivity within neuromodulatory networks in multiple sclerosis and clinical implications. J Neurol 267:2042–2053
Kurtzke JF (1983) Rating neurologic impairment in multiple sclerosis: an expanded disability status scale (EDSS). Neurology 33:1444–1452
Montgomery SA, Asberg M (1979) A new depression scale designed to be sensitive to change. Br J Psychiatry 134:382–389
Flachenecker P, Kumpfel T, Kallmann B, Gottschalk M, Grauer O, Rieckmann P, Trenkwalder C, Toyka KV (2002) Fatigue in multiple sclerosis: a comparison of different rating scales and correlation to clinical parameters. Mult Scler 8:523–526
Rooney S, McFadyen DA, Wood DL, Moffat DF, Paul PL (2019) Minimally important difference of the fatigue severity scale and modified fatigue impact scale in people with multiple sclerosis. Mult Scler Relat Disord 35:158–163
Chard DT, Jackson JS, Miller DH, Wheeler-Kingshott CA (2010) Reducing the impact of white matter lesions on automated measures of brain gray and white matter volumes. J Magn Reson Imaging 32:223–228
Smith SM, Zhang Y, Jenkinson M, Chen J, Matthews PM, Federico A, De Stefano N (2002) Accurate, robust, and automated longitudinal and cross-sectional brain change analysis. Neuroimage 17:479–489
Hesse S, Moeller F, Petroff D, Lobsien D, Luthardt J, Regenthal R, Becker GA, Patt M, Thomae E, Seese A, Meyer PM, Bergh FT, Sabri O (2014) Altered serotonin transporter availability in patients with multiple sclerosis. Eur J Nucl Med Mol Imaging 41:827–835
Garcia-Gomez FJ, Lojo-Ramirez J, Huertas I, Garcia-Solis D (2018) Elaboración de una plantilla de SPM para la normalización de imágenes de PET con 18F-DOPA. Imagen Diagnostica 9:23–25
Schmidt E, Schinke C, Rullmann M, Luthardt J, Becker GA, Haars S, Stoppe M, Lobsien D, Hoffmann KT, Sabri O, Hesse S, Then BF (2020) Changes of central noradrenaline transporter availability in immunotherapy-naive multiple sclerosis patients. Sci Rep 10:14651
Group TCMR (1987) A randomized controlled trial of amantadine in fatigue associated with multiple sclerosis. The Canadian MS Research Group. Can J Neurol Sci 14:273–278
Stankoff B, Waubant E, Confavreux C, Edan G, Debouverie M, Rumbach L, Moreau T, Pelletier J, Lubetzki C, Clanet M, French Modafinil Study G (2005) Modafinil for fatigue in MS: a randomized placebo-controlled double-blind study. Neurology 64:1139–1143
Capone F, Collorone S, Cortese R, Di Lazzaro V, Moccia M (2020) Fatigue in multiple sclerosis: the role of thalamus. Mult Scler 26:6–16
Marchesi O, Vizzino C, Meani A, Conti L, Riccitelli GC, Preziosa P, Filippi M, Rocca MA (2020) Fatigue in multiple sclerosis patients with different clinical phenotypes: a clinical and magnetic resonance imaging study. Eur J Neurol 27:2549–2560
Calabrese M, Rinaldi F, Grossi P, Mattisi I, Bernardi V, Favaretto A, Perini P, Gallo P (2010) Basal ganglia and frontal/parietal cortical atrophy is associated with fatigue in relapsing-remitting multiple sclerosis. Mult Scler 16:1220–1228
Engstrom M, Flensner G, Landtblom AM, Ek AC, Karlsson T (2013) Thalamo-striato-cortical determinants to fatigue in multiple sclerosis. Brain Behav 3:715–728
Filippi M, Rocca MA, Colombo B, Falini A, Codella M, Scotti G, Comi G (2002) Functional magnetic resonance imaging correlates of fatigue in multiple sclerosis. Neuroimage 15:559–567
Hidalgo de la Cruz M, d’Ambrosio A, Valsasina P, Pagani E, Colombo B, Rodegher M, Falini A, Comi G, Filippi M, Rocca MA (2018) Abnormal functional connectivity of thalamic sub-regions contributes to fatigue in multiple sclerosis. Mult Scler 24:1183–1195
Rocca MA, Meani A, Riccitelli GC, Colombo B, Rodegher M, Falini A, Comi G, Filippi M (2016) Abnormal adaptation over time of motor network recruitment in multiple sclerosis patients with fatigue. Mult Scler 22:1144–1153
Kostic VS, Tomic A, Jecmenica-Lukic M (2016) The pathophysiology of fatigue in Parkinson’s disease and its pragmatic management. Mov Disord Clin Pract 3:323–330
Pavese N, Metta V, Bose SK, Chaudhuri KR, Brooks DJ (2010) Fatigue in Parkinson’s disease is linked to striatal and limbic serotonergic dysfunction. Brain 133:3434–3443
Politis M, Loane C (2011) Serotonergic dysfunction in Parkinson’s disease and its relevance to disability. Sci World J 11:1726–1734
