Abstract
Introduction
Progressive supranuclear palsy (PSP) and corticobasal syndrome (CBS) are progressive neurodegenerative syndromes characterised by Parkinsonism with additional features including cognitive dysfunction, falls, and oculomotor abnormalities. Understanding the epidemiology of these conditions is critical to planning for future service provision.
Methods
We conducted a systematic review of studies reporting incidence and prevalence of CBS and PSP. A search of the PubMed and EMBASE data bases was conducted from their date of inception to 13th July 2021. Meta-analysis of studies sharing similar methodologies was carried out to generate estimated pooled prevalence and incidence.
Results
We found 32 studies meeting our criteria for inclusion. There were 20 studies with data on prevalence and 12 with incidence data of PSP. Prevalence of CBS was reported in eight studies while seven studies reported incidence. Reported estimates of prevalence for PSP ranged from 1.00 (0.9–1.1) to 18 (8–28) per 100,000 while prevalence rates for CBS ranged from 0.83 (0.1–3.0) to 25 (0–59). Incidence rates for PSP and CBS respectively ranged from 0.16 (0.07–0.39) to 2.6 per 100,000 person-years and 0.03 (0–0.18) to 0.8 (0.4–1.3) per 100,000 person-years. A random effects model meta-analysis of studies with similar methodologies yielded a pooled prevalence estimate for PSP of 6.92 (4.33–11.06, I2 = 89%, τ2 = 0.3907) and 3.91 (2.03–7.51, I2 = 72%, τ2 = 0.2573) per 100,000 for CBS.
Conclusion
Studies of the epidemiology of PSP and CBS report highly heterogeneous findings. There is a need for further studies using rigorous phenotyping and the most recent diagnostic criteria to understand the true burden of these conditions.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Progressive supranuclear palsy (PSP) and corticobasal syndrome (CBS) are progressive neurodegenerative syndromes which present with parkinsonism and a variety of additional features [1]. There is considerable clinical and neuropathological overlap between the two conditions [2, 3]. Both are associated with the proliferation of the four-repeat (4R) isoform tau, although a significant degree of pathological heterogeneity is recognised in both conditions [4, 5]. Initially described in 1964, the classic PSP phenotype (Richardson’s syndrome) is characterised by a supranuclear vertical gaze palsy, pseudobulbar palsy, and axial rigidity [6]. However, a broad range of phenotypes have since been recognised and the most recent diagnostic criteria recognise eight distinct phenotypes including a PSP-CBS overlap syndrome [7]. CBS is associated with an combination of asymmetrical cortical and extrapyramidal signs. Apraxia, myoclonus, and dystonia are commonly seen [3]. While some epidemiological studies have used the term corticobasal degeneration (CBD) we have used the term CBS throughout, as denoting the clinical syndrome. We have reserved the term CBD for the pathological diagnosis.
Epidemiological data on CBS and PSP are limited due to the relative rarity of the conditions; their protean presentation and the degree of pathological heterogeneity which they exhibit. However, an accurate understanding of the burden of disease is important for planning of medical services, provision of care, and the administration of disease modifying treatments. Previous studies have provided systematic analysis of the prevalence of PSP and CBS [8]. In this study, we aim to supplement and expand on previous studies by drawing together data on the prevalence and incidence of PSPS and CBS. Therefore, we performed a systematic review and meta-analysis to review and synthesise the available evidence with the aim of appraising the existing literature and identifying areas in need of future study.
Methods
Search strategy
A systematic review was conducted according to a pre-determined protocol based on the PRISMA statement for systematic review and meta-analysis (see Supplementary Table 3). A search of PubMed and EMBASE from their date of inception to 13th July 2021 was performed using the search strategies detailed below (Table 1). The search was restricted to studies in English. The study protocol was registered with the PROSPERO registry before the initial review of the titles and abstracts (crd.york.ac.uk/PROSPERO/; registration number: CRD 42021266193) [9].
Study selection
Studies were screened by the first author (SL). Where it was unclear whether to include a study, the senior author (SOD) acted as arbiter. No automated tools were used in the selection of studies. For the purposes of the systematic review portion of the study, we adopted broad inclusion criteria defined using the CoCoPop model (Supplementary Table 1) [10]. We considered any study which attempted to enumerate neurodegenerative disease in a general population and which reported cases of PSP or CBS as part of that enumeration. Studies were excluded if cases of PSP or CBS were not reported or could not be calculated from reported data. We considered a prevalence study to consist of a study of a general population which attempted to identify cases of PSP/CBS in a comprehensive way (for example by multiple source referrals within a defined population, screening of medical records within a region, or a standardised system of notification) and reported prevalence or reported data such that prevalence could be calculated. We considered an incidence study to consist of a study of a general population in which a comprehensive attempt was made to ascertain all cases of PSP and CBS within a defined time-period, and incidence was reported, or data reported such that incidence could be calculated. We included in the systematic review studies that identified PSP and CBS in a variety of ways: (i) in person examination of patients with identification based on published guidelines or diagnosis by expert clinicians, (ii) identification from notes and letters detailing patient diagnosis and (iii) notification to registries by treating physicians, (iv) association of patients with an appropriate code or entry in database and registry studies. Papers and conference abstracts meeting these criteria were selected for full-text review. The references of included articles and relevant review articles were hand searched for additional articles. All additional articles were evaluated in the same manner as those identified in the initial search. When studies were identified in both abstract and paper format, the paper format was included as representing a more complete data set.
