Abstract
Paraneoplastic neurologic syndromes (PNSs) are a wide spectrum of neurologic diseases characterized by different clinical features, associated with a neoplasia, and triggered by an immune-mediated process. In most cases, it is possible to detect specific neuronal antibodies and the Hu protein is one of the most frequently recognized intracellular antigens in patients with PNSs. Small-cell lung cancer is the most common cancer associated with PNSs, followed by urological, gynecological and hematological malignancies. Otherwise, extra-pulmonary small-cell carcinomas, including Merkel cell carcinoma (MCC), have been rarely described as related to PNSs. In this article we report, for the first time in the published literature, a case of anti-Hu antibody-related subacute sensory neuronopathy in association with MCC.


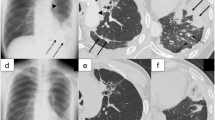
taken from the inguinal lymph node. Solid sheets of neoplastic cells with dark nuclei and high N/C ratio can be appreciated (A) in the hematoxylin and eosin staining. The tumoral cells resulted immunoreactive to cytokeratin AE1/AE3 (B) and neurofilaments (C), with a peculiar dot-like paranuclear pattern in both the stains. The cells appeared also positive for synaptophysin staining (D) with cytoplasmic reactivity


Similar content being viewed by others
References
Denny-Brown D (1948) Primary sensory neuropathy with muscular changes associated with carcinoma. J Neurol Neurosurg Psychiatry 11(2):73–87
Camdessanche JP, Jousserand G, Franques J et al (2012) A clinical pattern-based etiological diagnostic strategy for sensory neuronopathies: a French collaborative study. J Peripher Nerv Syst JPNS. 17(3):331–340
Graus F, Vogrig A, Muñiz-Castrillo S et al (2021) Updated diagnostic criteria for paraneoplastic neurologic syndromes. Neurol Neuroimmunol Neuroinflamm 8(4):e1014
Graus F, Keime-Guibert F, Reñe R et al (2001) Anti-Hu-associated paraneoplastic encephalomyelitis: analysis of 200 patients. Brain 124(Pt 6):1138–1148
Dubey D, Lennon VA, Gadoth A et al (2018) Autoimmune CRMP5 neuropathy phenotype and outcome defined from 105 cases. Neurology 90(2):e103–e110
Dubey D, Jitprapaikulsan J, Bi H et al (2019) Amphiphysin-IgG autoimmune neuropathy: a recognizable clinicopathologic syndrome. Neurology 93(20):e1873–e1880
Côté-Mantha E, Savard M (2012) Paraneoplastic anti-Hu syndrome associated with uterine tumor. Can J Neurol Sci 39:254–255
Sano T, Tanaka K, Ito N (2010) Anti-Hu-antibody-associated paraneoplastic neurological syndrome accompanying testicular cancer. Int J Urol 17(1):99
Lukacs S, Szabo N, Woodhams S (2012) Rare association of anti-Hu antibody positive paraneoplastic neurological syndrome and transitional cell bladder carcinoma. Case Rep Urol 2012:724940
Cowley A, Pascoe S (2011) Paraneoplastic subacute sensory neuronopathy in association with adenocarcinoma of the prostate. BMJ Case Rep 2011:bcr0420114077
Matsui T, Hori Y, Nagano H et al (2015) Poorly differentiated hepatocellular carcinoma accompanied by anti-Hu antibody-positive paraneoplastic peripheral neuropathy. Pathol Int 65(7):388–392
Graus F, Delattre JY, Antoine JC et al (2004) Recommended diagnostic criteria for paraneoplastic neurological syndromes. J Neurol Neurosurg Psychiatry 75(8):1135–1140
Stanoszek LM, Chan MP, Palanisamy N et al (2019) Neurofilament is superior to cytokeratin 20 in supporting cutaneous origin for neuroendocrine carcinoma. Histopathology 74(3):504–513
Walsh NM, Cerroni L (2021) Merkel cell carcinoma: a review. J Cutan Pathol 48(3):411–421
Sibley RK, Dehner LP, Rosai J (1985) Primary neuroendocrine (Merkel cell?) carcinoma of the skin I. A. clinicopathological and ultrastructural study of 43 cases. Am J Surg Pathol 9(2):95–108
Feng H, Shuda M, Chang Y, Moore PS (2008) Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science 319(5866):1096–1100
Heath M, Jaimes N, Lemos B et al (2008) Clinical characteristics of Merkel cell carcinoma at diagnosis in 195 patients: the AEIOU features. J Am Acad Dermatol 58(3):375–381
Paulson KG, Iyer JG, Blom A et al (2013) Systemic immune suppression predicts diminished Merkel cell carcinoma-specific survival independent of stage. J Invest Dermatol 133(3):642–646
