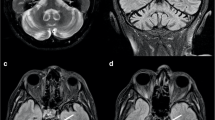
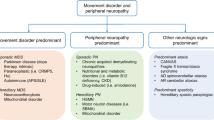
Abstract
With complicated conditions and a large number of potentially causative genes, the diagnosis of a patient with complex inherited peripheral neuropathies (IPNs) is challenging. To provide an overview of the genetic and clinical features of 39 families with complex IPNs from central south China and to optimize the molecular diagnosis approach to this group of heterogeneous diseases, a total of 39 index patients from unrelated families were enrolled, and detailed clinical data were collected. TTR Sanger sequencing, hereditary spastic paraplegia (HSP) gene panel, and dynamic mutation detection in spinocerebellar ataxia (SCAs) were performed according to the respective additional clinical features. Whole-exome sequencing (WES) was used in patients with negative or unclear results. Dynamic mutation detection in NOTCH2NLC and RCF1 was applied as a supplement to WES. As a result, an overall molecular diagnosis rate of 89.7% was achieved. All 21 patients with predominant autonomic dysfunction and multiple organ system involvement carried pathogenic variants in TTR, among which nine had c.349G > T (p.A97S) hotspot variants. Five out of 7 patients (71.4%) with muscle involvement harbored biallelic pathogenic variants in GNE. Five out of 6 patients (83.3%) with spasticity reached definite genetic causes in SACS, KIF5A, BSCL2, and KIAA0196, respectively. NOTCH2NLC GGC repeat expansions were identified in all three cases accompanied by chronic coughing and in one patient accompanied by cognitive impairment. The pathogenic variants, p.F284S and p.G111R in GNE, and p.K4326E in SACS, were first reported. In conclusion, transthyretin amyloidosis with polyneuropathy (ATTR-PN), GNE myopathy, and neuronal intranuclear inclusion disease (NIID) were the most common genotypes in this cohort of complex IPNs. NOTCH2NLC dynamic mutation testing should be added to the molecular diagnostic workflow. We expanded the genetic and related clinical spectrum of GNE myopathy and ARSACS by reporting novel variants.


Similar content being viewed by others
Data availability
The original contributions presented in the study are included in the article, and further inquiries can be directed to the corresponding authors.
References
Carroll AS, Burns J, Nicholson G, Kiernan MC, Vucic S (2019) Inherited neuropathies. Semin Neurol 39(5):620–639. https://doi.org/10.1055/s-0039-1693006
Rossor AM, Tomaselli PJ, Reilly MM (2016) Recent advances in the genetic neuropathies. Curr Opin Neurol 29(5):537–548
Rossor AM, Carr AS, Devine H et al (2017) Peripheral neuropathy in complex inherited diseases: an approach to diagnosis. J Neurol Neurosurg Psychiatry 88(10):846–863. https://doi.org/10.1136/jnnp-2016-313960
Mathis S, Goizet C, Tazir M et al (2015) Charcot-Marie-Tooth diseases: an update and some new proposals for the classification. J Med Genet 52(10):681–690. https://doi.org/10.1136/jmedgenet-2015-103272
Laurá M, Pipis M, Rossor AM, Reilly MM (2019) Charcot-Marie-Tooth disease and related disorders: an evolving landscape. Curr Opin Neurol 32(5):641–650. https://doi.org/10.1097/WCO.0000000000000735
Tian Y, Wang JL, Huang W et al (2019) Expansion of human-specific GGC repeat in neuronal intranuclear inclusion disease-related disorders. Am J Hum Genet 105(1):166–176. https://doi.org/10.1016/j.ajhg.2019.05.013
Cortese A, Simone R, Sullivan R et al (2019) Biallelic expansion of an intronic repeat in RFC1 is a common cause of late-onset ataxia. Nat Genet 51(4):649–658. https://doi.org/10.1038/s41588-019-0372-4