Data extraction and study quality
Data was extracted from articles using a standardised data collection form by the first authors (SL) in cases where data the relevant data was unclear or questionable, these cases were reviewed with the senior author (SOD). Studies were recorded as reporting data on prevalence and/or incidence of PSP, CBS, or both. If multiple articles reported data on the same study population the most comprehensive data were utilised. In cases where multiple studies reported prevalence or incidence data for the same geographic area at different time points all studies were included as separate estimates. Demographic data extracted included age, sex, and study location. Diagnostic criteria used, data on phenotypic subtypes, and methods of case recruitment and identification were recorded. The number of cases of PSP or CBS, and the size of the population studied, and/or person-years used to calculate incidence rates were extracted. Incidence and/or prevalence estimates from each study were extracted and reported per 100,000 population or 100,000 person-years as applicable. Study quality was assessed using the Joanna Briggs Inventory (Supplementary Table 4) [11].
Data analysis
In cases where prevalence or incidence was not reported but could be calculated from given data this was performed. 95% confidence intervals were not reported, these were calculated assuming a binomial distribution. All statistical analyses were carried out in R (version 4.1.1 the R Foundation for statistical computing, Vienna, Austria). The forestploter package was employed to produce unweighted forest plots for incidence and prevalence in existing studies. Reported figures for incidence and prevalence were sub grouped by region.
Meta-analysis was limited to a subset of studies with the following characteristics: (1) full articles, excluding abstracts, (2) were designed to investigate PSP/CBS, parkinsonism, Parkinson’s disease, or FTLD. Studies of dementia were excluded as they may fail to identify movement-predominant presentations, (3) data on individual cases accessed either by review of clinical notes or examination of participants (as opposed to databases or registries) (4) did not restrict recruitment by age beyond that required by diagnostic criteria (i.e. 40 years of age) (5) specified established diagnostic criteria/guidelines, excluding the 2017 MDS Diagnostic Criteria (these were used only in one study and the expansion of the phenotype recognised by these criteria makes meta-analysis unreliable), (6) studies based on sampling were excluded.
Number of cases (numerator) and population and/or person-years (denominator) in included studies were used to calculate pooled prevalence and incidence of PSP and CBS using a random effects model. Meta-analyses were conducted using the metafor package [12]. Heterogeneity was assessed by the X2 test on Cochrane’s Q statistic, which was quantified by I2, assuming I2 values of 25%, 50%, and 75% respectively representing low, medium, and high heterogeneity [13]. Outliers were identified using studentised residuals. We conducted a leave-one-out sensitivity analysis to explore the sensitivity of the pooled prevalence estimates to the exclusion of individual studies.
Results
Identification and description of studies
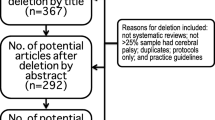
The search strategy yielded 1,650 citations (1,020 from EMBASE, 630 from PubMed) (Fig. 1) carried out between 1988 and 2021. Of the studies reviewed, 32 reporting the incidence and/or prevalence of PSP and/or CBS were included in our systematic review [14,15,16,17,18,19,20,21,22,23,24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45]. Nine studies reported data on both conditions. A variety of ascertainment methods, often overlapping within a single study, were used (see Table 2 for summary details of study contents and Supplementary Table 2 and 3 for more details, Supplementary Table 6 contains a complete list of included studies). Seven studies assessed populations of fewer than 20,000 people (Supplementary Table 1 and 2).
Studies reporting the prevalence of PSP and CBS
Prevalence data on PSP and/or CBS was reported in 23 studies. This included 20 papers and three conference abstracts. 15 studies reported prevalence data on PSP only, three reported on CBS only, and five reported data on both PSP and CBS. One paper reported prevalence figures for PSP in three nested populations using a “Russian doll” design, thus, there were 22 estimates of PSP prevalence. Studies identified PSP/CBS in the context of a range of conditions as follows: PSP/CBS specifically (n = 7), parkinsonism (n = 7), Parkinson’s disease (n = 5), dementia (n = 3), frontotemporal lobar degeneration (n = 1).