Skelton HG, Smith KJ, Hitchcock CL et al (1997) Merkel cell carcinoma: analysis of clinical, histologic, and immunohistologic features of 132 cases with relation to survival. J Am Acad Dermatol 37(5 Pt 1):734–739
Llombart B, Monteagudo C, López-Guerrero JA et al (2005) Clinicopathological and immunohistochemical analysis of 20 cases of Merkel cell carcinoma in search of prognostic markers. Histopathology 46(6):622–634
Chan JK, Suster S, Wenig BM et al (1997) Cytokeratin 20 immunoreactivity distinguishes Merkel cell (primary cutaneous neuroendocrine) carcinomas and salivary gland small cell carcinomas from small cell carcinomas of various sites. Am J Surg Pathol 21(2):226–234
Simmons DB, Duginski TM, McClean JC et al (2016) Lambert-Eaton myasthenic syndrome and merkel cell carcinoma. Muscle Nerve 53(2):325–326
Gandhi L, Johnson BE (2006) Paraneoplastic syndromes associated with small cell lung cancer. J Natl Compr Canc Netw 4(6):631–638
Honnorat J, Antoine JC (2007) Paraneoplastic neurological syndromes. Orphanet J Rare Dis 4(2):22
Saltos A, Shafique M, Chiappori A (2020) Update on the biology, management, and treatment of small cell lung cancer (SCLC). Front Oncol 10:1074
Pavolucci L, Giannini G, Giannoccaro MP et al (2017) Paraneoplastic cerebellar degeneration and Lambert-Eaton myasthenia in a patient with Merkel cell carcinoma and voltage-gated calcium channel antibodies. Muscle Nerve 56(5):998–1000
Agostini A, Merli M, Avallone G et al (2021) Lambert-Eaton myasthenic syndrome and paraneoplastic cerebellar degeneration associated with Merkel cell carcinoma with unknown primary: a case report. Acta Derm Venereol 101(5):adv00452
Balegno S, Ceroni M, Corato M et al (2005) Antibodies to cerebellar nerve fibres in two patients with paraneoplastic cerebellar ataxia. Anticancer Res 25(5):3211–3214
Cher LM, Hochberg FH, Teruya J et al (1995) Therapy for paraneoplastic neurologic syndromes in six patients with protein a column immunoadsorption. Cancer 75(7):1678–1683
Shalhout SZ, Emerick KS, Sadow PM et al (2020) Regionally metastatic Merkel cell carcinoma associated with paraneoplastic anti-N-methyl-D-aspartate receptor encephalitis. Case Rep Oncol Med 2020:1257587
Moll JW, Antoine JC, Brashear HR et al (1995) Guidelines on the detection of paraneoplastic anti-neuronal-specific antibodies: report from the workshop to the fourth meeting of the international society of neuro-immunology on paraneoplastic neurological disease, held october 22–23, 1994, in Rotterdam. Neth Neurol 45(10):1937–1941
Dalmau J, Furneaux HM, Cordon-Cardo C, Posner JB (1992) The expression of the Hu (paraneoplastic encephalomyelitis/sensory neuronopathy) antigen in human normal and tumor tissues. Am J Pathol 141(4):881–886
Gultekin SH, Rosai J, Demopoulos A et al (2000) Hu immunolabeling as a marker of neural and neuroendocrine differentiation in normal and neoplastic human tissues: assessment using a recombinant anti-Hu fab fragment. Int J Surg Pathol 8:109–117
Titulaer MJ, Lang B, Verschuuren JJ (2011) Lambert-Eaton myasthenic syndrome: from clinical characteristics to therapeutic strategies. Lancet Neurol 10(12):1098–1107
Ahmadi Moghaddam P, Cornejo KM, Hutchinson L et al (2016) Complete spontaneous regression of Merkel cell carcinoma after biopsy: a case report and review of the literature. Am J Dermatopathol 38(11):e154–e158
Cirillo F (2015) Spontaneous regression of primitive Merkel cell carcinoma. Rare Tumors 7(4):5961
Pang C, Sharma D, Sankar T (2015) Spontaneous regression of Merkel cell carcinoma: a case report and review of the literature. Int J Surg Case Rep 7C:104–108
Lawrence LEB, Saleem A, Sahoo MK et al (2020) Is Merkel cell carcinoma of lymph node actually metastatic cutaneous Merkel cell carcinoma? Am J Clin Pathol 154(3):369–380
Sehgal IS, Kaur H, Dhooria S et al (2016) Extrapulmonary small cell carcinoma of lymph node: pooled analysis of all reported cases. World J Clin Oncol 7(3):308–320
Lemos BD, Storer BE, Iyer JG et al (2010) Pathologic nodal evaluation improves prognostic accuracy in Merkel cell carcinoma: analysis of 5823 cases as the basis of the first consensus staging system. J Am Acad Dermatol 63(5):751–761