Rocca MA, Agosta F, Colombo B, Mezzapesa DM, Falini A, Comi G, Filippi M (2007) fMRI changes in relapsing-remitting multiple sclerosis patients complaining of fatigue after IFNβ-1a injection. Hum Brain Mapp 28:373–382
Genova HM, Rajagopalan V, Deluca J, Das A, Binder A, Arjunan A, Chiaravalloti N, Wylie G (2013) Examination of cognitive fatigue in multiple sclerosis using functional magnetic resonance imaging and diffusion tensor imaging. PLoS One 8:e78811
Zhou F, Gong H, Chen Q, Wang B, Peng Y, Zhuang Y, Zee CS (2016) Intrinsic functional plasticity of the thalamocortical system in minimally disabled patients with relapsing-remitting multiple sclerosis. Front Hum Neurosci 10:2
Manjaly ZM, Harrison NA, Critchley HD, Do CT, Stefanics G, Wenderoth N, Lutterotti A, Muller A, Stephan KE (2019) Pathophysiological and cognitive mechanisms of fatigue in multiple sclerosis. J Neurol Neurosurg Psychiatry 90:642–651
Salamone PC, Esteves S, Sinay VJ, Garcia-Cordero I, Abrevaya S, Couto B, Adolfi F, Martorell M, Petroni A, Yoris A, Torquati K, Alifano F, Legaz A, Cassara FP, Bruno D, Kemp AH, Herrera E, Garcia AM, Ibanez A, Sedeno L (2018) Altered neural signatures of interoception in multiple sclerosis. Hum Brain Mapp 39:4743–4754
Chen MH, DeLuca J, Genova HM, Yao B, Wylie GR (2020) Cognitive fatigue is associated with altered functional connectivity in interoceptive and reward pathways in multiple sclerosis. Diagnostics (Basel) 10(11):930
Benedetti F (2014) Placebo effects: from the neurobiological paradigm to translational implications. Neuron 84:623–637
Piedimonte A, Benedetti F, Carlino E (2015) Placebo-induced decrease in fatigue: evidence for a central action on the preparatory phase of movement. Eur J Neurosci 41:492–497
Funding
This work has been partially supported by grants from Italian Ministry of Healthy (grant GR-2008-1138784).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
M.A. Rocca received consulting fees from Biogen, Bristol Myers Squibb, Eli Lilly, Janssen, Roche; and speaker honoraria from Bayer, Biogen, Bristol Myers Squibb, Bromatech, Celgene, Genzyme, Merck Healthcare Germany, Merck Serono SpA, Novartis, Roche, and Teva. She receives research support from the MS Society of Canada and Fondazione Italiana Sclerosi Multipla. She is Associate Editor for Multiple Sclerosis and Related Disorders. P. Valsasina received speaker honoraria from Biogen Idec. M.T. Lamanna and B. Colombo have nothing to disclose. V. Martinelli received travel support and speaker’s honoraria for activities with Biogen Idec, Merck Serono, Bayer, Teva Pharma, Novartis, Almirall and Genzyme. Prof. Filippi is Editor-in-Chief of the Journal of Neurology, Associate Editor of Human Brain Mapping, Neurological Sciences, and Radiology; received compensation for consulting services from Alexion, Almirall, Biogen, Merck, Novartis, Roche, Sanofi; speaking activities from Bayer, Biogen, Celgene, Chiesi Italia SpA, Eli Lilly, Genzyme, Janssen, Merck-Serono, Neopharmed Gentili, Novartis, Novo Nordisk, Roche, Sanofi, Takeda, and TEVA; participation in Advisory Boards for Alexion, Biogen, Bristol-Myers Squibb, Merck, Novartis, Roche, Sanofi, Sanofi-Aventis, Sanofi-Genzyme, Takeda; scientific direction of educational events for Biogen, Merck, Roche, Celgene, Bristol-Myers Squibb, Lilly, Novartis, Sanofi-Genzyme; he receives research support from Biogen Idec, Merck-Serono, Novartis, Roche, Italian Ministry of Health, and Fondazione Italiana Sclerosi Multipla.
Ethical approval
Approval was received from the local ethical standards committee on human experimentation of IRCCS Ospedale San Raffaele (registration number: GR-2008-1138784); all subjects signed written informed consent before study participation.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Rocca, M.A., Valsasina, P., Lamanna, M.T. et al. Functional connectivity modifications in monoaminergic circuits occur in fatigued MS patients treated with fampridine and amantadine. J Neurol 270, 4697–4706 (2023). https://doi.org/10.1007/s00415-023-11858-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-11858-0