17 articles and three abstracts provided information on PSP prevalence (see Fig. 2(A) and Supplementary Table 1). Reported estimates of population prevalence ranged from 1.00 (0.9–1.1) [22] to 18 (8–28) [32] per 100,000. Age-adjusted prevalence was reported in 10 populations with estimates ranging from 1.39 to 17.26 per 100,000 [16, 39]. In a study of a large US database prevalence peaked in the age range 75–79, with a prevalence of < 2 per 100,000 before the age of 60 [45]. A UK study demonstrated a peak prevalence in the 70–74 age range [38], however, a large Swiss study reported a peak prevalence of 72.3 per 100,000 in the oldest age group (80–89 years) [40]. Prevalence data by sex was available for seven studies [16, 21, 22, 27, 32, 39, 40]. Reported prevalence figures for men ranged from 1.00 (0.9–1.1) to 25 (8–43), and from 1.0 (0.8–1.1) to 17.76 (10.38–30.39) per 100,000 for women. One study reported data on phenotypic subtypes. Richardson’s syndrome was the most common phenotype (16/25, 64%), followed by PSP-parkinsonism (3/25, 12%), and PSP-progressive gait freezing (2/25, 8%) [14]. Two studies of PSP prevalence in Yonago City, Japan conducted 11 years apart demonstrated an increase in the recorded prevalence of PSP in this area during that time (from 5.82 to 17.9 per 100,000) [27, 39]. Meta-analysis of nine studies yielded a pooled prevalence estimate of 6.92 (4.33–11.06) per 100,000, with a high degree of heterogeneity (I2 = 89%, τ2 = 0.3907, P < 0.01) (Fig. 4(A)). One study [38] was a significant outlier (studentised residual = – 2.00), exclusion of this study resulted in a prevalence estimate of 8.29 (5.50–12.49). Pooled prevalence figures of relevant subgroups are available in Fig. 5(A). Pooled prevalence for three studies from Japan yielded a prevalence of 12.85 (10.97–19.68) (I2 = 76%, τ2 = 0.2969, P = 0.02) while six studies carried out in Europe yielded a pooled prevalence of 4.95 (3.19–7.68) (I2 = 81%, τ2 = 0.1789, P < 0.01).
Unweighted forest plots of crude prevalence and incidence rates for: A PSP and B CBS prevalence per 100,000 population, C PSP and D CBS incidence per 100,000 patient-years with studies reported by region. a = Study restricted to population > 55, b = Study restricted to population > 65, c = Study restricted to population < 65, d = Study restricted to population > 50. In cases where rates were not reported but could be calculated from provided data (cases and population and/or person-years) this was done
Eight studies (seven papers and one abstract) reported the prevalence of CBS (Table 2 and Supplementary Table 2). Reported prevalence figures ranged from 0.77 (0.02–4.32) to 25 (0–59) per 100,000. Age adjusted prevalence was available for two studies of CBS with rates of 3.2 (1.5–4.8) and 6 (0–12) per 100,000 [32, 40]. Prevalence by sex were available for two studies, with male vs female prevalence figures of 2.6 vs 3.3[40], 16 vs 3 per 100,000 [32]. Meta-analysis of three studies of the prevalence of CBS yielded a pooled prevalence 3.91 (2.03- 7.51, I2 = 72%, τ2 = 0.2573, P = 0.03).
Studies reporting the incidence of PSP and CBS
The incidence of PSP/CBS was reported in 12 studies (PSP only n = 5, PSP and CBS n = 7, no studies with CBS alone), including 11 papers and one abstract. Two additional studies reported an indirectly calculated incidence rate, using prevalence and survival data and were not included in the analysis of incidence [16, 21]. Included studies reported on the incidence of parkinsonism (n = 6), PSP and CBS specifically (n = 2), dementia (n = 1), FTLD (n = 2), and a single study reported on a number of uncommon neurological diseases. Age-adjusted rates were available for four studies of PSP [14, 31, 40, 45] and two for CBS [14, 40]. Three of these studies used direct standardisation to a national population with details of standardisation not reported in one study.
Reported incidence rates for PSP ranged from 0.3 to 2.6 per 100,000 person-years (see Fig. 2(C) and Supplementary Table 2), while age-adjusted rates ranged from 0.12 to 2.0 per 100,000 person-years. Incidence rates in populations > 60 years ranged from 0.84 (0.27–1.96) [14] to 9.7 per 100,000 person [18] years reported. Incidence of PSP was reported in comparable age increments in three studies (Fig. 3). PSP incidence was reported by sex in eight studies. Incidence among men ranged from 0.17–3.9 per 100,000 person-years and from 0.1–0.8 per 100,000 person-years among women. Incidence of PSP was reported in studies of the population in Olmsted County, Minnesota over two time periods, the first study reported on the period 1976–1990 produced an incidence of 0.9 per 100,000 person-years [18], while a study of the 1991–2005 population using the 2017 MDS criteria returned an incidence rate of 2.6 per 100,000 person-years [43]. Meta-analysis of eight studies yielded a pooled incidence of 0.81 per 100,000 person-years (0.48–1.37, I2 = 86%, τ2 = 0.4847, p < 0.01, Fig. 4(C)). One study [14] was a significant outlier (studentised residual = – 2.57); when this was excluded from the analysis, a pooled incidence of 0.99 (0.65–1.51) was found.
The incidence of CBS ranged from 0.03 [14] to 0.8 per 100,000 person-years [18] (Fig. 2(D) and Supplementary Table 2). Age-adjusted rates were available for two studies of CBS with rates of 0.02 (0.01–0.12) [14] and 1.4 (0.3–2.4) [40] per 100,000 person-years reported. Meta-analysis of six studies yielded an incidence of 0.28 per 100,000 person-years (0.15–0.52, I2 = 70%, τ2 = 0.3896, p < 0.01, Fig. 4(D)). Pooled incidence figures for relevant subgroups are reported in Fig. 5(B) and (C).
Discussion
A true and reliable description of the epidemiology of PSP and CBS remains elusive. This study reviews and synthesises existing data on the prevalence and incidence of these disorders, but significant challenges remain. Estimates of prevalence and incidence confirm that PSP and CBS are uncommon conditions but there is a high degree of heterogeneity in the existing literature. Studies generally accord with the reported preponderance of male cases in PSP, although one high quality study demonstrated an equal prevalence of male and female cases. PSP is more common in older age bands, where these are reported. Only one population-based study of PSP prevalence reported phenotypic subtypes, suggesting a predominance of Richardson’s Syndrome (64%).