Yang HL, Jiang L, Pan HX et al (2021) Assessment of the association between NUS1 variants and essential tremor. Neurosci Lett 740:135441. https://doi.org/10.1016/j.neulet.2020.135441
Martindale JE (2017) Diagnosis of spinocerebellar ataxias caused by trinucleotide repeat expansions. Curr Protoc Hum Genet 92:1–22. https://doi.org/10.1002/cphg.30
Wang JL, Cao L, Li XH et al (2011) Identification of PRRT2 as the causative gene of paroxysmal kinesigenic dyskinesias. Brain 134(Pt 12):3493–3501. https://doi.org/10.1093/brain/awr289
Richards S, Aziz N, Bale S et al (2015) Standards and guidelines for the interpretation of sequence variants: a joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet Med 17(5):405–424. https://doi.org/10.1038/gim.2015.30
Xie Y, Lin Z, Liu L et al (2021) Genotype and phenotype distribution of 435 patients with Charcot–Marie–Tooth disease from central south China. Eur J Neurol 00:1–10. https://doi.org/10.1111/ene.15024
Adams D, Ando Y, Beirão JM et al (2021) Expert consensus recommendations to improve diagnosis of ATTR amyloidosis with polyneuropathy. J Neurol 268(6):2109–2122. https://doi.org/10.1007/s00415-019-09688-0
Cortese A, Vegezzi E, Lozza A et al (2017) Diagnostic challenges in hereditary transthyretin amyloidosis with polyneuropathy: avoiding misdiagnosis of a treatable hereditary neuropathy. J Neurol Neurosurg Psychiatry 88(5):457–458. https://doi.org/10.1136/jnnp-2016-315262
Hawkins PN, Ando Y, Dispenzeri A et al (2015) Evolving landscape in the management of transthyretin amyloidosis. Ann Med 47(8):625–638. https://doi.org/10.3109/07853890.2015.1068949
Rapezzi C, Quarta CC, Obici L et al (2013) Disease profile and differential diagnosis of hereditary transthyretin-related amyloidosis with exclusively cardiac phenotype: an Italian perspective. Eur Heart J 34(7):520–528. https://doi.org/10.1093/eurheartj/ehs123
Castaño A, Drachman BM, Judge D, Maurer MS (2015) Natural history and therapy of TTR-cardiac amyloidosis: emerging disease-modifying therapies from organ transplantation to stabilizer and silencer drugs. Heart Fail Rev 20(2):163–178. https://doi.org/10.1007/s10741-014-9462-7
Chen Z, Koh JS, Saini M et al (2021) Hereditary transthyretin amyloidosis- clinical and genetic characteristics of a multiracial south-east asian cohort in Singapore. J Neuromuscul Dis 8(4):723–733. https://doi.org/10.3233/JND-210656
Liu YT, Yen YJ, Ricardo F et al (2019) Biophysical characterization and modulation of Transthyretin Ala97Ser. Ann Clin Transl Neurol 6(10):1961–1970. https://doi.org/10.1002/acn3.50887
Carrillo N, Malicdan MC, Huizing M (2018) GNE myopathy: etiology, diagnosis, and therapeutic challenges. Neurotherapeutics 15(4):900–914. https://doi.org/10.1007/s13311-018-0671-y
Liu X, Zhang Y, Zhang S et al (2022) Different electrophysiology patterns in GNE myopathy. Orphanet J Rare Dis 17(1):206. https://doi.org/10.1186/s13023-022-02355-0
Nuria C, May CM, Petcharat L et al (2021) Safety and efficacy of N-acetylmannosamine (ManNAc) in patients with GNE myopathy: an open-label phase 2 study. Genet Med 23:2067–2075. https://doi.org/10.1038/s41436-021-01259-x
Celeste FV, Vilboux T, Ciccone C et al (2021) Mutation update for GNE gene variants associated with GNE myopathy. Hum Mutat 35(8):915–926. https://doi.org/10.1002/humu.22583
Zhao J, Wang Z, Hong D et al (2015) Mutational spectrum and clinical features in 35 unrelated mainland Chinese patients with GNE myopathy. J Neurol Sci 354(1–2):21–26. https://doi.org/10.1016/j.jns.2015.04.028
Tian Y, Zhou L, Gao J et al (2022) Clinical features of NOTCH2NLC-related neuronal intranuclear inclusion disease. J Neurol Neurosurg Psychiatry 93(12):1289–1298. https://doi.org/10.1136/jnnp-2022-329772