Tarantola TI, Vallow LA, Halyard MY et al (2013) Unknown primary Merkel cell carcinoma: 23 new cases and a review. J Am Acad Dermatol 68(3):433–440
Kaufman HL, Russell J, Hamid O et al (2016) Avelumab in patients with chemotherapy-refractory metastatic Merkel cell carcinoma: a multicentre, single-group, open-label, phase 2 trial. Lancet Oncol 17(10):1374–1385
Emge DA, Cardones AR (2019) Updates on Merkel cell carcinoma. Dermatol Clin 37(4):489–503
Gill A, Perez MA, Perrone CM et al (2019) A case series of PD-1 inhibitor-associated paraneoplastic neurologic syndromes. J Neuroimmunol 334:576980
Raibagkar P, Ho D, Gunturu KS, Srinivasan J (2020) Worsening of anti-Hu paraneoplastic neurological syndrome related to anti-PD-1 treatment: case report and review of literature. J Neuroimmunol 341:577184
Matsuoka H, Kimura H, Koba H et al (2018) Nivolumab-induced limbic encephalitis with anti-Hu antibody in a patient with advanced pleomorphic carcinoma of the lung. Clin Lung Cancer 19(5):e597–e599
Iyer JG, Parvathaneni K, Bhatia S et al (2016) Paraneoplastic syndromes (PNS) associated with Merkel cell carcinoma (MCC): A case series of 8 patients highlighting different clinical manifestations. J Am Acad Dermatol 75(3):541–547
Hocar O, Poszepczynska-Guigné E, Faye O et al (2011) Severe necrotizing myopathy subsequent to Merkel cell carcinoma. Ann Dermatol Venereol 138(2):130–134
Batchelor TT, Platten M, Hochberg FH (1998) Immunoadsorption therapy for paraneoplastic syndromes. J Neurooncol 40(2):131–136
Bombelli F, Lispi L, Calabrò F et al (2015) Lambert-Eaton myasthenic syndrome associated to Merkel cell carcinoma: report of a case. Neurol Sci 36(8):1491–1492
Dohrn MF, Schöne U, Küppers C et al (2020) Immunoglobulins to mitigate paraneoplastic Lambert Eaton myasthenic syndrome under checkpoint inhibition in Merkel cell carcinoma. Neurol Res Pract 2:52
Eggers SD, Salomao DR, Dinapoli RP, Vernino S (2001) Paraneoplastic and metastatic neurologic complications of Merkel cell carcinoma. Mayo Clin Proc 76(3):327–330
Greenlee JE, Steffens JD, Clawson SA et al (2002) Anti-Hu antibodies in Merkel cell carcinoma. Ann Neurol 52(1):111–115
Lopez M-C, Pericay C, Agustí M et al (2004) Merkel cell carcinoma associated with a paraneoplastic neurological syndrome. Histopathology 44(6):628–629
Nguyen ND, Simmons DB, Bersabe AR et al (2019) Lambert-Eaton myasthenic syndrome and Merkel cell carcinoma. Cutis 103(5):E19-23
Queen D, Gu Y, Lopez A et al (2019) Paraneoplastic cerebellar ataxia in Merkel cell carcinoma of unknown primary. JAAD Case Rep 5(5):398–400
Sharobeam A, Ray J, Dong J, Chong V (2017) Subacute cerebellar degeneration due to a paraneoplastic phenomenon associated with metastatic Merkel cell carcinoma: a case report. Case Rep Oncol 10(2):764–768
Siau RT, Morris A, Karoo RO (2014) Surgery results in complete cure of Lambert-Eaton myasthenic syndrome in a patient with metastatic Merkel cell carcinoma. J Plast Reconstr Aesthet Surg JPRAS 67(6):e162–e164
Zhang C, Emery L, Lancaster E (2014) Paraneoplastic cerebellar degeneration associated with noncutaneous Merkel cell carcinoma. Neurol Neuroimmunol Neuroinflamm 1(2):e17
Funding
None.
Author information
Authors and Affiliations
Contributions
RM contributed to study design, literature review, data analyses and paper drafting. MC contributed to study design, data analyses and paper drafting. GA and FF contributed to data collection and paper drafting. FC contributed to study design and paper drafting. MF contributed to paper conception, supervised data analysis and interpretation, critically reviewed the paper.
Corresponding author
Ethics declarations
Conflicts of interest
No relevant competing interest was declared by the Authors.
Ethical approval
The patient gave explicit informed consent to report his clinical case.
Rights and permissions
About this article
Cite this article
Milani, R., Cannizzaro, M., Arrigoni, G. et al. Subacute sensory neuronopathy associated with Merkel cell carcinoma with unknown primary: a case report with literature review. J Neurol 269, 4080–4088 (2022). https://doi.org/10.1007/s00415-022-11116-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00415-022-11116-9