There are fewer studies of the epidemiology of CBS than PSP. Most studies support the view that CBS is significantly rarer than PSP [10] although it is worth noting that in one well-designed study of the prevalence of CBS was the same as that of PSP.[38] Since the conclusion of our study selection in July 2021 several studies have added to the field. A study of a large health insurance database in Israel has provided registry data on a large population. The epidemiological advantages of this database include the fact that enrolment in a healthcare database is mandatory in Israel, it provides stable database population, and this single insurance database includes 25% of the national population [46]. Estimates from this study accord reasonably well with the most widely cited data with prevalence estimates of 5.3 per 100,000 and incidence of 1 per 100,000 for PSP as well as our pooled estimate of European studies.
Study design and methodology
Registry and database-based studies have generally reported lower prevalence and incidence numbers than studies which have sought referral from a comprehensive array of sources [44, 45]. The 2001 study by Nath et al. demonstrates the limitations of a database and registry-based approach, with a Russian-doll model demonstrating higher prevalence figures with more granular and comprehensive methods of ascertainment [22]. While database and registry studies provide the sample size necessary to study rare diseases, they may lack the ability to accurately capture cases in disease where reliable biomarkers are lacking, such as PSP and CBS. However, studies with more complete ascertainment may be limited in size, identifying relatively few cases in smaller populations and generating estimates of incidence and prevalence with very broad confidence intervals.
Changes in prevalence and incidence of PSP and CBS over time
The degree of heterogeneity between studies makes inferences regarding longitudinal trajectories of prevalence difficult, however, two studies conducting in Yonago City, Japan 11 years apart, demonstrated an increase in prevalence over that time, from 5.82 (1.78–9.86) to 17.9 (12.12–26.42) per 100,000 [27, 39]. This increase remained statistically significant following adjustment for age and sex. However, although PSP-RS was numerically increased this did not reach statistical significance, suggesting that increased recognition of non-RS subtypes might have played a role in this apparent trend. This may be driven by several factors which are likely to be replicated in other settings, such as an aging population and increased recognition of PSP subtypes, and some which are unique to the local context, such as the inclusion of PSP in the Japanese Specified Treatment Research Program in 2003. However, the later of these studies is a relative outlier in terms of reported prevalence and, as geographical clusters of PSP are known to occur, may not be generalisable. Studies of the population of Olmsted County, Minnesota demonstrated an incidence rate which was essentially unchanged in the periods 1976–1990 and 1991–2005. Two studies published since the conclusion of our data collection two studies have added more information on secular trends in the prevalence and incidence of PSP and CBS. Swallow et al. have demonstrated stable incidences of PSP and CBS over a 20 year period in a Northern European population [47]. Logroscino et al., whose earlier study is included in our analysis have published updated data on the incidence of a range of fronto-temporal lobar degeneration syndromes in nine countries with combined estimated incidence of PSP and CBS of 0.51 (0.22–1.20) per 100,000 person-years [48].
Impact of diagnostic criteria on studies of PSP/CBS
Evolution in diagnostic criteria may explain a significant proportion of apparent change over time. Many existing studies used the NINDS-SPSP criteria for PSP which, despite excellent specificity, has poor sensitivity for Richardson’s syndrome early in the disease course and does not account well for variant presentations, which may be the initial presentation in up to 60–75% of PSP cases [49, 50]. Therefore, these studies may underestimate the true prevalence and incidence of PSP. In one study, only 25% of cases in which PSP was demonstrated pathologically received a PSP diagnosis at initial evaluation [50]. Two studies conducted in Olmsted County, Minnesota provide insight into the impact of the 2017 MDS criteria on PSP incidence: in a 2013 study using the NINDS-SPSP criteria the incidence of PSP was 0.9, in a 2020 study of the same population using the 2017 criteria, an incidence rate of 2.6 was reported [34, 43]. The most common criteria used in the diagnosis of CBS were the 2013 Armstrong criteria which despite usefully expanding the range of phenotypes seen in CBS may have limited specificity [51].
Regional and socio-economic difference
In common with many other neurological conditions the epidemiology of PSP/CBS is much more extensively studied in high-income countries with Europe, America, and Japan being particular areas of activity with low-and-middle-income-countries (LMICs) underrepresented. As the prevalence of neurodegenerative disease increases with age, and the populations of many LMICs are aging[52], there is an urgent need to expand the study of PSP/CBS to these areas. In addition, of particular interest is the relatively high prevalence of PSP reported in studies from Japan, and further studies from that region may provide clues to genetic and environmental risk factors [32, 39].