Jiao B, Zhou L, Zhou Y et al (2020) Identification of expanded repeats in NOTCH2NLC in neurodegenerative dementias. Neurobiol Aging 89:142.e1-142.e7
Yu J, Deng J, Guo X et al (2021) The GGC repeat expansion in NOTCH2NLC is associated with oculopharyngodistal myopathy type 3. Brain 144(6):1819–1832
Ogasawara M, Iida A, Kumutpongpanich T et al (2020) CGG expansion in NOTCH2NLC is associated with oculopharyngodistal myopathy with neurological manifestations. Acta Neuropathol Commun 8(1):204
Fang P, Yu Y, Yao S et al (2020) Repeat expansion scanning of the NOTCH2NLC gene in patients with multiple system atrophy. Ann Clin Transl Neurol 7(4):517–526
Ma D, Tan YJ, Ng ASL et al (2020) Association of NOTCH2NLC repeat expansions with Parkinson disease. JAMA Neurol 77(12):1559–1563
Shi CH, Fan Y, Yang J et al (2021) NOTCH2NLC Intermediate-length repeat expansions are associated with Parkinson disease. Ann Neurol 89(1):182–187
Sun QY, Xu Q, Tian Y et al (2021) Expansion of GGC repeat in the human-specific NOTCH2NLC gene is associated with essential tremor. Brain 143(1):222–233
Ng ASL, Lim WK, Xu Z et al (2020) NOTCH2NLC GGC repeat expansions are associated with sporadic essential tremor: variable disease expressivity on long-term follow-up. Ann Neurol 88(3):614–618
Yuan Y, Liu Z, Hou X et al (2020) Identification of GGC repeat expansion in the NOTCH2NLC gene in amyotrophic lateral sclerosis. Neurology 95(24):e3394–e3405
Okubo M, Doi H, Fukai R et al (2021) GGC repeat expansion of NOTCH2NLC in adult patients with leukoencephalopathy. Ann Neurol 86(6):962–968
Liao YC, Chang FP, Huang HW et al (2021) GGC repeat expansion of NOTCH2NLC in Taiwanese patients with inherited neuropathies. Neurology 98(2):e199–e206. https://doi.org/10.1212/WNL.0000000000013008
Yu J, Luan XH, Yu M et al (2021) GGC repeat expansions in NOTCH2NLC causing a phenotype of distal motor neuropathy and myopathy. Ann Clin Transl Neurol 8(6):1330–1342. https://doi.org/10.1002/acn3.51371
Acknowledgements
The authors would like to thank the patients and their family members for their participation in this study.
Funding
This study was supported by the National Natural Science Foundation of China (8177366 and 82001338), the China International Medical Foundation (CIMF-Z-2016-20-1801), the Hunan Provincial Natural Science Foundation (2021JJ40934), and Changsha Municipal Natural Science Foundation (kq2014260).
Author information
Authors and Affiliations
Contributions
RZ was responsible for the concept, design, and manuscript editing; MW and HY performed the data analysis and the original draft preparation; ZL, XL, LL, SH, HZ, XZ, QX performed the data acquisition and analysis; JW, SZ, and BT supervised the study and reviewed the manuscript. The version to be published has been read and approved by all the authors, and each author believes the manuscript represents honest work.
Corresponding author
Ethics declarations
Conflicts of interest
None declared.
Ethical approval
This study conformed with the World Medical Association Declaration of Helsinki and was approved by the ethics committee of the Third Xiangya Hospital (No. 2020-S035). Written informed consent was obtained from all participants or their first-grade relatives.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, M., Yang, H., Lin, Z. et al. The genetic and clinical spectrum in a cohort of 39 families with complex inherited peripheral neuropathies. J Neurol 270, 4959–4967 (2023). https://doi.org/10.1007/s00415-023-11821-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-11821-z