Study strengths and limitations
This study comprehensively collates and analyses the existing literature on the incidence and prevalence of PSP/CBS. A recent systematic review and meta-analysis of the prevalence of PSP and CBS by Swallow et al. identified similar issues with heterogeneity of study design and a wide range of reported prevalence figures [8]. Our study complements this work, including a discussion on data from existing registry based studies and data and meta-analysis on incidence of PSP and CBS. Our study has several limitations. There is a high degree of heterogeneity between the identified studies, which limits the ability to generalise from recorded observations, especially when the possibility of geographic or genetic clustering may occur. Many of the studies are small and underpowered to estimate the prevalence of rare conditions such as PSP and CBS. Nevertheless, significant heterogeneity remains even when such studies are excluded. Several studies restricted their recruitment by age [17, 26, 33] and these studies reported prevalence of PSP/CBS higher than unrestricted studies, consistent with an increasing prevalence of neurodegenerative disease in older age. The relatively small number of studies makes identifying trends problematic. All studies of PSP and CBS are limited by the degree of phenotypic heterogeneity and the absence of reliable antemortem biomarkers of the underlying neuropathology, and any inference derived from meta-analysis will be susceptible to similar limitations. PSP has also demonstrated a tendency to cluster for environmental reasons [53, 54] and genetic reasons [55] which may further confound attempts to generalise from existing studies, especially from small sample sizes. Lastly, because of the recognition of the wider phenotype associated with PSP and CBD pathology it is difficult to extrapolate epidemiological estimates from studies which use older diagnostic criteria.
Future directions in the epidemiology of PSP/CBS
Future epidemiological studies of PSP and CBS face several challenges. Studies of PSP/CBS require relatively large populations to generate accurate estimates. Accurate detection of variant presentations of PSP and CBS will require collaboration of multiple specialties, including psychiatry of older age, neurology, medicine for the elderly, and family physicians. Given the possibility of evolution of clinical syndromes and consequent diagnostic instability studies should include a range of neurodegenerative diagnoses and longitudinal follow-up. Large, prospective registries of neurodegenerative diseases with comprehensive reporting requirements and including detailed clinical information might offer the best hope for accurate population level data, although the logistic and data management challenges would be formidable. In the future, with the development of fluid and imaging based biomarkers, epidemiology informed by in-vivo molecular profiles may improve the reliability of epidemiological data [56].
Conclusion
Data regarding the prevalence and incidence of PSP and CBS are rare, with information on CBS particularly limited. Existing estimates are varied, influenced by study design and size. Evolving diagnostic criteria constitute a particular challenge in the study of these conditions. There is a need for further epidemiological studies, adequately powered to assess the epidemiology of rare diseases, with robust methods of clinical assessment using the most recent diagnostic criteria.
References
Pantelyat A (2022) Progressive supranuclear palsy and corticobasal syndrome. Continuum (Minneap Minn) 28:1364–1378. https://doi.org/10.1212/CON.0000000000001158
Rowe JB, Holland N, Rittman T (2021) Progressive supranuclear palsy: diagnosis and management. Pract Neurol 21:376–383. https://doi.org/10.1136/practneurol-2020-002794
Constantinides VC, Paraskevas GP, Paraskevas PG, Stefanis L, Kapaki E (2019) Corticobasal degeneration and corticobasal syndrome: a review. Clin Park Relat Disord 1:66–71. https://doi.org/10.1016/j.prdoa.2019.08.005
Dickson DW (1999) Neuropathologic differentiation of progressive supranuclear palsy and corticobasal degeneration. J Neurol 246(Suppl 2):II6-15. https://doi.org/10.1007/BF03161076
Boeve BF, Maraganore DM, Parisi JE, Ahlskog JE, Graff-Radford N, Caselli RJ, Dickson DW, Kokmen E, Petersen RC (1999) Pathologic heterogeneity in clinically diagnosed corticobasal degeneration. Neurology 53:795–800. https://doi.org/10.1212/wnl.53.4.795
Steele JC, Richardson JC, Olszewski J (1964) Progressive supranuclear palsy. A heterogenous degeneration involving the brain stem, basal ganglia and cerebellum with vertical gaze and pseudobulbar palsy, nuchal dystonia and dementia. Arch Neurol 10:333–359. https://doi.org/10.1001/archneur.1964.00460160003001
Hoglinger GU, Respondek G, Stamelou M, Kurz C, Josephs KA, Lang AE, Mollenhauer B, Muller U, Nilsson C, Whitwell JL, Arzberger T, Englund E, Gelpi E, Giese A, Irwin DJ, Meissner WG, Pantelyat A, Rajput A, van Swieten JC, Troakes C, Antonini A, Bhatia KP, Bordelon Y, Compta Y, Corvol JC, Colosimo C, Dickson DW, Dodel R, Ferguson L, Grossman M, Kassubek J, Krismer F, Levin J, Lorenzl S, Morris HR, Nestor P, Oertel WH, Poewe W, Rabinovici G, Rowe JB, Schellenberg GD, Seppi K, van Eimeren T, Wenning GK, Boxer AL, Golbe LI, Litvan I, Movement Disorder Society-endorsed PSPSG (2017) Clinical diagnosis of progressive supranuclear palsy: the movement disorder society criteria. Mov Disord 32:853–864. https://doi.org/10.1002/mds.26987
Swallow DMA, Zheng CS, Counsell CE (2022) Systematic review of prevalence studies of progressive supranuclear palsy and corticobasal syndrome. Mov Disord Clin Pract 9:604–613. https://doi.org/10.1002/mdc3.13489
Lyons S, O'Dowed S (2021) The prevalence and incidence of progressive supranuclear palsy and corticobasal syndrome: a systematic review. PROSPERO CRD420021266193. https://www.crd.york.ac.uk/PROSPERO/display_record.php?RecordID=266193
Munn Z, Moola S, Lisy K, Riitano D, Tufanaru C (2015) Methodological guidance for systematic reviews of observational epidemiological studies reporting prevalence and cumulative incidence data. Int J Evid Based Health 13:147–153. https://doi.org/10.1097/XEB.0000000000000054
Moola S MZ, Tufanaru C, Aromataris E, Sears K, Sfetcu R, Currie M, Qureshi R, Mattis P, Lisy K, Mu P-F. (2017) Chapter 7: Systematic reviews of etiology and risk. In: Aromataris E, Munn Z (Ed). Joanna Briggs Reviewer’s Manual. The Joanna Briggs Institute. Available from http://reviewersmanual.joannabriggs.org/.
Viechtbauer W (2010) Conducting meta-analyses in R with the metafor Package. J Stat Softw 36:1–48
Higgins JP, Thompson SG (2002) Quantifying heterogeneity in a meta-analysis. Stat Med 21:1539–1558. https://doi.org/10.1002/sim.1186
Winter Y, Bezdolnyy Y, Katunina E, Avakjan G, Reese JP, Klotsche J, Oertel WH, Dodel R, Gusev E (2010) Incidence of Parkinson’s disease and atypical parkinsonism: Russian population-based study. Mov Disord 25:349–356. https://doi.org/10.1002/mds.22966
Radhakrishnan K, Thacker AK, Maloo JC, Gerryo SE, Mousa ME (1988) Descriptive epidemiology of some rare neurological diseases in Benghazi, Libya. Neuroepidemiology 7:159–164. https://doi.org/10.1159/000110150
Golbe LI, Davis PH, Schoenberg BS, Duvoisin RC (1988) Prevalence and natural history of progressive supranuclear palsy. Neurology 38:1031–1034. https://doi.org/10.1212/wnl.38.7.1031
de Rijk MC, Breteler MM, Graveland GA, Ott A, Grobbee DE, van der Meche FG, Hofman A (1995) Prevalence of Parkinson’s disease in the elderly: the Rotterdam study. Neurology 45:2143–2146. https://doi.org/10.1212/wnl.45.12.2143
Bower JH, Maraganore DM, McDonnell SK, Rocca WA (1997) Incidence of progressive supranuclear palsy and multiple system atrophy in Olmsted county, Minnesota, 1976 to 1990. Neurology 49:1284–1288. https://doi.org/10.1212/wnl.49.5.1284
Wermuth L, Joensen P, Bünger N, Jeune B (1997) High prevalence of Parkinson’s disease in the Faroe Islands. Neurology 49:426–432. https://doi.org/10.1212/wnl.49.2.426
Chio A, Magnani C, Schiffer D (1998) Prevalence of Parkinson’s disease in Northwestern Italy: comparison of tracer methodology and clinical ascertainment of cases. Mov Disord 13:400–405. https://doi.org/10.1002/mds.870130305
Schrag A, Ben-Shlomo Y, Quinn NP (1999) Prevalence of progressive supranuclear palsy and multiple system atrophy: a cross-sectional study. Lancet 354:1771–1775. https://doi.org/10.1016/s0140-6736(99)04137-9
Nath U, Ben-Shlomo Y, Thomson RG, Morris HR, Wood NW, Lees AJ, Burn DJ (2001) The prevalence of progressive supranuclear palsy (Steele-Richardson-Olszewski syndrome) in the UK. Brain 124:1438–1449. https://doi.org/10.1093/brain/124.7.1438
Yamada T, Hattori H, Miura A, Tanabe M, Yamori Y (2001) Prevalence of Alzheimer’s disease, vascular dementia and dementia with lewy bodies in a Japanese population. Psychiatry Clin Neurosci 55:21–25. https://doi.org/10.1046/j.1440-1819.2001.00779.x
Harvey RJ, Skelton-Robinson M, Rossor MN (2003) The prevalence and causes of dementia in people under the age of 65 years. J Neurol Neurosurg Psychiatry 74:1206–1209. https://doi.org/10.1136/jnnp.74.9.1206
Zhang ZX, Anderson DW, Huang JB, Li H, Hong X, Wei J, Yang EL, Maraganore DM (2003) Prevalence of Parkinson’s disease and related disorders in the elderly population of greater Beijing. China Mov Disord 18:764–772. https://doi.org/10.1002/mds.10445
Bergareche A, De La Puente E, Lopez de Munain A, Sarasqueta C, de Arce A, Poza JJ, Marti-Masso JF (2004) Prevalence of Parkinson’s disease and other types of Parkinsonism. A door-to-door survey in Bidasoa. Spain J Neurol 251:340–345. https://doi.org/10.1007/s00415-004-0333-3
Kawashima M, Miyake M, Kusumi M, Adachi Y, Nakashima K (2004) Prevalence of progressive supranuclear palsy in Yonago. Japan Mov Disord 19:1239–1240. https://doi.org/10.1002/mds.20149
Tan LC, Venketasubramanian N, Hong CY, Sahadevan S, Chin JJ, Krishnamoorthy ES, Tan AK, Saw SM (2004) Prevalence of Parkinson disease in Singapore: Chinese vs Malays vs Indians. Neurology 62:1999–2004. https://doi.org/10.1212/01.wnl.0000128090.79756.10
Wermuth L, Bech S, Petersen MS, Joensen P, Weihe P, Grandjean P (2008) Prevalence and incidence of Parkinson’s disease in The Faroe Islands. Acta Neurol Scand 118:126–131. https://doi.org/10.1111/j.1600-0404.2007.00991.x
Tartari JPSC, Bauso DJ, Giunta D, Rojas JI, Cristiano E (2010) Prevalence of parkinsonism in a health maintenance organization from Buenos Aires City. Mov Disord 25:S264. https://doi.org/10.1002/mds.23162
Linder J, Stenlund H, Forsgren L (2010) Incidence of Parkinson’s disease and parkinsonism in northern Sweden: a population-based study. Mov Disord 25:341–348. https://doi.org/10.1002/mds.22987
Osaki Y, Morita Y, Kuwahara T, Miyano I, Doi Y (2011) Prevalence of Parkinson’s disease and atypical parkinsonian syndromes in a rural Japanese district. Acta Neurol Scand. 124:182–187. https://doi.org/10.1111/j.1600-0404.2010.01442.x
Nakashita SWIK, Uemura Y, Yamawaki M, Tanaka K, Nakashima K (2011) Parkinsonism in a community dwelling elderly population sample in Japan. Eur J Neurol 18:545. https://doi.org/10.1111/ene.14306
Savica R, Grossardt BR, Bower JH, Ahlskog JE, Rocca WA (2013) Incidence and pathology of synucleinopathies and tauopathies related to parkinsonism. JAMA Neurol 70:859–866. https://doi.org/10.1001/jamaneurol.2013.114
Caslake R, Taylor K, Scott N, Harris C, Gordon J, Wilde K, Murray A, Counsell C (2014) Age-, and gender-specific incidence of vascular parkinsonism, progressive supranuclear palsy, and parkinsonian-type multiple system atrophy in North East Scotland: the PINE study. Parkinsonism Relat Disord 20:834–839. https://doi.org/10.1016/j.parkreldis.2014.04.013
Withall A, Draper B, Seeher K, Brodaty H (2014) The prevalence and causes of younger onset dementia in Eastern Sydney. Australia Int Psychogeriatr 26:1955–1965. https://doi.org/10.1017/S1041610214001835
Khedr EM, Fawi G, Abbas MA, Mohammed TA, El-Fetoh NA, Attar GA, Zaki AF (2015) Prevalence of parkinsonism and parkinson’s disease in Qena governorate/Egypt: a cross-sectional community-based survey. Neurol Res 37:607–618. https://doi.org/10.1179/1743132815Y.0000000020
Coyle-Gilchrist IT, Dick KM, Patterson K, Vazquez Rodriquez P, Wehmann E, Wilcox A, Lansdall CJ, Dawson KE, Wiggins J, Mead S, Brayne C, Rowe JB (2016) Prevalence, characteristics, and survival of frontotemporal lobar degeneration syndromes. Neurology 86:1736–1743. https://doi.org/10.1212/WNL.0000000000002638
Takigawa H, Kitayama M, Wada-Isoe K, Kowa H, Nakashima K (2016) Prevalence of progressive supranuclear palsy in Yonago: change throughout a decade. Brain Behav 6:e00557. https://doi.org/10.1002/brb3.557
Fleury V, Brindel P, Nicastro N, Burkhard PR (2018) Descriptive epidemiology of parkinsonism in the canton of Geneva, Switzerland. Parkinsonism Relat Disord 54:30–39. https://doi.org/10.1016/j.parkreldis.2018.03.030
Calvo-Perxas L, Belchi O, Turon-Estrada A, Van Eendenburg C, Linares M, Vinas M, Manzano A, Turro-Garriga O, Vilalta-Franch J, Garre-Olmo J (2019) Incidence and characteristics of uncommon dementia subtypes: results from 10 years of clinical surveillance by the registry of dementia of girona. Alzheimers Dement. 15:917–926. https://doi.org/10.1016/j.jalz.2019.03.017
Logroscino G, Piccininni M, Binetti G, Zecca C, Turrone R, Capozzo R, Tortelli R, Battista P, Bagoj E, Barone R, Fostinelli S, Benussi L, Ghidoni R, Padovani A, Cappa SF, Alberici A, Borroni B (2019) Incidence of frontotemporal lobar degeneration in Italy: the salento-brescia registry study. Neurology 92:e2355–e2363. https://doi.org/10.1212/WNL.0000000000007498
Stang CD, Turcano P, Mielke MM, Josephs KA, Bower JH, Ahlskog JE, Boeve BF, Martin PR, Upadhyaya SG, Savica R (2020) Incidence and trends of progressive supranuclear palsy and corticobasal syndrome: a population-based study. J Parkinsons Dis 10:179–184. https://doi.org/10.3233/JPD-191744
Viscidi E, Morris H, Harrington A, Inuzaka Y, Li L, Eaton S, Orlovic M (2020) The epidemiology of progressive supranuclear palsy in the United Kingdom: evidence from the clinical practice research datalink GP online database (CPRD GOLD). Eur J Neurol 27:1–102. https://doi.org/10.1111/ene.14306
Viscidi E, Litvan I, Dam T, Juneja M, Li L, Krzywy H, Eaton S, Hall S, Kupferman J, Hoglinger GU (2021) Clinical Features of Patients With Progressive supranuclear palsy in an US insurance claims database. Front Neurol. 12:5718. https://doi.org/10.3389/fneur.2021.571800
Barer Y, Chodick G, Cohen R, Grabarnik-John M, Ye X, Zamudio J, Gurevich T (2022) Epidemiology of progressive supranuclear palsy: real world data from the second largest health plan in Israel. Brain Sci. https://doi.org/10.3390/brainsci12091126
Swallow DMA, Counsell CE (2022) Prevalence of progressive supranuclear palsy and corticobasal syndrome in Scotland. Neuroepidemiology 56:291–297. https://doi.org/10.1159/000525334
Logroscino G, Piccininni M, Graff C, Hardiman O, Ludolph AC, Moreno F, Otto M, Remes AM, Rowe JB, Seelaar H, Solje E, Stefanova E, Traykov L, Jelic V, Rydell MT, Pender N, Anderl-Straub S, Barandiaran M, Gabilondo A, Krüger J, Murley AG, Rittman T, van der Ende EL, van Swieten JC, Hartikainen P, Stojmenovic GM, Mehrabian S, Benussi L, Alberici A, Dell’Abate MT, Zecca C, Borroni B (2023) Incidence of syndromes associated with frontotemporal lobar degeneration in 9 European countries. JAMA Neurol 80:279–286. https://doi.org/10.1001/jamaneurol.2022.5128
Respondek G, Stamelou M, Kurz C, Ferguson LW, Rajput A, Chiu WZ, van Swieten JC, Troakes C, Al Sarraj S, Gelpi E, Gaig C, Tolosa E, Oertel WH, Giese A, Roeber S, Arzberger T, Wagenpfeil S, Hoglinger GU, Movement Disorder Society-endorsed PSPSG (2014) The phenotypic spectrum of progressive supranuclear palsy: a retrospective multicenter study of 100 definite cases. Mov Disord. 29:1758–1766. https://doi.org/10.1002/mds.26054
Respondek G, Kurz C, Arzberger T, Compta Y, Englund E, Ferguson LW, Gelpi E, Giese A, Irwin DJ, Meissner WG, Nilsson C, Pantelyat A, Rajput A, van Swieten JC, Troakes C, Josephs KA, Lang AE, Mollenhauer B, Muller U, Whitwell JL, Antonini A, Bhatia KP, Bordelon Y, Corvol JC, Colosimo C, Dodel R, Grossman M, Kassubek J, Krismer F, Levin J, Lorenzl S, Morris H, Nestor P, Oertel WH, Rabinovici GD, Rowe JB, van Eimeren T, Wenning GK, Boxer A, Golbe LI, Litvan I, Stamelou M, Hoglinger GU, Movement Disorder Society-Endorsed PSPSG (2017) Which ante mortem clinical features predict progressive supranuclear palsy pathology? Mov Disord. 32:995–1005. https://doi.org/10.1002/mds.27034
Alexander SK, Rittman T, Xuereb JH, Bak TH, Hodges JR, Rowe JB (2014) Validation of the new consensus criteria for the diagnosis of corticobasal degeneration. J Neurol Neurosurg Psychiatry 85:925–929. https://doi.org/10.1136/jnnp-2013-307035
Tan MP (2022) Healthcare for older people in lower and middle income countries. Age Aging 51(4):016. https://doi.org/10.1093/ageing/afac016
Caparros-Lefebvre D, Sergeant N, Lees A, Camuzat A, Daniel S, Lannuzel A, Brice A, Tolosa E, Delacourte A, Duyckaerts C (2002) Guadeloupean parkinsonism: a cluster of progressive supranuclear palsy-like tauopathy. Brain 125(Pt 4):801–811. https://doi.org/10.1093/brain/awf086
Park HK, Ilango SD, Litvan I (2021) Environmental risk factors for progressive supranuclear palsy. J Mov Disord 14:103–113. https://doi.org/10.14802/jmd.20173
Wen Y, Zhou Y, Jiao B, Shen L (2021) Genetics of progressive supranuclear palsy: a review. J Parkinsons Dis 11:93–105. https://doi.org/10.3233/JPD-202302
Lee EB (2018) Integrated neurodegenerative disease autopsy diagnosis. Acta Neuropathol 135:643–646. https://doi.org/10.1007/s00401-018-1827-x
Acknowledgements
Three of the authors of this paper are members of the European Reference Network for Rare Neurological Diseases – Project ID No 101085584.
Funding
Open Access funding provided by the IReL Consortium.
Author information
Authors and Affiliations
Contributions
SL: Conceptualisation, methodology, formal analysis, writing-original draft. DT: Methodology, writing- review and editing. TL: Conceptualisation, methodology, writing-review and editing. RW: Methodology, writing-review and editing. SO: Conceptualisation, methodology, supervision.
Corresponding author
Ethics declarations
Conflicts of interest
This research did not receive specific grants from funding agencies in the public, commercial, or not-for-profit sectors. Dr Lyons has been funded by a Fellowship from the Meath Foundation.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Lyons, S., Trépel, D., Lynch, T. et al. The prevalence and incidence of progressive supranuclear palsy and corticobasal syndrome: a systematic review and meta-analysis. J Neurol 270, 4451–4465 (2023). https://doi.org/10.1007/s00415-023-11791-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-023-11791-2